PF3D7_1212000 glutathione peroxidase-like thioredoxin peroxidase (TPx(Gl))
Disruptability [+]
| Species | Disruptability | Reference | Submitter |
|---|---|---|---|
| P. falciparum 3D7 |
Refractory |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen |
Mutant phenotypes [+]
None reported yet. Please press the '+' button above to add one.Imaging data (from Malaria Metabolic Pathways)
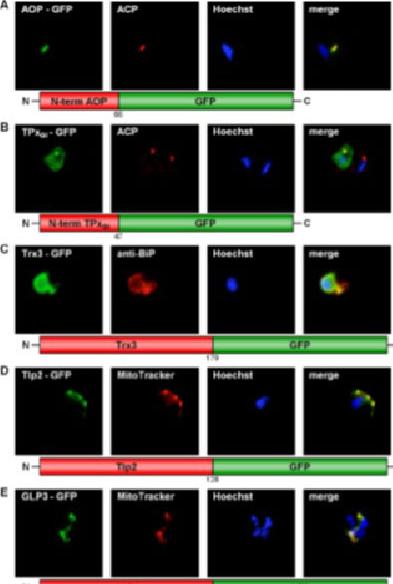
GFP targeting by various P. falciparum redox proteins. (A) Apicoplast targeting of the AOP N-terminus. (B) Dual localization (cytosol and apicoplast) of the TPxGl N-terminal amino acid sequence. (C) ER-targeting of Trx3. (D) Mitochondrial targeting of Tlp2. (E) Mitochondrial targeting of GLP3. (F) Apicoplast targeting of the GILP N-terminal sequence. Colocalization of GFP with the mitochondrial dye MitoTrackerOrange in fixed cells. Colocalization of GFP and the apicoplast marker ACP PFB0385w or the ER marker BiP PF11_0098 in fixed, immunodecorated cells. Kehr S, Sturm N, Rahlfs S, Przyborski JM, Becker K. Compartmentation of redox metabolism in malaria parasites. PLoS Pathog. 2010 6:e1001242.
See original on MMP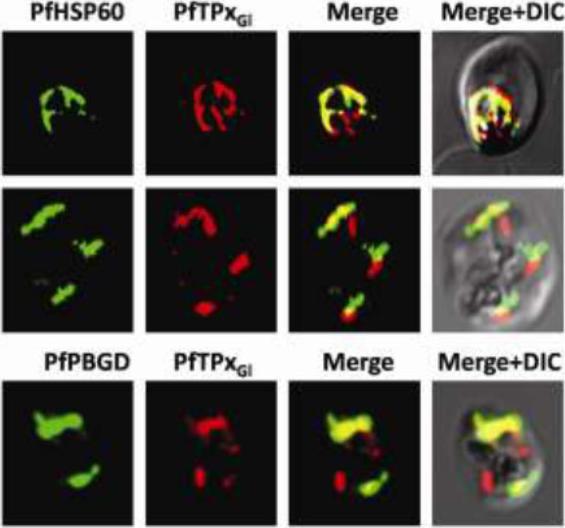
Immunofluorescence images showing multiple localizations of PfTPxGL. PfTPxGL-red signal (anti-mouse Alexa fluor 568), PfPBGD – apicoplast marker, green signal (anti-rabbit FITC), HSP60 – mitochondrial marker, green signal (anti-rabbit FITC). The imaging was carried out with scale bars varying from 5-20 mm The infected RBCs imaged are 6-8 mm across. Parasites showing dual localization of PfTPxGL to the apicoplast and mitochondrionChaudhari R, Narayan A, Patankar S. A novel trafficking pathway in Plasmodium falciparum for the organellar localization of glutathione peroxidase-like thioredoxin peroxidase. FEBS J. 2012 279(20):3872-88
See original on MMP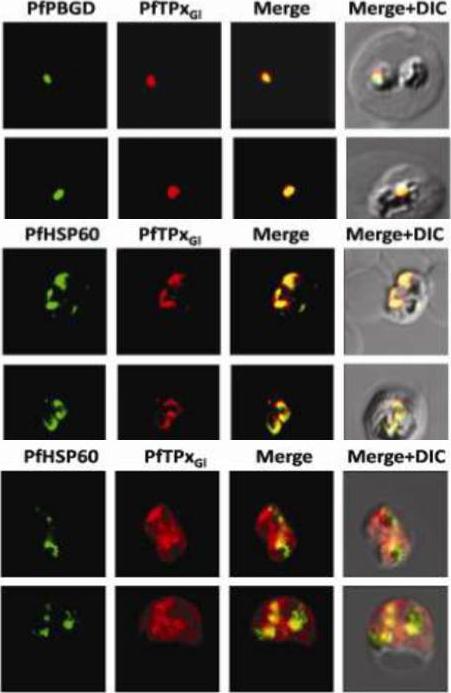
Immunofluorescence images showing multiple localizations of PfTPxGL. PfTPxGL-red signal (anti-mouse Alexa fluor 568), PfPBGD – apicoplast marker, green signal (anti-rabbit FITC), HSP60 – mitochondrial marker, green signal (anti-rabbit FITC). The imaging was carried out with scale bars varying from 5-20 mm. The infected RBCs imaged are 6-8 mm across. (Upper panel) Parasites showing only apicoplast localization of PfTPxGL, (Middle panel) Parasite showing only mitochondrial localization of PfTPxGL, (Lower panel) Parasite showing organellar as well as cytosolic localization of PfTPxGL. Chaudhari R, Narayan A, Patankar S. A novel trafficking pathway in Plasmodium falciparum for the organellar localization of glutathione peroxidase-like thioredoxin peroxidase. FEBS J. 2012 279(20):3872-88.
See original on MMP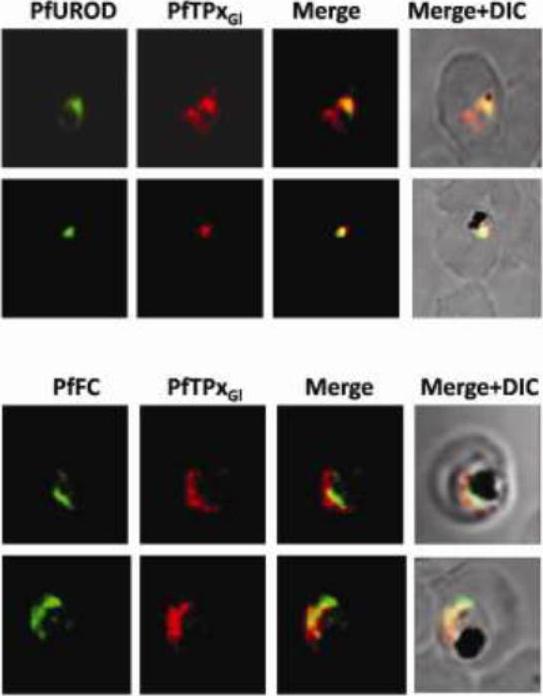
Immunofluorescence images showing multiple localizations of PfTPxGL. PfTPxGL-red signal (anti-mouse Alexa fluor 568). The imaging was carried out with scale bars varying from 5-20 mm. The infected RBCs imaged are 6-8 mm across. Parasites showing localization of PfTPxGl using antibodies purified against rPfTPxGL. In several parasites, PfTPxGL was localized exclusively to only one organelle per parasite. In some parasites, PfTPxGL co-localized with exclusively the mitochondrial markers PfUROD or PfFC. Chaudhari R, Narayan A, Patankar S. A novel trafficking pathway in Plasmodium falciparum for the organellar localization of glutathione peroxidase-like thioredoxin peroxidase. FEBS J. 2012 279(20):3872-88 PMID:
See original on MMP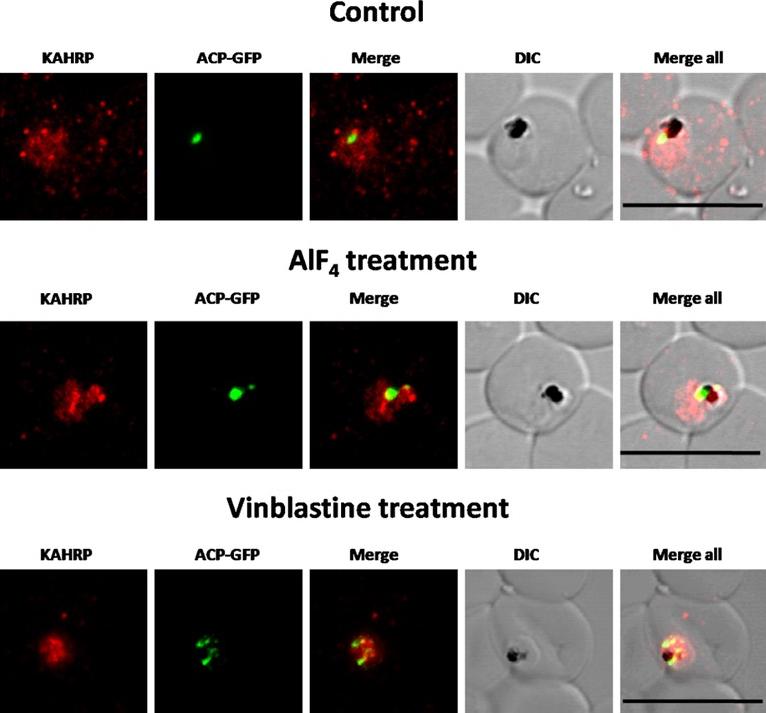
Immunofluorescence images showing PfTPxGl and PfFC (mitochondrial marker protein) trafficking in AlF4−, nocodazole and vineblsatine treated 3D7 parasites(A) PfTPxGl and PfFC co-localization in AlF4−-treated parasites, (B) PfTPxGl and PfFC co-localization in vinblastine-treated parasites, (C) PfTPxGl and PfFC co-localization in nocodazole-treated parasites. In this experiment, PfTPxGl was found to be co-localized with the mitochondrial marker protein PfFC in 40% of the treated parasites suggesting that trafficking of PfTPxGl to the mitochondrion may be partially disrupted by the treatments. Scale Bar: 10 µm. When the parasites from the same treated cultures were analyzed for co-localization of the disrupted PfTPxGl signal with mitochondrial marker protein ferrochelatase (PfFC), we observed that in 35-40% of cells the PfTPxGl signal showed some overlap.Chaudhari R, Dey V, Narayan A, Sharma S, Patankar S. Membrane and luminal proteins reach the apicoplast by different trafficking pathways in the malaria parasite Plasmodium falciparum. PeerJ. 2017 Apr 27;5:e3128. PMID: 28462015
See original on MMP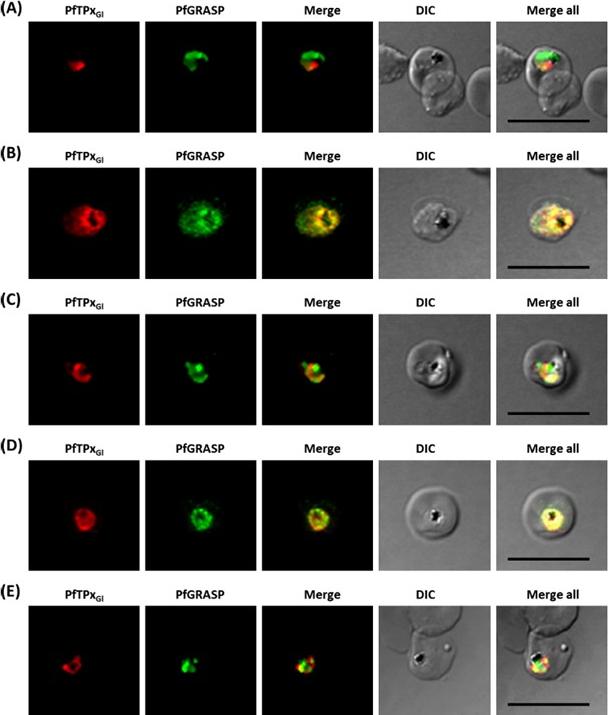
Immunofluorescence images showing the Golgi morphology in AlF4 and vinblastine treated parasites. (A) PfGRASP localization in control parasites for AlF4treatment, (B) Golgi morphology in AlF4- treated parasites (17 parasites counted, Golgi structure was dispersed in 95% parasites), (C) Pf-GRASP localization in solvent (PBS) control parasites for vinblastine treatment, (D) Golgi morphology in vinblastine-treated parasites (18 parasites counted, Golgi structure was dispersed in 95% parasites),(E) Golgi morphology in parasites reverted after vinblastine treatment (11 parasites counted, Intact Golgi structure was observed in 90% parasites). Scale Bar: 10 mm.Chaudhari R, Dey V, Narayan A, Sharma S, Patankar S. Membrane and luminal proteins reach the apicoplast by different trafficking pathways in the malaria parasite Plasmodium falciparum. PeerJ. 2017 Apr 27;5:e3128.
See original on MMP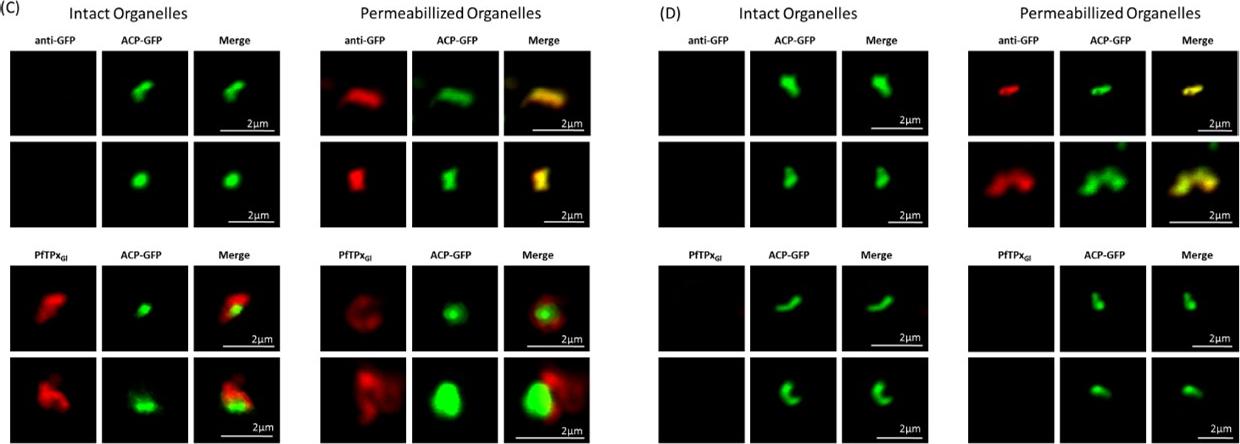
PfTPxGl localization to the outermost membrane of the apicoplast in D10-ACPleader-GFP parasites. (C) Localization of PfTPxGl to the membranes of intact/permeabilized apicoplasts from control parasites, (D) Absence of PfTPxGl in the membranes of intact/permeabilized apicoplasts from AlF4- treated parasites. Scale Bars as indicated in the figures. anti-GFP antibodies stained the apicoplast lumen (red signal) only in the permeabilized organelles, but not in the intact ones (C). This immunofluorescence signal colocalized with the intrinsic GFP fluorescence (green signal) of ACP-GFP. In both fractions, anti-PfTPxGl antibodies clearly showed staining surrounding the fluorescent signals from luminal ACP-GFP. While the size and morphology of free intact apicoplasts (based on ACP-GFP staining) remained unaltered, no peri-organellar PfTPxGl signal was observed in the immunofluorescence analysis of intact as well as permeabilized organelles, indicating the involvement of vesicles in the membrane targeting of this protein (D).Chaudhari R, Dey V, Narayan A, Sharma S, Patankar S. Membrane and luminal proteins reach the apicoplast by different trafficking pathways in the malaria parasite Plasmodium falciparum. PeerJ. 2017 Apr 27;5:e3128.
See original on MMP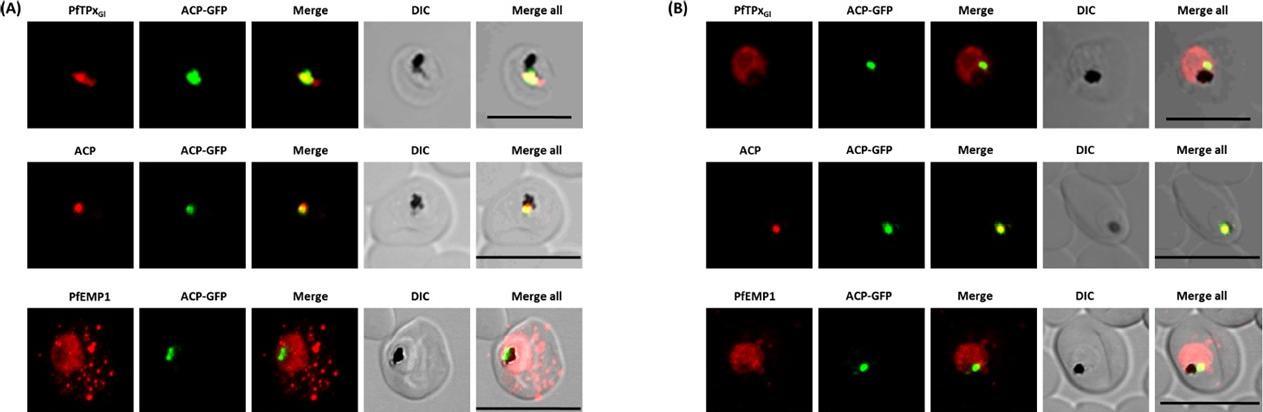
Immunofluorescence images show PfTPxGl, microtubules, ACP-GFP, PfACP and PfEMP1 trafficking in AlF4-treated D10-ACPleader-GFP parasites. (A) PfTPxGl, ACP-GFP, PfACP and PfEMP1 localization in control parasites, (B) PfTPxGl, ACP-GFP, PfACP and PfEMP1 localization in AlF4-treated parasites. For D10-ACPleader-GFP parasites, 98% of the 115 parasites analyzed showed disrupted PfTPxGl signal while 96% of the 147 parasites analyzed showed arrest of PfEMP1 in the parasites. Scale Bar: 10 mm. In control as well as in treated parasites, endogenous ACP colocalized perfectly with ACP-GFP signal suggesting that it was unaltered by the treatment. PfEMP1 was found both in the parasite and in punctate structures in the host RBC.Chaudhari R, Dey V, Narayan A, Sharma S, Patankar S. Membrane and luminal proteins reach the apicoplast by different trafficking pathways in the malaria parasite Plasmodium falciparum. PeerJ. 2017 Apr 27;5:e3128.
See original on MMP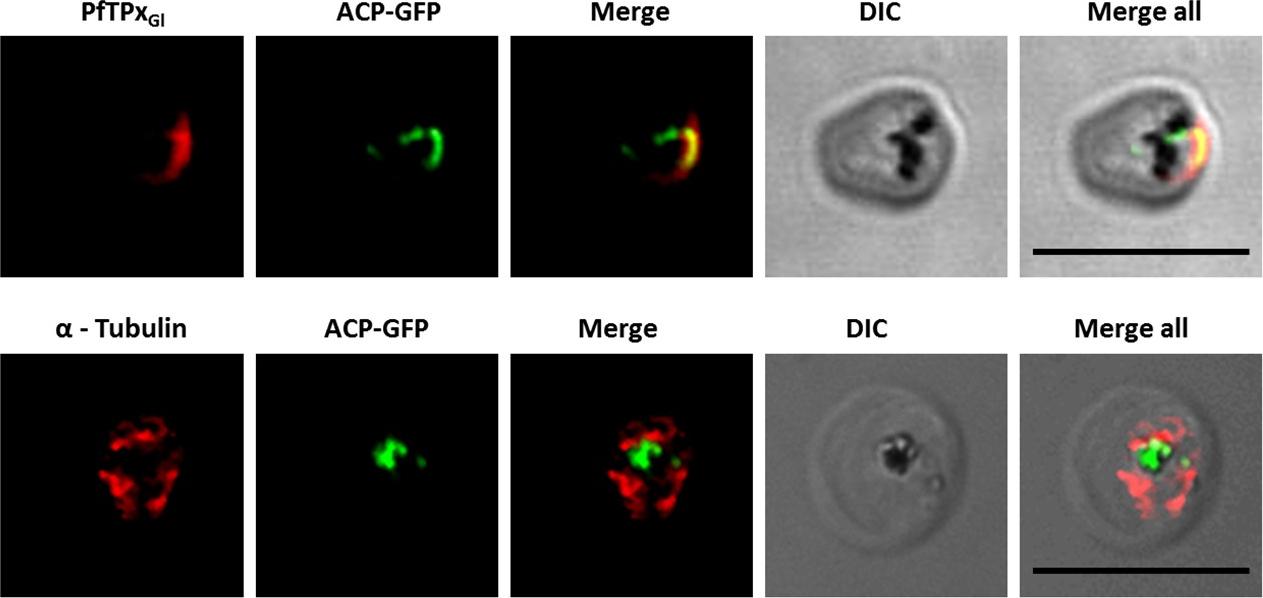
Immunofluorescence images showing PfTPxGl and microtubules in D10-ACPleader-GFP parasites with drug washed out. Reversion of PfTPxGl localization to the organelles and intact microtubular structures observed in parasites in drug washed out medium after vinblastine treatment. In these experiments, localization of PfTPxGl was reverted to the apicoplast in 47% parasites, while remaining 53% parasites showed mitochondrial localization (23 parasites counted). Scale Bar: 10 mm.Chaudhari R, Dey V, Narayan A, Sharma S, Patankar S. Membrane and luminal proteins reach the apicoplast by different trafficking pathways in the malaria parasite Plasmodium falciparum. PeerJ. 2017 Apr 27;5:e3128.
See original on MMP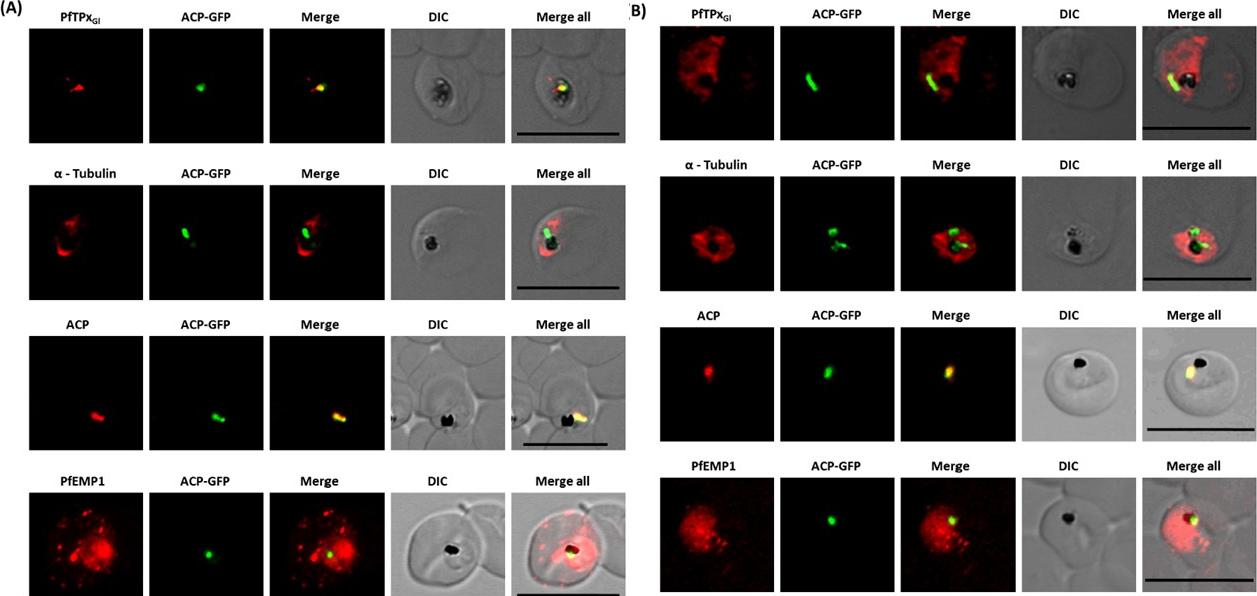
Immunofluorescence images show PfTPxGl, microtubules, ACP-GFP, PfACP and PfEMP1 trafficking in vinblastine-treated D10-ACPleader-GFP parasites. (A) PfTPxGl, microtubules, ACP-GFP, PfACP and PfEMP1 localization in solvent control parasites, (B) PfTPxGl, microtubules, ACP-GFP, PfACP and PfEMP1 localization in vinblastine treated parasites. In these experiments, targeting to the apicoplast was inhibited in 94% of the parasites with vinblastine treatment (33 parasites counted) while an arrest of PfEMP1 was observed in 97% of the parasites (134 parasites counted). Scale Bar: 10 mm.Chaudhari R, Dey V, Narayan A, Sharma S, Patankar S. Membrane and luminal proteins reach the apicoplast by different trafficking pathways in the malaria parasite Plasmodium falciparum. PeerJ. 2017 Apr 27;5:e3128.
See original on MMP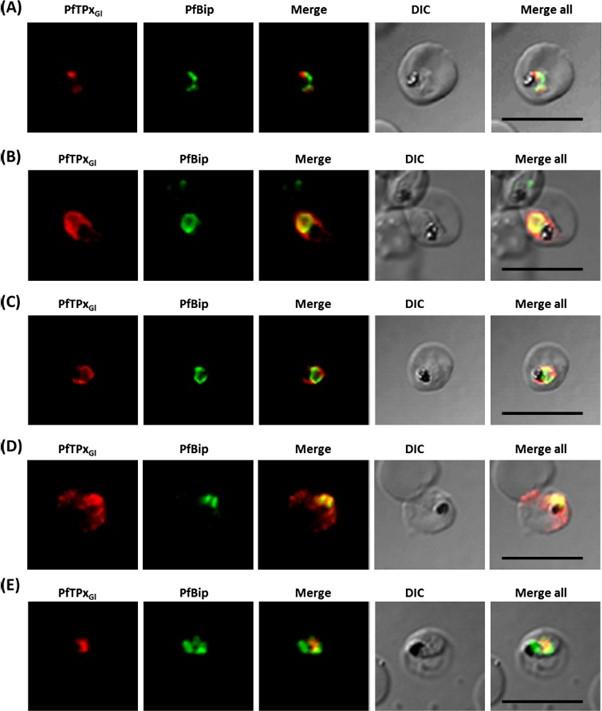
Immunofluorescence images showing the endoplasmic reticulum (ER) morphology in AlF4 and vinblastine treated parasites. (A) PfBiP localization in control parasites for AlF4- treatment, (B) ER morphology in AlF4- treated parasites (14 parasites were counted, none showed dispersal of ER struc-ture), (C) PfBiP localization in solvent (PBS) control parasites for vinblastine treatment, (D) ER morphology in vinblastine-treated parasites (26 parasites were counted, none showed dispersal), (E) ER morphology in parasites reverted after vinblastine treatment (27 parasites counted). Scale Bar: 10 mm. Unlike mammalian cells, no disintegration of the ER was observed in parasites subjected to vinblastine and AlF4-.Both control parasites and drug-treated parasites showed perinuclear ER morphology consistent with the normal development of the ER.Chaudhari R, Dey V, Narayan A, Sharma S, Patankar S. Membrane and luminal proteins reach the apicoplast by different trafficking pathways in the malaria parasite Plasmodium falciparum. PeerJ. 2017 Apr 27;5:e3128.
See original on MMPMore information
| PlasmoDB | PF3D7_1212000 |
| GeneDB | PF3D7_1212000 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | 2277.t00120, MAL12P1.119, PFL0595c |
| Orthologs | PBANKA_0610500 , PCHAS_0612200 , PKNH_1312100 , PVP01_1311200 , PVX_084630 , PY17X_0613000 |
| Google Scholar | Search for all mentions of this gene |