PF3D7_1149000 antigen 332, DBL-like protein (Pf332)
Disruptability [+]
| Species | Disruptability | Reference | Submitter |
|---|---|---|---|
| P. falciparum 3D7 |
Possible |
18614010 | Theo Sanderson, Wellcome Trust Sanger Institute |
| P. falciparum 3D7 |
Possible |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen |
| P. falciparum 3D7 |
Possible |
19007413 | Theo Sanderson, Francis Crick Institute |
Mutant phenotypes [+]
| Species | Stage | Phenotype | Reference | Submitter |
|---|---|---|---|---|
| P. falciparum 3D7 | Asexual |
No difference |
19007413 | Theo Sanderson, Francis Crick Institute |
Imaging data (from Malaria Metabolic Pathways)
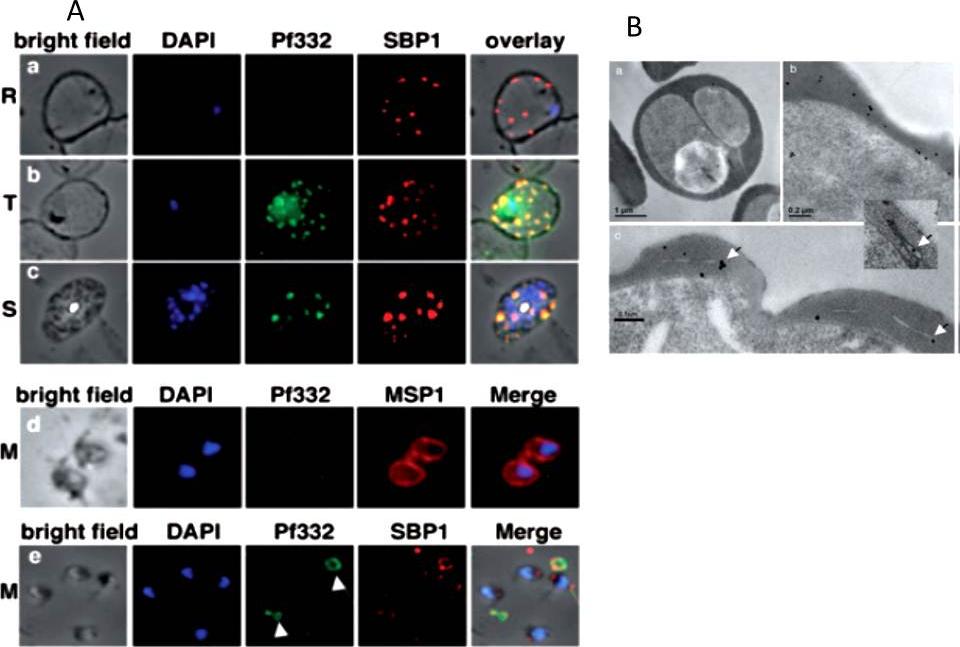
A. Localization of Pf332 in ring stage-infected erythrocytes (R) (a), trophozoite-infected erythrocytes (T) (b) and schizont-infected erythrocytes (S) (c). Each panel contains from left to right: bright field, DAPI-stained nuclei, anti-Pf322, anti-SBP1 PFE0065w and overlay of all four. Free merozoites (M) are shown in d. d is as above except that the fourth panel was labelled with anti-MSP1 PFI1475w antibodies. Panel row e contains free merozoites and material positive for both Pf332 and SBP1 suggesting Maurer’s clefts (white arrows) that were released during schizont rupture.B. High pressure frozen and freeze substituted sections were labelled with either rabbit or mouse anti-Pf332 antibody and show the distribution of Pf332 in the red blood cell cytosol (a and b) and on Maurer’s clefts (c and inset).Hodder AN, Maier AG, Rug M, Brown M, Hommel M, Pantic I, Puig-de-Morales-Marinkovic M, Smith B, Triglia T, Beeson J, Cowman AF. Analysis of structure and function of the giant protein Pf332 in Plasmodium falciparum. Mol Microbiol. 2009 71:48-65. Copyright John Wiley & Sons Ltd. 2010.
See original on MMP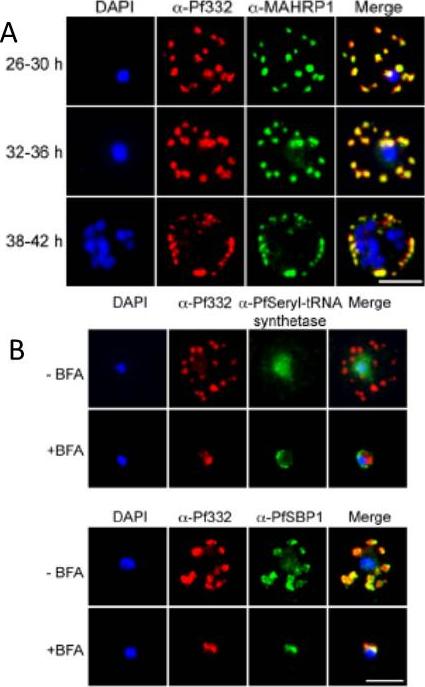
A. IFA of air-dried monolayers of pRBC collected at each of the three indicated time-points. Slides were probed with mouse monoclonal anti-Pf332-DBL (red) and rabbit polyclonal anti-PfMAHRP1 antibodies (green). The parasite was counterstained with DAPI (blue). Scale bar indicates 5 mm. Pf332 displayed a close association with Maurer’s clefts throughout trophozoite maturation and schizogonyB. Air-dried monolayers of BFA+ and BFA2 pRBC were probed with monoclonal anti-Pf332-DBL (red) and polyclonal anti-PfSeryl-tRNA synthetase antibodies (green), or polyclonal anti-Pf332-E200 (red) and polyclonal anti-PfSBP1 antibodies (green). The parasite was counterstained with DAPI (blue). Scale bar indicates 5 mm. Synchronous early ring-stage pRBC were treated with (+) or without BFA for 20 h and inhibition of protein export was verified by IFA. As controls, we included antibodies towards the BFA sensitive SBP1, and the nonexported Seryl-tRNA synthetase. In the absence of BFA, both the anti-Pf332 and anti-SBP1 antibodies stained Maurer’s clefts, whereas the Seryl-tRNA synthetase antibodies only stained the parasiteNilsson S, Angeletti D, Wahlgren M, Chen Q, Moll K. Plasmodium falciparum Antigen 332 Is a Resident Peripheral Membrane Protein of Maurer's Clefts. PLoS One. 2012;7(11):e46980.
See original on MMP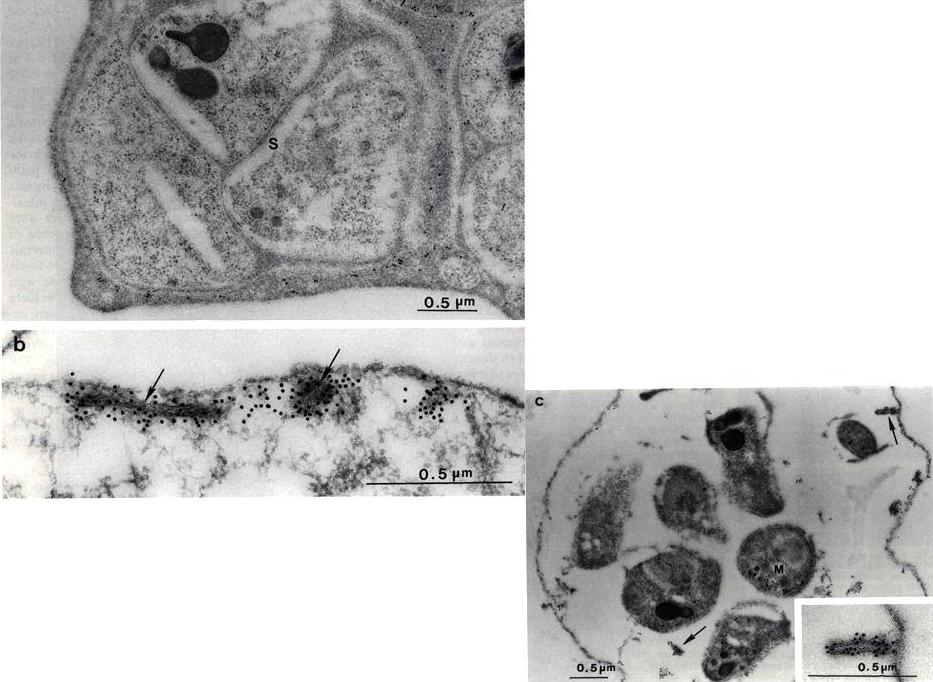
Electron micrograph of P. falciparum schizont-infected red blood cells labeled with Mab51-22. Gold labeling is associated with Maurer’s clefts( arrows) and the parasitophorous vacuolar membrane (PVM). b) Saponin-treated infected cell showing slit-like, membranous Maurer’s cleft and electron dense material in the red blood cell cytoplasm labeled with anti-Pf332 Mab 51-22. c) Saponin-treated segmenter reacted with anti-Pf332 Mab 51-22. Gold label is located on the Maurer’s clefts (arrows) in the cytoplasm and in association with the red blood cell membrane. The inset shows a Maurer’s cleft in close association with the erythrocyte membrane.Hinterberg K, Scherf A, Gysin J, Toyoshima T, Aikawa M, Mazie JC, da Silva LP, Mattei D. Plasmodium falciparum: the Pf332 antigen is secreted from the parasite by a brefeldin A-dependent pathway and is translocated to the erythrocyte membrane via the Maurer's clefts. Exp Parasitol. 1994 79:279-91. Copyright Elsevier 2010.
See original on MMP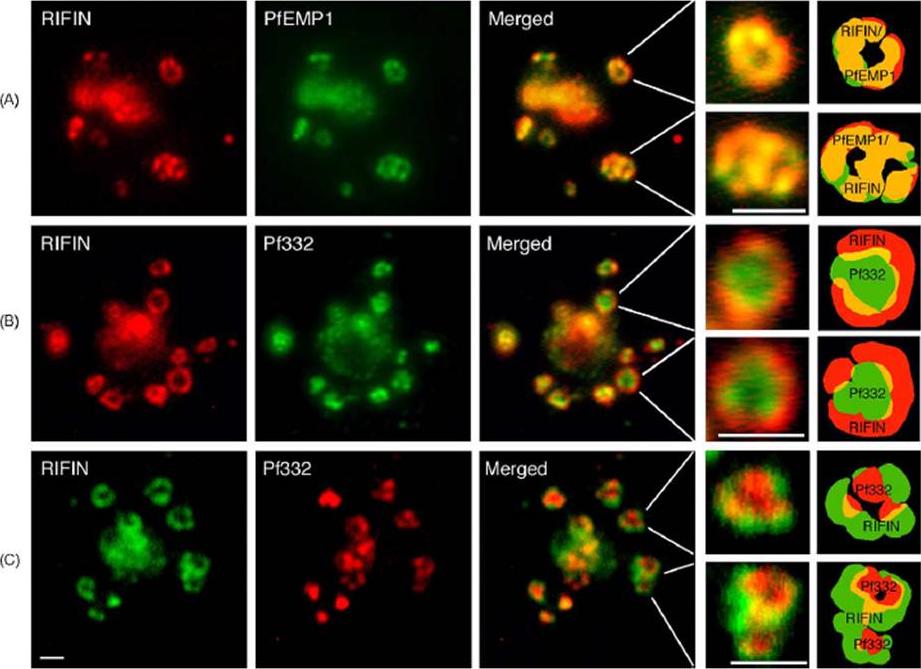
RIFIN and PfEMP1 polypeptides co-localize in the same complex in which the Maurer’s cleft antigen Pf332 is transiently co-transported. (A) Immunofluorescence staining with anti-RIFIN and anti-PfEMP1 antibodies. (B, C) Staining with anti-RIFIN and anti-Pf332 sera. Labeling of proteins was performed on air-dried monolayers of FCR3S1.2(K−) cultures at the trophozoite stage (18–24 h) as described in Section 2. Co-localization of FITC-labeled PfEMP1, Pf332 or RIFIN (green) and TRITC- or CY3-labeled RIFINS and Pf332, respectively (red), is shown in merged images (yellow). Details and illustrations show co-transport of RIFINS, PfEMP1 and Pf332 in individual large multimeric vesicles (LMV). Pf332 often occupies the center or core of the transport complex whereas RIFINS (and PfEMP1) are located in the outer rim of the assembly. Scale bar: 1 mm.Haeggström M, Kironde F, Berzins K, Chen Q, Wahlgren M, Fernandez V. Common trafficking pathway for variant antigens destined for the surface of the Plasmodium falciparum-infected erythrocyte. Mol Biochem Parasitol. 2004 133:1-14. Copyright Elsevier 2010.
See original on MMP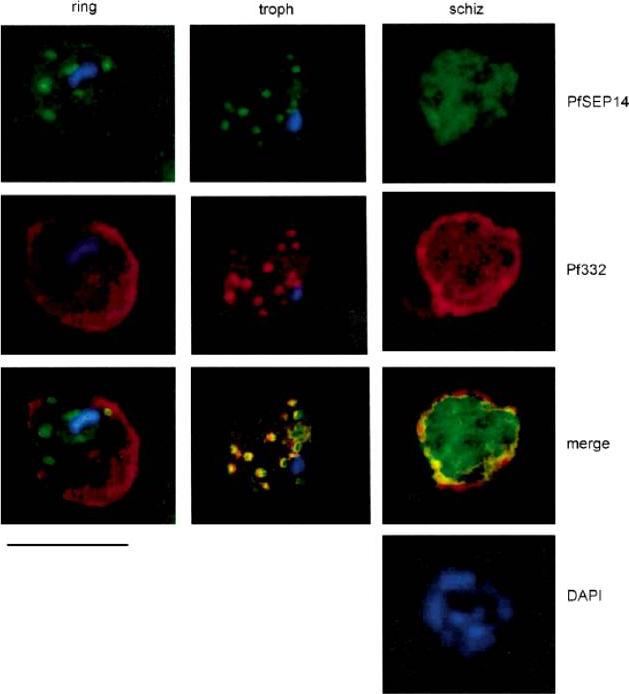
Immuno-localization of PfSEP14 on synchronous P. falciparum cultures. Double immunofluorescence assay was performed on unfixed parasites with PfSEP14-specific rat immune serum (dilution 1:200), revealed with FITC-conjugated anti-rat IgG (green) and with anti-Pf332 monoclonal antibody Mab51-22, revealed with TRITC-conjugated anti-mouse IgG (red). Parasite nuclei are stained with DAPI. Bar represents 5mm. At the stage of ring the distribution of PfSEP14 did not co-localize with that obtained with Mab51-22 which, at this stage, cross reacts with RESA. A similar pattern of fluorescence for both antigens appeared instead in trophozoites, while at the schizont stage patterns were distinguishable, in that SEP14-associated fluorescence was diffuse within the parasitized cell and did not appear concentrated at the edges of the schizonts.Birago C, Albanesi V, Silvestrini F, Picci L, Pizzi E, Alano P, Pace T, Ponzi M. A gene-family encoding small exported proteins is conserved across Plasmodium genus. Mol Biochem Parasitol. 2003 126:209-18. Copyright Elsevier 2010.
See original on MMP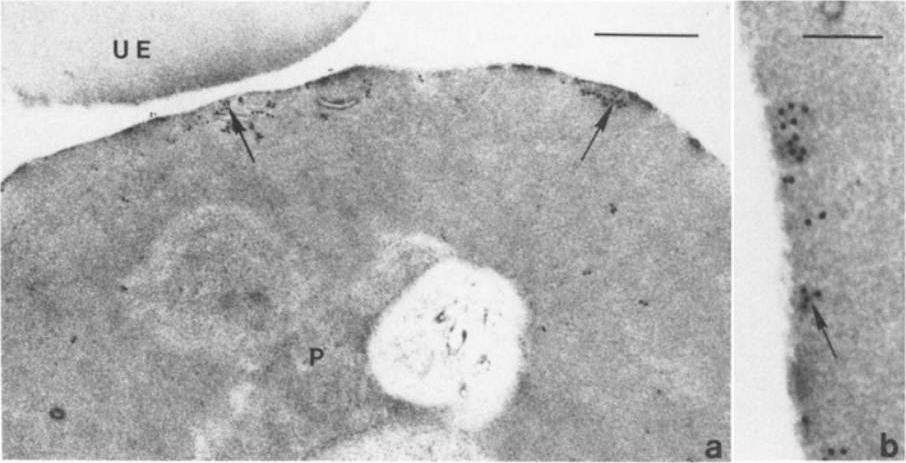
lmmunoelectron micro-graphs showing reactivity of mAb 33G2 (arrow) with P. falciparum (P)-infected erythrocytes. Note the absence of gold particles in an uninfected erythrocyte (UE). Bars: a. 1 mm: b. 0.25 mm. The localization of Ag reacting with mAb 3362 to the membrane of P. falciparum-infected erythrocytes was confirmed. The binding of the mAb was mainly confined to the area just under the erythrocyte membrane. Scattered clusters of labeling were also observed within the intracellular parasite as well as in the erythrocyte cytoplasm. mAb 3362 reacts with RESA and antigens 332 and Pf11.1.Udomsangpetch R, Carlsson J, Wåhlin B, Holmquist G, Ozaki LS, Scherf A, Mattei D, Mercereau-Puijalon O, Uni S, Aikawa M, Berzins K, Perlmann P. Reactivity of the human monoclonal antibody 33G2 with repeated sequences of three distinct Plasmodium falciparum antigens. J Immunol. 1989 142:3620-6. Copyright Journal of Immunology 2009.
See original on MMP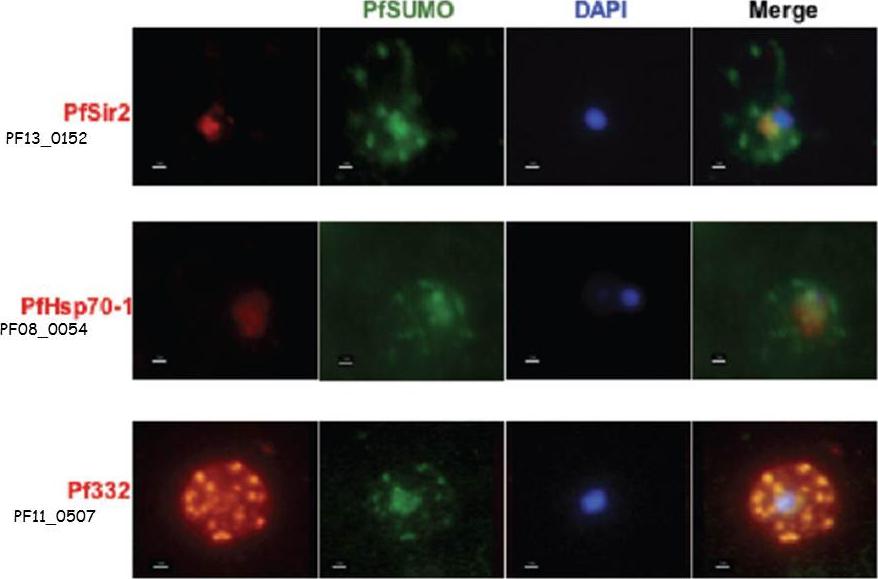
Co-localization assays of PfSUMO antiserum with nuclear marker PfSir2, cytoplasmic marker, PfHsp70-1 and Maurer’s clefts marker, Pf332 reveal compartmentalization. Size bar corresponds to 1 mm.Issar N, Roux E, Mattei D, Scherf A. Identification of a novel post-translational modification in Plasmodium falciparum: protein sumoylation in different cellular compartments. Cell Microbiol. 2008 10:1999-2011.
See original on MMP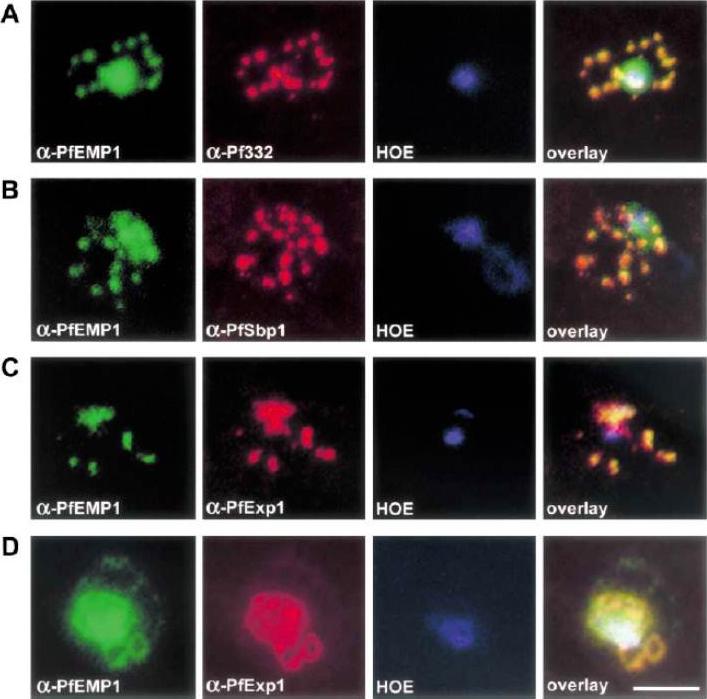
Co-localization of PfEMP1 with Pf332, PfSbp1 and PfExp-1 in P. falciparum-infected erythrocytes. Double immuno-fluorescence images of infected erythrocytes at the trophozoite stage with antibodies against: (A) PfEMP1 (a-PfEMP1) and Pf332 (a-Pf332); B) PfEMP1 (a-PfEMP1) and PfSbp1 (a-PfSbp1); (C, D) PfEMP1 (a-PfEMP1) and PfExp-1 (a-PfExp1). Different morphological appearances of the structures stained with antibodies against PfEMP1 and PfExp-1 are shown in (C and D). (HOE), DNA staining by Hoechst. (overlay), Overlay of the micrographs shown in a row. Representative examples of three independent experiments are shown. Bar, 5 mm.Wickert H, Wissing F, Andrews KT, Stich A, Krohne G, Lanzer M. Evidence for trafficking of PfEMP1 to the surface of P. falciparum-infected erythrocytes via a complex membrane network. Eur J Cell Biol. 2003 82:271-84.
See original on MMP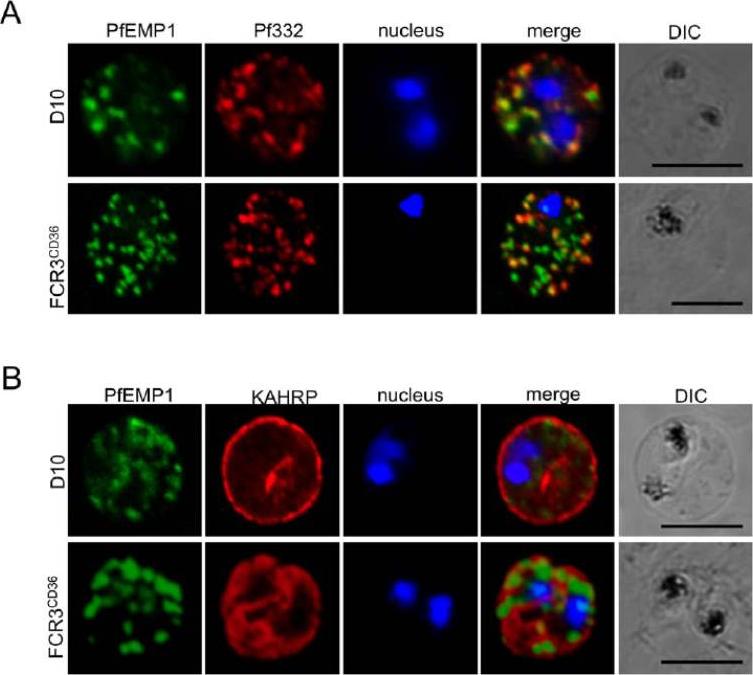
Localization of parasite exported proteins. A. Double-labelling of synchronous air-dried D10 (top row) and FCR3 panned on CD36 (FCR3CD36) (bottom row) with guinea pig anti-PfEMP1 antibodies (1:500) and purified rabbit anti-Pf332 IgG (1:200) revealed with a mixture of goat anti-guinea pig Alexa Fluor 488 (1:500) and Alexa Fluor 594-conjugated anti-rabbit IgG (1:1000). 332 (a Maurer’s clefts marker) and PfEMP1 were associated with the Maurer’s clefts in transit through the RBC cytoplasm. B. Labeling of PfEMP1 (1:500, green) and knobs on the PRBC surface with monoclonal anti-KAHRP (mAb89, 1:800, red), followed by anti-guinea pig Alexa Fluor 488 and anti-mouse Alexa Fluor 594. Scale bars: 5 mm. KAHRP, which interacts with the PfEMP1 ATS region, is correctly addressed to the RBC membrane.Nacer A, Roux E, Pomel S, Scheidig-Benatar C, Sakamoto H, Lafont F, Scherf A, Mattei D. clag9 is not essential for PfEMP1 surface expression in non-cytoadherent Plasmodium falciparum parasites with a chromosome 9 deletion. PLoS One. 2011;6(12):e29039.
See original on MMP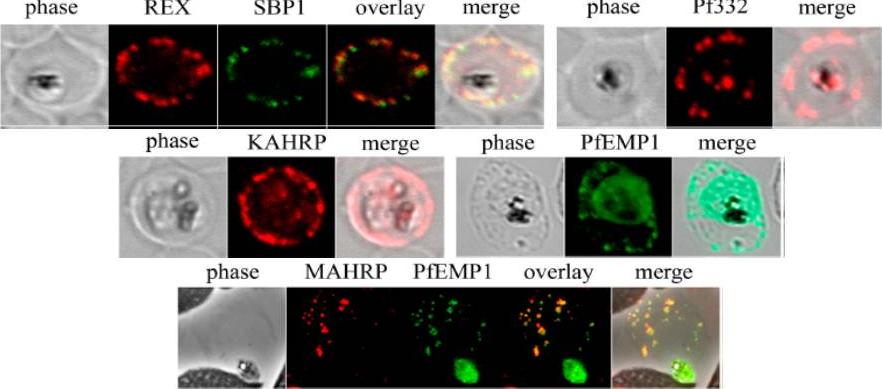
Analysis of expression and localization of Maurer’s cleft–associated proteins in IRBCs. Confocal immunofluorescence analysis of resident (REX-PFI1735c, SBP1-PFE0065w, and Pf332-PF11_0507) or transiently associated (KAHRP-PFB0100c and PfEMP1) Maurer’s cleft proteins. Colocalization of the resident Maurer’s cleft marker MAHRP-MAL13P1.413 with PfEMP1 in streptolysin O–pretreated IRBCs demonstrates that PfEMP1 is associated with Maurer’s clefts.Cooke BM, Buckingham DW, Glenister FK, Fernandez KM, Bannister LH, Marti M, Mohandas N, Coppel RL. A Maurer's cleft-associated protein is essential for expression of the major malaria virulence antigen on the surface of infected red blood cells. J Cell Biol. 2006 172:899-908.
See original on MMP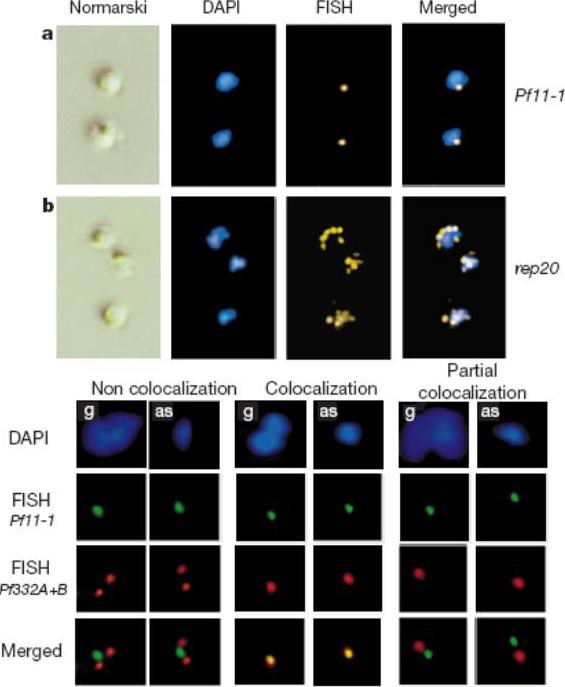
Upper panel: Physical clustering of P. falciparum subtelomeric regions detected in trophozoites detected by FISH. The first column of panels shows Nomarski images of parasites, the second shows fluorescent images after DAPI staining (blue), and the third shows FISH signals (yellow) from the single copy Pf11-1 gene probe or the P. falciparum telomere associated rep20 probe. The FISH and DAPI signals are superimposed in the fourth column.Lower panel: Promiscuous bouquet formation of P. falciparum chromosome ends. Colocalization FISH analysis of heterologous telomeres revealed with Pf11-1-specific (green) and Pf332-specific (red) gene probes. A typical example of an asexual (as) and gametocyte (g) of HB3 parasites is shown for each category.Freitas-Junior LH, Bottius E, Pirrit LA, Deitsch KW, Scheidig C, Guinet F, Nehrbass U, Wellems TE, Scherf A. Frequent ectopic recombination of virulence factor genes in telomeric chromosome clusters of P. falciparum. Nature. 2000 407(6807):1018-10122.
See original on MMP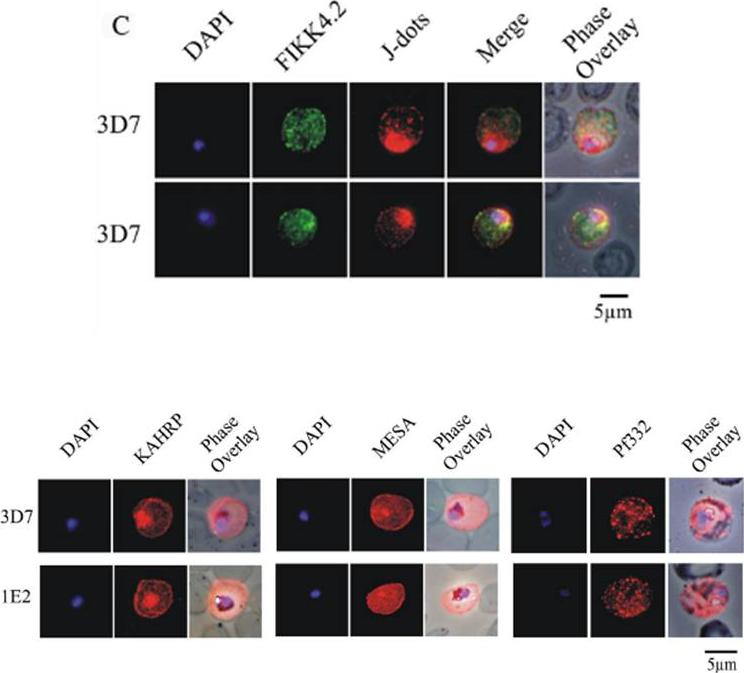
(C) Immunofluorescence analysis of red blood cells infected with parental 3D7 parasites co-labelled with a-FIKK4.2 and a-PFB0090c (J-dots) antibodies. Merge and phase overlay images indicate that FIKK4.2 and J-dots do not co-localise in infected red blood cells. In all panels, parasite nuclei are counterstained with DAPI. (D) Immunofluorescence analysis of red blood cells infected with either parental 3D7 or FIKK4.2 knockout (1E2) parasites. Infected red blood cells were incubated with antibodies raised against the knob-associated histidine-rich protein (KAHRP), mature-parasite-infected erythrocyte surface antigen (MESA) or P. falciparum antigen 332 (Pf332). Parasite nuclei were counterstained with DAPI. KAHRP, MESA and Pf332 as three examples of other very well described P. falciparum-encoded exported proteins.Kats LM, Fernandez KM, Glenister FK, Herrmann S, Buckingham DW, Siddiqui G, Sharma L, Bamert R, Lucet I, Guillotte M, Mercereau-Puijalon O, Cooke BM. An exported kinase (FIKK4.2) that mediates virulence-associated changes in Plasmodium falciparum-infected red blood cells. Int J Parasitol. 2014 Feb 14. [Epub ahead of print]
See original on MMP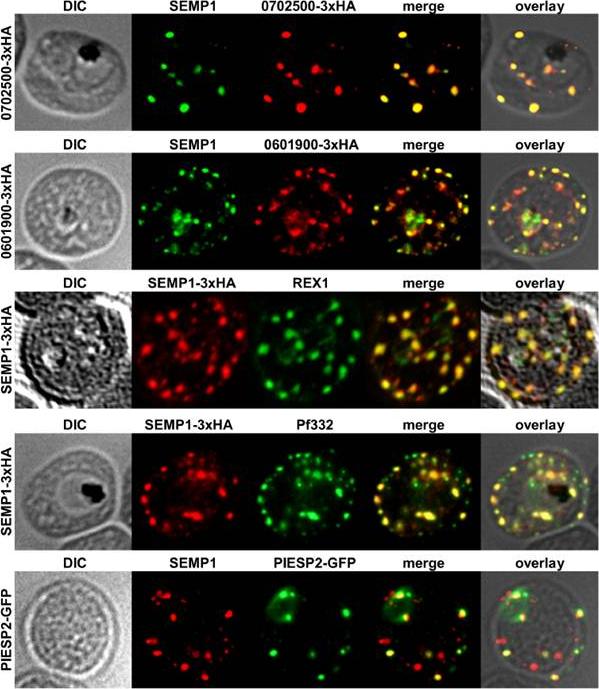
Localization of potential SEMP1 interacting proteins. Immunofluorescence assays of MeOH-fixed RBCs infected with 3D7 expressing EMP1/PF3D7_0702500 (PF07_0008)/PF3D7_0601900 (PFF0090w) with a C-terminal 3xHA tag and 3D7 expressing PIESP2 with a C-terminal GFP-tag, co-labelled with mouse a-SEMP1 and rat a-HA (PF3D7_0702500-3xHA & PF3D7_0601900-3xHA) /a-GFP (PIESP2-GFP). For co-labelling of SEMP1-3xHA with REX1 and Pf332, rat a-HA and rabbit a-REX1 / mouse a-Pf332 antibodies were used. All potential interaction partners co-localized at least partially with SEMP1, but in particular with Pf332 or PIESP2 antibodies only a fraction of SEMP1 positive MCs were labelled with either antibody suggesting that distinct subpopulations of MCs exist. Dietz O, Rusch S, Brand F, Mundwiler-Pachlatko E, Gaida A, Voss T, Beck HP. Characterization of the Small Exported Plasmodium falciparum Membrane Protein SEMP1. PLoS One. 2014 9(7):e103272.
See original on MMPMore information
| PlasmoDB | PF3D7_1149000 |
| GeneDB | PF3D7_1149000 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | PF11_0506, PF11_0507 |
| Orthologs | |
| Google Scholar | Search for all mentions of this gene |