PY17X_0420500 alpha tubulin 1
Disruptability [+]
| Species | Disruptability | Reference | Submitter | |
|---|---|---|---|---|
| P. berghei ANKA |
Refractory |
RMgm-8 | Imported from RMgmDB | |
| P. berghei ANKA |
Possible |
PlasmoGEM (Barseq) | PlasmoGEM | |
| P. berghei ANKA |
Possible |
RMgm-4636 | Imported from RMgmDB | |
| P. berghei ANKA |
Possible |
RMgm-4637 | Imported from RMgmDB | |
Mutant phenotypes [+]
| Species | Stage | Phenotype | Reference | Submitter |
|---|---|---|---|---|
| P. berghei ANKA | Asexual |
No difference |
PlasmoGEM (Barseq) | PlasmoGEM |
| P. berghei ANKA | Asexual |
No difference |
RMgm-4636 | Imported from RMgmDB |
| P. berghei ANKA | Asexual |
No difference |
RMgm-4637 | Imported from RMgmDB |
| P. berghei ANKA | Gametocyte |
No difference |
RMgm-4636 | Imported from RMgmDB |
| P. berghei ANKA | Gametocyte |
No difference |
RMgm-4637 | Imported from RMgmDB |
| P. berghei ANKA | Ookinete |
No difference |
RMgm-4636 | Imported from RMgmDB |
| P. berghei ANKA | Ookinete |
No difference |
RMgm-4637 | Imported from RMgmDB |
| P. berghei ANKA | Oocyst |
Difference from wild-type |
RMgm-4636
Both a1-tubulin(-) parasite lines showed no defect in blood-stage growth or initial mosquito infection. However, no sporozoites were found in the midgut or salivary glands of the mosquito; thus, a main defect seems to occur at the oocyst stage. This defect could be completely rescued by complementing the gene in the a1-tubulin(-) parasite line.Deletion of a1-tubulin affects Plasmodium sporozoite formation during budding. |
Imported from RMgmDB |
| P. berghei ANKA | Oocyst |
Difference from wild-type |
RMgm-4637
Both a1-tubulin(-) parasite lines showed no defect in blood-stage growth or initial mosquito infection. However, no sporozoites were found in the midgut or salivary glands of the mosquito; thus, a main defect seems to occur at the oocyst stage. This defect could be completely rescued by complementing the gene in the a1-tubulin(-) parasite line.Deletion of a1-tubulin affects Plasmodium sporozoite formation during budding. |
Imported from RMgmDB |
| P. berghei ANKA | Sporozoite |
Difference from wild-type |
RMgm-4636
Both a1-tubulin(-) parasite lines showed no defect in blood-stage growth or initial mosquito infection. However, no sporozoites were found in the midgut or salivary glands of the mosquito; thus, a main defect seems to occur at the oocyst stage. This defect could be completely rescued by complementing the gene in the a1-tubulin(-) parasite line.Deletion of a1-tubulin affects Plasmodium sporozoite formation during budding. |
Imported from RMgmDB |
| P. berghei ANKA | Sporozoite |
Difference from wild-type |
RMgm-4637
Both a1-tubulin(-) parasite lines showed no defect in blood-stage growth or initial mosquito infection. However, no sporozoites were found in the midgut or salivary glands of the mosquito; thus, a main defect seems to occur at the oocyst stage. This defect could be completely rescued by complementing the gene in the a1-tubulin(-) parasite line.Deletion of a1-tubulin affects Plasmodium sporozoite formation during budding. |
Imported from RMgmDB |
Imaging data (from Malaria Metabolic Pathways)
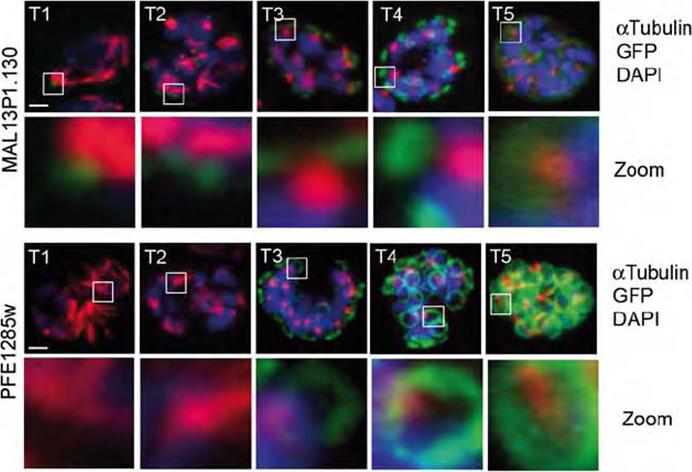
Co-localization of the TMD protein MAL13P1.130 and the alveolin PFE1285w with the disassembling spindle apparatus during schizogony (T1-5). Upper: While the mitotic spindles (T1, anti-tubulin, red) are still present, the cramp-like structure highlighted by the TMD protein MAL13P1.130-GFP is formed (T2). Lower: In contrast, the alveolin-defined structure only emerges after the spindles have retracted forming MTOC bundles (T3). Nuclei stained with DAPI. Scale bars, 2μm.Kono M, Herrmann S, Loughran NB, Cabrera A, Engelberg K, Lehmann C, Sinha D, Prinz B, Ruch U, Heussler V, Spielman T, Parkinson J, Gilberger TW. Evolution and Architecture of the Inner Membrane Complex in Asexual and Sexual Stages of the Malaria Parasite. Mol Biol Evol. 2012 29(9):2113-32.
See original on MMP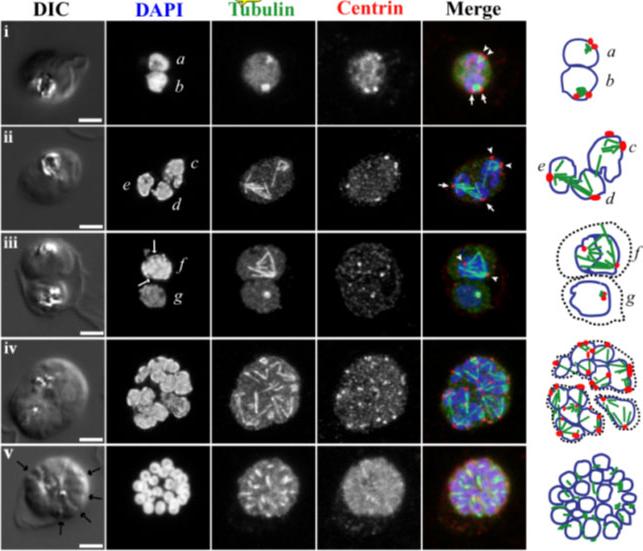
Fluorescence light microscopy images of mitotic 450 spindle during blood stage schizogony in P. falciparum parasites. 3D confocal microscopy views of blood-stage P.f. parasite cell morphology (DIC microscopy), nuclei (DAPI, blue in merged images), mitotic spindle (anti-alpha tubulin antibody, green in merged images), and mitotic spindle MTOCs (anti-PfCEN3 antibody in panels i, ii, iv, and v or anti-Cr Centrin1 antibody 20H5 in panel iii, red in merged images). Schematic cartoons are drawn for each example indicating spindle MTOCs (red circles), microtubules (green lines), and outlines of stained DNA (blue lines). Dotted black lines indicate separate parasites in multiply infected host cells. Panel i, a-b) Short mitotic spindles bounded by MTOCs. The spindle in nucleus b which resembles an oblong tubulin spot has a pole-to-pole distance that is approximately 1 micron long. Panels ii-iii, c-f) Larger spindle structures. In d and e, spindle extends across separate nuclear bodies. In f, a dark line is visible down the center of the DAPI-stained DNA near the spindle midzone (DAPI, arrows). Panel iv) Asynchronous schizont nuclei have spindles with different geometries in multiple stages of extension. Panel v) Segmented parasites after the final mitosis of schizogony. Furrows are visible between daughters (arrows), nuclei are condensed, and microtubules appear in the daughter cytoplasms. Panel i is a single confocal slice from a 3D series. Panels ii-v are maximum projection images of full 3D series. Panels ii-iv were processed with deconvolution software. Scale bar = 2 microns. Gerald N, Mahajan B, Kumar S. Mitosis in the human malaria parasite Plasmodium falciparum. Eukaryot Cell. 2011 10(4):474-82.
See original on MMP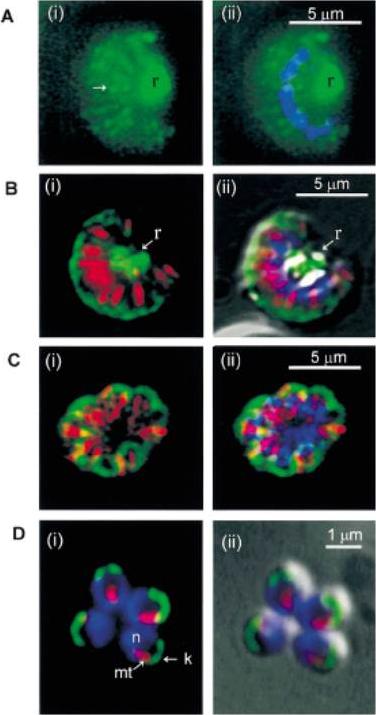
Fluorescent images of schizonts undergoing merogony (A–C), and four merozoites (D), labelled with antibodies to kinesin heavy chains (green) and a-tubulin (red), which labels the microtubules of f-MASTs, (merozoite assemblage of sub-pellicular mt) and DAPI nuclear stain (blue). A. Kinesin labelling is primarily associated with the residual body (r), but also radiates out in a series of linear arrays of discrete fluorescent points (Arrow) (i). Nuclei are shown by the DAPI staining (ii). B(i), kinesin labelling is seen at the periphery of the schizont, where the merozoite apices lie, and in the residual body (r), which is distinguished by the white glow of the pigment seen in the overlaid DIC image (ii). C(i), kinesin staining is apparent only at the periphery of the schizont where the merozoite apices are located, and is not apparent in the residual body area. D(i), the nuclei (n) are situated at the base of the merozoites, the f-MASTs (mt) run laterally, and the kinesin labelling (k) appears to be located close to the membrane at the apices, as judged by its proximity to the cell surface in the overlaid DIC image (ii).Fowler RE, Smith AM, Whitehorn J, Williams IT, Bannister LH, Mitchell GH. Microtubule associated motor proteins of Plasmodium falciparum merozoites. Mol Biochem Parasitol. 2001 117:187-200.
See original on MMP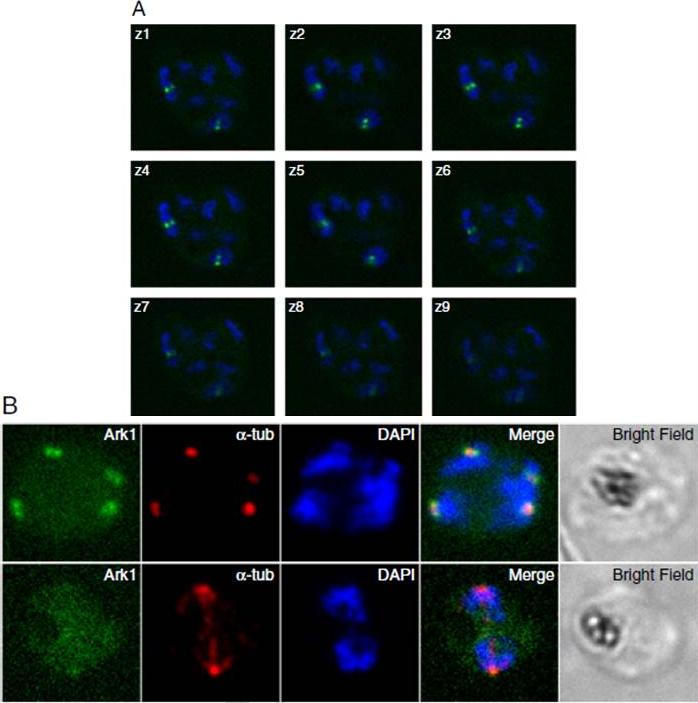
Pfark-1 expression in schizont stage P. falciparum parasites. A. Z-stacks live images of Pfark-1-GFP transgenic parasite undergoing schizogony showing the presence of pairs of dots (green) restricted to two out of seven nuclei (blue), reflecting the asynchrony of nuclear multiplication in schizont stage malaria parasites. B. Immunofluorescence assay of Pfark-1-GFP transgenic parasites. The upper panel shows that the Pfark-1-GFP protein is observed as pairs of dots (green) in close vicinity to DAPI-stained nuclear bodies (blue). Anti-α-tubulin antibody (DM1A) recognizes short intranuclear microtubule spindles (red) which appear between duplicated spindle pole bodies. The lower panel shows the absence of Pfark1-GFP protein at the spindle poles when fully developed spindle microtubules are visible.Doerig C, Reininger L. Nima- and Aurora-related kinases of malaria parasites. Biochim Biophys Acta. 2013 1834(7):1336-45. Copyright Elsevier
See original on MMP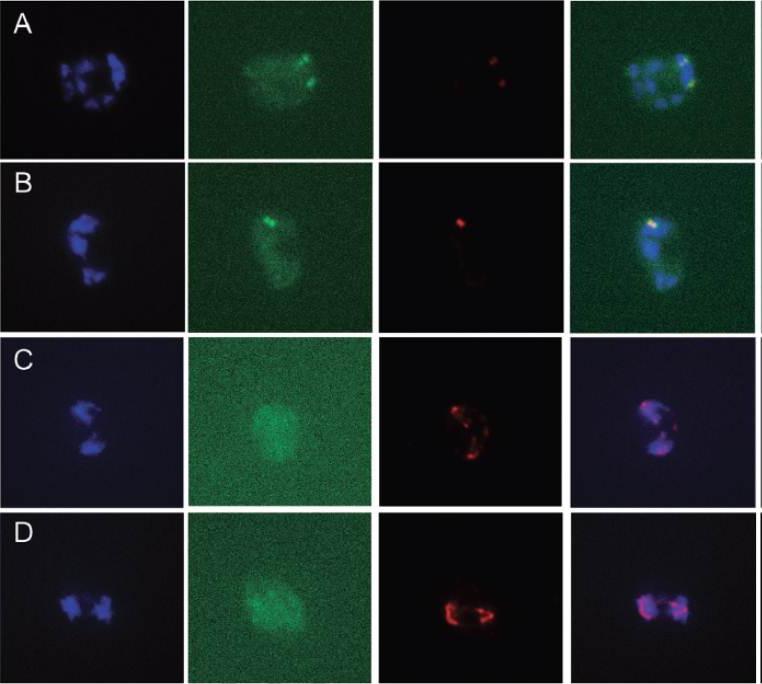
Colocalization experiments of Pfark-1–GFP with a-tubulin. Fluorescence microscopy of fixed schizont stage transgenic parasites expressing Pfark-1–GFP. A and B. An anti-a-tubulin mAb recognizes discrete foci (red) near DAPI-stained nuclear DNA (blue). The red fluorescent pattern is consistent with early-stage mitotic spindle formation developing from the spindle pole bodies anchored in the nuclear membrane. In colour merge images, the Pfark-1–GFP protein appears to localize at opposite sides of the early mitotic spindle, consistent with a localization at the mitotic spindle poles. C and D. In these two panels the anti-a-tubulin mAb recognizes partly disassembled bipolar mitotic spindles (red) aligned with DAPI-stained nuclear bodies (blue), representing daughter nuclear bodies in the process of completion of nuclear division. The Pfark-1–GFP protein does not appear now to be confined to mitotic spindle poles. A diffuse cytosolic green fluorescence is observable, presumably representing the GFP degradation product observed by Western blot analysis.Reininger L, Wilkes JM, Bourgade H, Miranda-Saavedra D, Doerig C. An essential Aurora-related kinase transiently associates with spindle pole bodies during Plasmodium falciparum erythrocytic schizogony. Mol Microbiol. 2011 79(1):205-21.
See original on MMP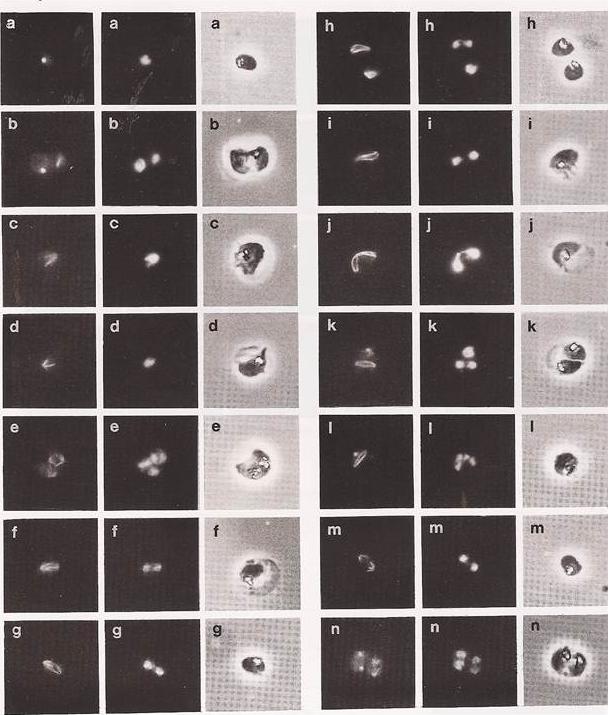
Immunofluorescence of parasites undergoing the first mitotic division. Each set of 3 photographs has the tubulin fluorescence on the left the nuclear stain in the middle and the phase contrast on the right. The panels a to n derive from chronologically ordered samples. The first structure comprising polymerized microtubules visible by immuno-fluorescence is that associated with the centriolar plaque (panel a). Panel b shows a double infection, the left-hand parasite showing the centriolar plaque, while in the parasite on the right, half spindles (hemispindles) are developing from the centriolar plaque (cleared in c and d). Note the close alignment of the nuclear (DAPI-stained) material to the shape of the microtubular structure in panel c. The hemispindles then migrate into opposition, by movement of the centriolar plaques in the nuclear membrane (e, upper parasite of the double infection). When in opposition the hemispindles combine to produce the complete spindle. Nuclear material is now in the process of division and is located towards the spindle poles (f, g and h). The next stage is spindle elongation. Sister 'chromatids' are further separated by the physical elongation of the spindle (i). Panel j shows that the opposition of the hemispindles does not have to be at 1800 for successful karyokinesis. The spindle is then partly disassembled into its component hemispindles (m). Panel n shows a telophase analogue (the left-hand parasite of the double infection). A centriolar plaque is now associated with each daughter nuclear body. Diffuse tubulin staining returns, suggesting the export of tubulin molecules from the nucleus to the cytoplasm on the disassembly of the hemispindles. Read M, Sherwin T, Holloway SP, Gull K, Hyde JE. Microtubular organization visualized by immunofluorescence microscopy during erythrocytic schizogony in Plasmodium falciparum and investigation of post-translational modifications of parasite tubulin. Parasitology. 1993 106:223-32. Copyright Cambridge University Press Journals 2011
See original on MMP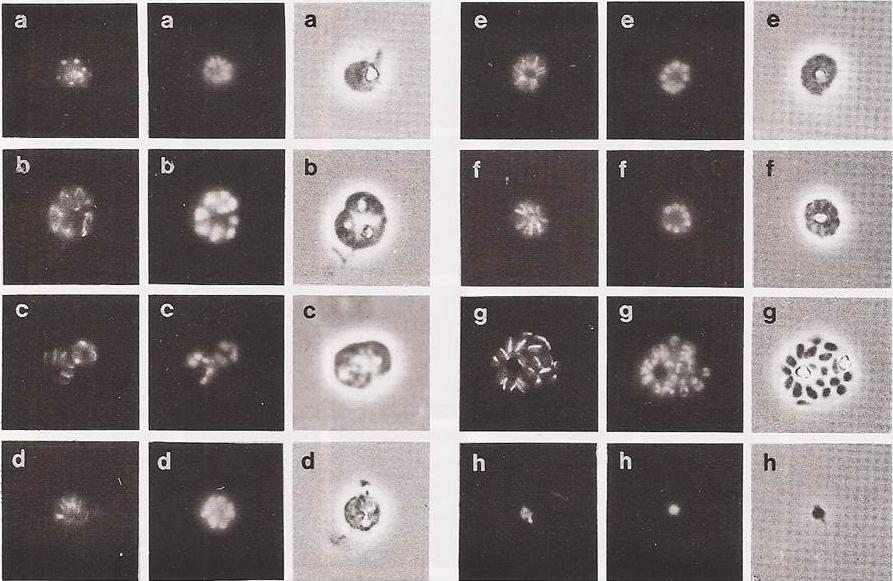
Immunofluorescence of late mitotic and post mitotic schizonts/ The panels a to h derive from chronologically ordered samples. Division in the schizont does not appear to be completely synchronous (see below). It appears that spindles can be in various stages of assembly within the same parasite. This can be seen in panel b (the uppermost parasite of the triple infection). Panels d, e and f show late schizonts displaying a very regular morphology. The microtubules are now arranged in a regular radial array, like the spokes of a cartwheel. Each spoke appears to be associated with a particular nucleus. The microtubular spokes seem to be extra-nuclear and to extend beyond the nuclei towards the centre of the schizont. These parasites are interpreted as being schizonts that have completed their mitotic divisions and are now in the cytodifferentiation phase leading to the production of merozoites.Read M, Sherwin T, Holloway SP, Gull K, Hyde JE. Microtubular organization visualized by immunofluorescence microscopy during erythrocytic schizogony in Plasmodium falciparum and investigation of post-translational modifications of parasite tubulin. Parasitology. 1993 106:223-32.
See original on MMP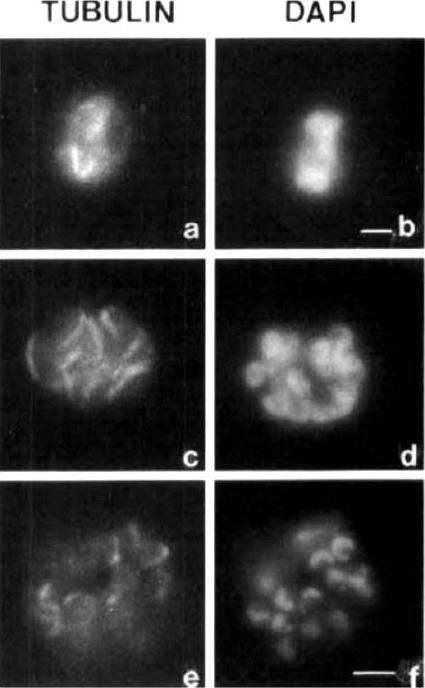
Mitotic spindle organization during the schizogony of P. fulciparum. The observations were performed at the 30-35 h (a, b), 35-40 h (c. d) and 43-48 h (e, f) periods of the erythrocytic cycle. Each set of two photographs has the tubulin immunofluorescence as revealed by the mAb TATl on the left and the nuclear staining with DAPI on the right. Bar = 0.1 mm.Sinou V, Boulard Y, Grellier P, Schrevel J. Host cell and malarial targets for docetaxel (Taxotere) during the erythrocytic development of Plasmodium falciparum. J Eukaryot Microbiol. 1998 45:171-83. Copyright John Wiley & Sons Ltd. 2010.
See original on MMP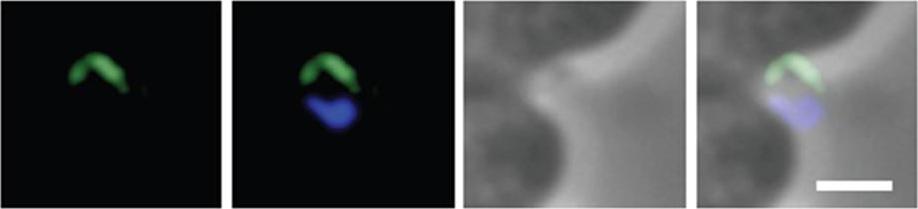
Schizont and merozoite culture was incubated overnight with 0.1µM cytochalasin D to inhibit invasion. Thin smeared on a glass slide, fixed in methanol, stained with commercial antibody against a-tubulin (mouse anti-tubulin DM1A, Sigma, 1:200). Two stripes are show from apex which are presumed to be the 2-3 microtubules present in the merozoite (see Fowler et al. Microtubule associated motor proteins of Plasmodium falciparum merozoites. Molecular and Biochemical Parasitology (2001) vol. 117 (2) pp. Baum J, Tonkin CJ, Paul AS, Rug M, Smith BJ, Gould SB, Richard D, Pollard TD, Cowman AF. A malaria parasite formin regulates actin polymerization and localizes to the parasite-erythrocyte moving junction during invasion. Cell Host Microbe. 2008 3:188-98. Copyright Elsevier 2009.
See original on MMP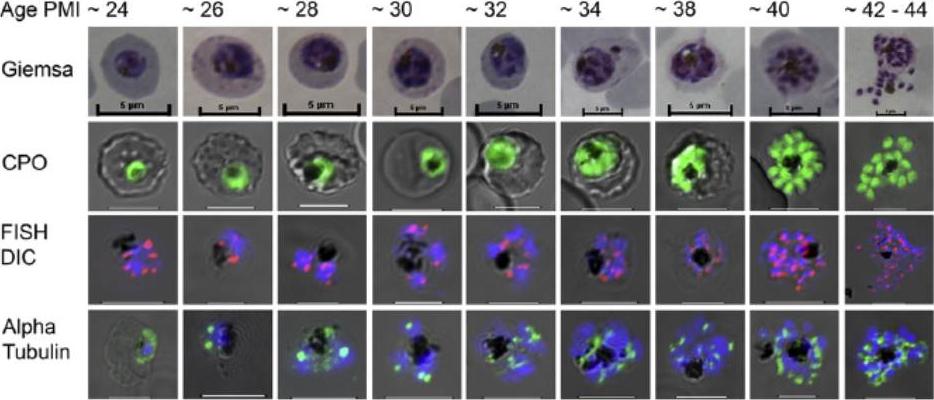
Comparative imaging of mitotic development in trophozoite and schizont stages of Plasmodium falciparum (24–44 h post-merozoite invasion (PMI)). Giemsa, Coriphosphine O (CPO; staining nuclei), Rep 20 fluorescence in situ hybridization (FISH) and microtubule staining of intra-erythrocytic (IE) parasites, compared from the first S phase, through subsequent mitotic divisions. Giemsa: Methanol-fixed, Giemsa-stained IE parasites. CPO: Differential interference contrast (DIC) images of CPO fluorescence from unfixed, live-stained parasites. FISH DIC: Rep 20 FISH marking of telomeres in DAPI-stained nuclei, superimposed on the DIC image of the IE. Alpha Tubulin: Microtubular structures detected in formaldehyde-fixed cells using a monoclonal antibody binding to a-tubulin. Arnot DE, Ronander E, Bengtsson DC. The progression of the intra-erythrocytic cell cycle of Plasmodium falciparum and the role of the centriolar plaques in asynchronous mitotic division during schizogony. Int J Parasitol. 2010 41:71-80. Copyright Elsevier 2011.
See original on MMP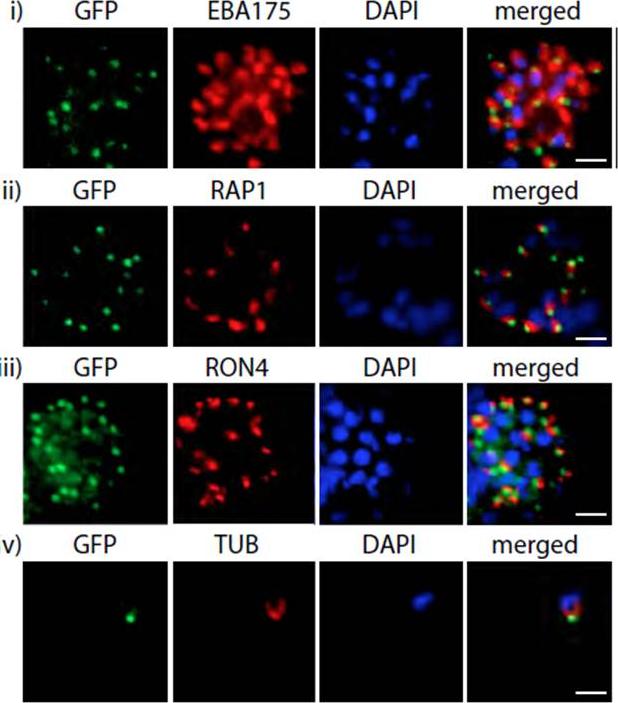
PfMyoB-GFP is located at the apical pole of the merozoite. Indirect immunofluorescence of PfMyoB-GFP (green) with various merozoite organelle markers (red), using antisera to i) EBA175 (microneme marker), ii) RAP1 (rhoptry bulb), iii) RON4 (rhoptry neck) and iv) α-tubulin. Samples were counterstained with DAPI (blue). The merged images are also shown. Rows i-iii show individual schizonts, row iv shows an individual merozoite. Scale bar: 2 μM. Antibodies to GFP produced a compact discrete dot pattern of fluorescence located at the very apical end of the MyoB-GFP parasites, near to the localisation of the apical markers EBA175, RON4, and RAP1. However, whilst in some cases the fluorescent signal partially overlapped with these markers, MyoB appeared to be in a distinct location within the cell, anterior to the microneme marker, the rhoptry bulb, and even to the rhoptry neck.Yusuf NA, Green JL, Wall RJ, Knuepfer E, Moon RW, Schulte-Huxel C, Stanway RR, Martin SR, Howell SA, Douse CH, Cota E, Tate EW, Tewari R, Holder AA. The Plasmodium Class XIV Myosin, MyoB has a Distinct Subcellular Location in Invasive and Motile Stages of the Malaria Parasite, and an Unusual Light Chain. J Biol Chem. 2015 Mar 23.
See original on MMP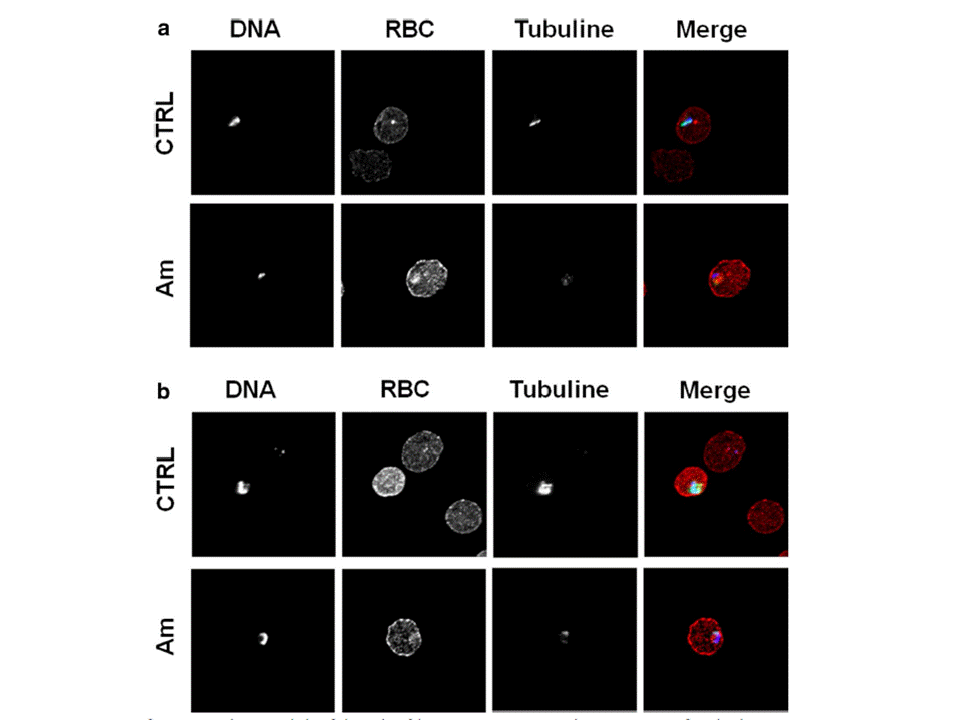
Effect of amitozyn on the microtubules of Plasmodium falciparum. Human RBC at 2 % haematocrit were infected with 3G8 strain of P. falciparum. At 0.5 % parasitaemia infected RBC (iRBC) were treated with 30 μg/mL amitozyn for 24 h. Untreated iRBC were used as a control. Fixed iRBC were stained with anti-α-tubulin (green), anti-RBC (red) and DAPI (blue) and analysed by confocal microscopy. a Rings and b trophozoite stages of P. falciparum, respectively. Amitozyn destroyed the filamentous structure of parasite microtubules at the ring and trophozoite stages.Tcherniuk SO, Chesnokova O, Oleinikov IV, Potopalsky AI, Oleinikov AV. Anti-malarial effect of semi-synthetic drug amitozyn. Malar J. 2015 14(1):425.
See original on MMP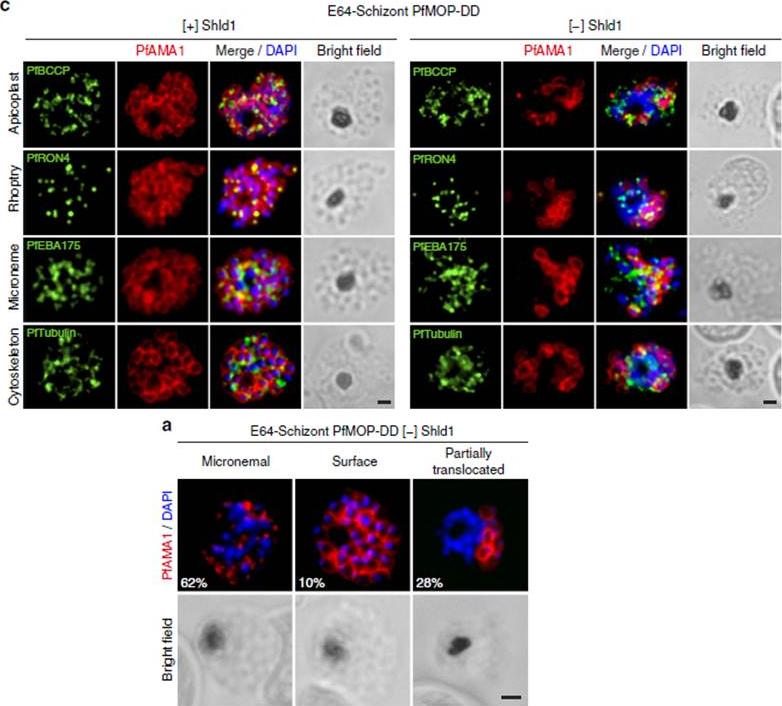
PfAMA1 translocation is aberrant in schizonts with PfMOP-knockdown. (a) Schizonts from [+]/[-] Shld1 PfMOP-DD parasites were E64-treated, fixed, probed with anti-PfAMA1 and scored as micronemal (M), partially translocated (PT) or surface (S), representative [+] Shld1 parasite IFAs shown. (c) Synchronized schizont stage (40–44 h) parasites, maintained with 250nM (left panel) or 0 nM (right panel) Shld1, were incubated 6 h in presence of 10 mM E64, methanol-fixed, permeabilized, and stained using antibodies against PfRON4, PfRhopH3, PfEBA175 and PfTubulin. Staining for these markers was similar in [+] and [-] Shld1 conditions. PfAMA1 staining was used to identify E64-treated schizonts that were sufficiently mature (that is, surface staining or partially translocated staining, but not micronemal staining, scale bar, 1 mm).Absalon S, Robbins JA, Dvorin JD. An essential malaria protein defines the architecture of blood-stage and transmission-stage parasites. Nat Commun. 2016 7:11449.
See original on MMP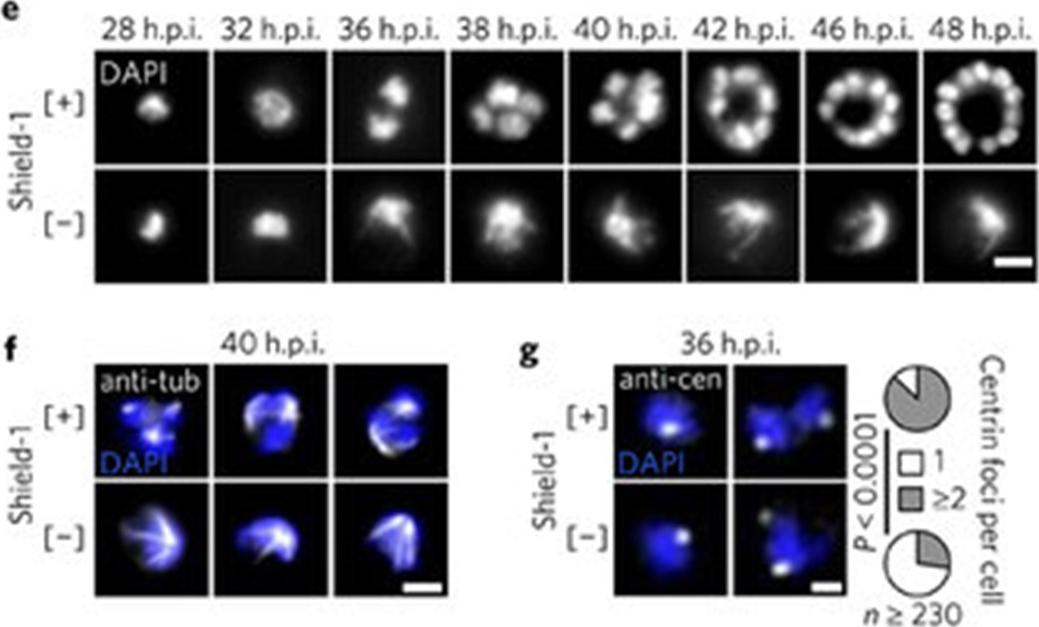
e, Nuclear development in PfCRK4-depleted parasites (D10 parent, representative of three biological replicates, and results with P2G12-PfCRK4-HA-DD). Scale bar, 2 μm. f, Immunofluorescence detection of spindle structures in PfCRK4-depleted parasites (P2G12 parent, representative of two biological replicates). Tub, tubulin. Scale bar, 2 μm. g, Quantification of centriolar plaques by immunofluorescence in PfCRK4-depleted parasites (D10 parent; P value, chi-square test; representative of two biological replicates and results with P2G12-PfCRK4-HA-DD). Cen, centrin. Scale bar, 1 μm. Analysis of nuclei stained with fluorescent DNA-specific dyes confirmed that the nuclei did not divide and revealed a substantiallydistorted nuclear morphology (e). Hemispindle structures were evident in PfCRK4-depleted cells (f); however, they were greatlyenlarged relative to spindles in wild-type parasites and might account for the nuclear distortion.Ganter M, Goldberg JM, Dvorin JD, Paulo JA, King JG, Tripathi AK, Paul AS, Yang J, Coppens I, Jiang RH, Elsworth B, Baker DA, Dinglasan RR, Gygi SP, Duraisingh MT. Plasmodium falciparum CRK4 directs continuous rounds of DNA replication during schizogony. Nat Microbiol. 2017 2:17017
See original on MMP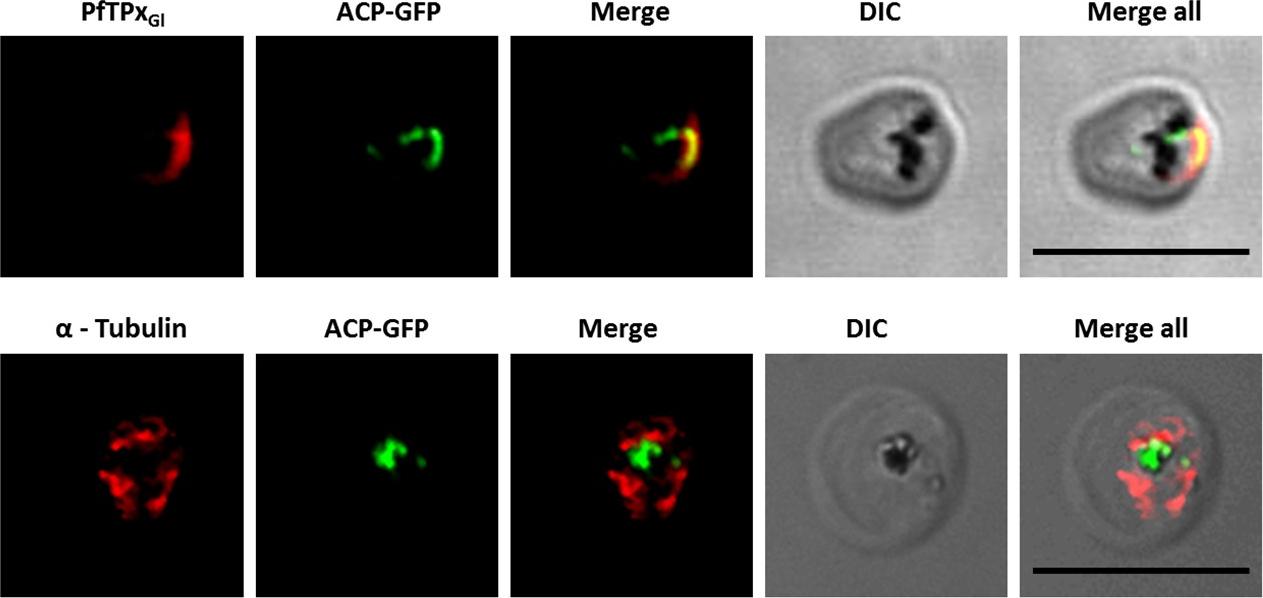
Immunofluorescence images showing PfTPxGl and microtubules in D10-ACPleader-GFP parasites with drug washed out. Reversion of PfTPxGl localization to the organelles and intact microtubular structures observed in parasites in drug washed out medium after vinblastine treatment. In these experiments, localization of PfTPxGl was reverted to the apicoplast in 47% parasites, while remaining 53% parasites showed mitochondrial localization (23 parasites counted). Scale Bar: 10 mm.Chaudhari R, Dey V, Narayan A, Sharma S, Patankar S. Membrane and luminal proteins reach the apicoplast by different trafficking pathways in the malaria parasite Plasmodium falciparum. PeerJ. 2017 Apr 27;5:e3128.
See original on MMP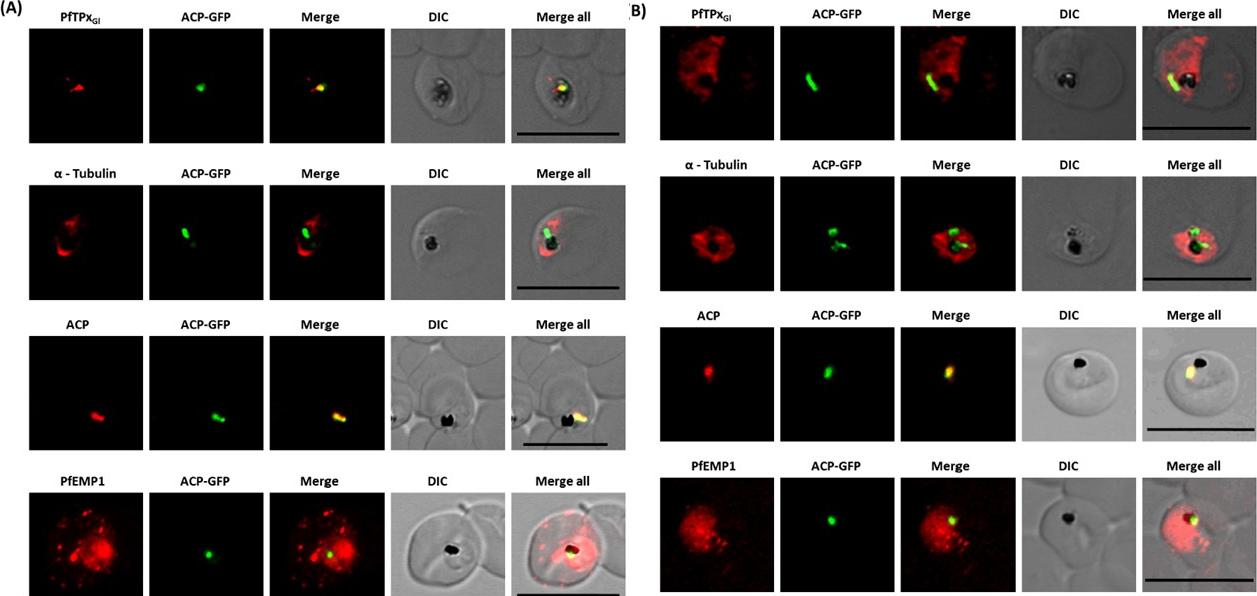
Immunofluorescence images show PfTPxGl, microtubules, ACP-GFP, PfACP and PfEMP1 trafficking in vinblastine-treated D10-ACPleader-GFP parasites. (A) PfTPxGl, microtubules, ACP-GFP, PfACP and PfEMP1 localization in solvent control parasites, (B) PfTPxGl, microtubules, ACP-GFP, PfACP and PfEMP1 localization in vinblastine treated parasites. In these experiments, targeting to the apicoplast was inhibited in 94% of the parasites with vinblastine treatment (33 parasites counted) while an arrest of PfEMP1 was observed in 97% of the parasites (134 parasites counted). Scale Bar: 10 mm.Chaudhari R, Dey V, Narayan A, Sharma S, Patankar S. Membrane and luminal proteins reach the apicoplast by different trafficking pathways in the malaria parasite Plasmodium falciparum. PeerJ. 2017 Apr 27;5:e3128.
See original on MMP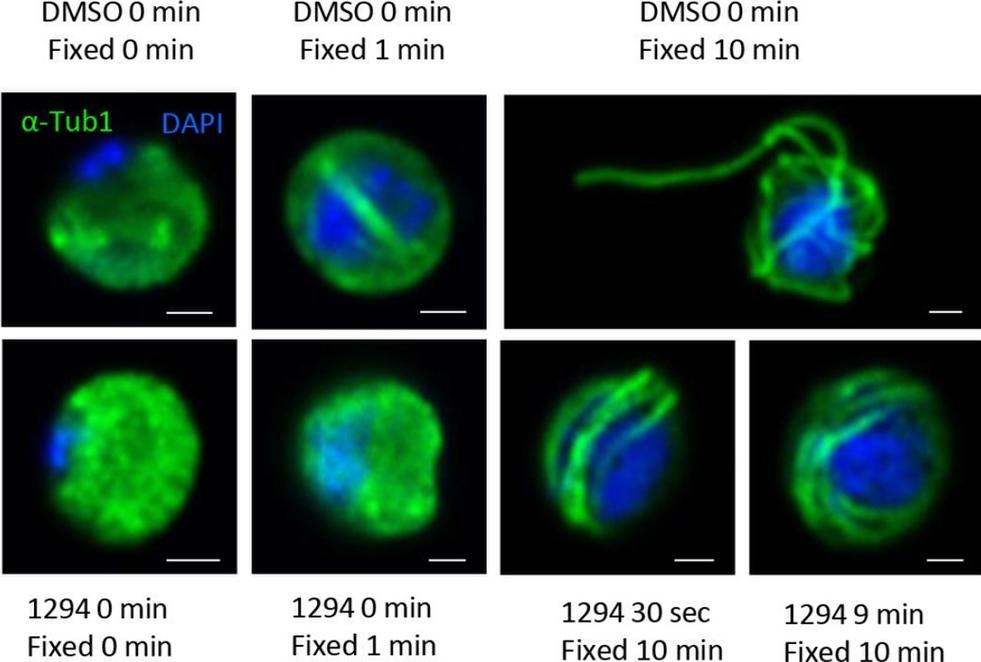
A chemical genetic approach reveals CDPK4 activity is required to initiate the first round of DNA replication, assemble the first mitotic spindle and initiate axoneme motility. Immunofluorescence assays showing the effect of 1294 at different time points after activation. Addition of 1 mM 1294 at the time of activation prevents the formation of mitotic spindles as observed 1 min after activation in presence of DMSO. Addition of 1294 at 30 s or at 9 min post-activation does not inhibit axoneme formation but blocks initiation of axoneme motility and condensation of chromatin. Absence of 1294 treatment leads to the exflagellation of male gametes 10 min after activation. Scale bars = 1 mm.Fang H, Klages N, Baechler B, Hillner E, Yu L, Pardo M, Choudhary J, Brochet M. Multiple short windows of calcium-dependent protein kinase 4 activity coordinate distinct cell cycle events during Plasmodium gametogenesis. Elife. 2017 May 8;6. pii: e26524.
See original on MMP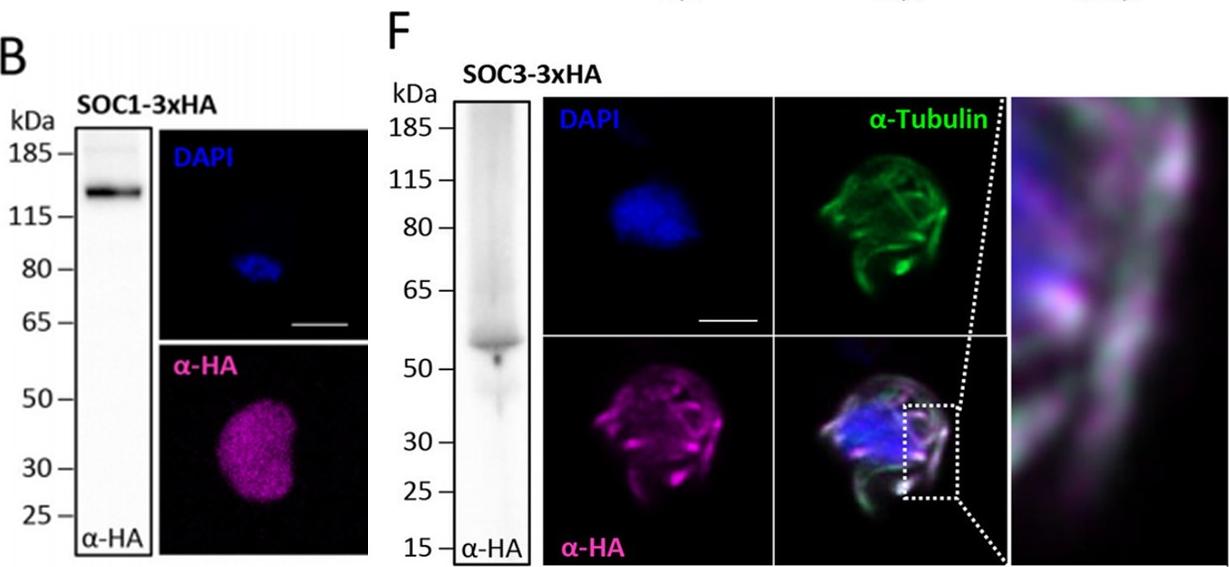
Functional characterisation of three identified substrates of CDPK4. (B) SOC1-3xHA shows a diffuse cytoplasmic distribution (scale bar = 2 mm) but interacts withCDPK4 and proteins of the MCM complex. (F) SOC3 colocalises with axonemal a-tubulin in exflagellating gametes. Scale bar = 2 mm.Fang H, Klages N, Baechler B, Hillner E, Yu L, Pardo M, Choudhary J, Brochet M. Multiple short windows of calcium-dependent protein kinase 4 activity coordinate distinct cell cycle events during Plasmodium gametogenesis. Elife. 2017 May 8;6. pii: e26524. PMID: 28481199
See original on MMPMore information
| PlasmoDB | PY17X_0420500 |
| GeneDB | PY17X_0420500 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | null |
| Orthologs | PBANKA_0417700 , PCHAS_0418600 , PF3D7_0903700 , PKNH_0701400 , PVP01_0702100 , PVX_098630 |
| Google Scholar | Search for all mentions of this gene |