PF3D7_0936000 ring-exported protein 2 (REX2)
Disruptability [+]
| Species | Disruptability | Reference | Submitter |
|---|---|---|---|
| P. falciparum 3D7 |
Refractory |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen |
Mutant phenotypes [+]
None reported yet. Please press the '+' button above to add one.Imaging data (from Malaria Metabolic Pathways)
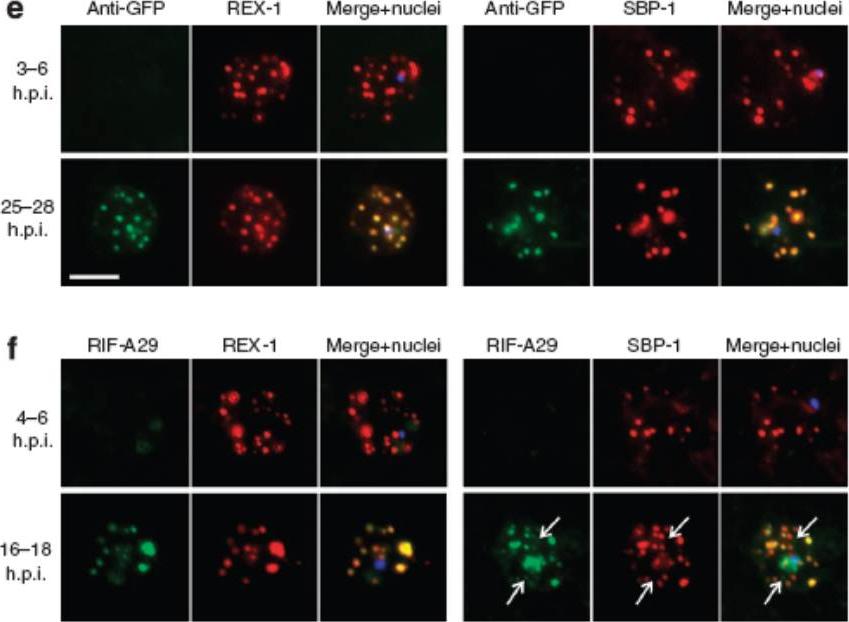
Export of membrane proteins is independent of cleft formation. IFA showing that in REX2-GFPcrt-expressing parasites (e) and 3D7 (f), early clefts contain REX1 and but not REX2-GFPcrt (e, upper panels) or RIFIN A29 (RIF-A29, f, upper panels). On progress of the culture to the trophozoite stage, the clefts containing REX1 and now also contain REX2-GFPcrt (e, lower panel) and RIFIN A29 (f, lower panel). Dots positive for RIFIN A29 only, found in ~50% of cells, are indicated by white arrows. Scale bars, 5 μm; h.p.i., hours post invasion. the clefts in early stages contained only the early markers (e,f). In contrast, later stages were positive for all four antigens (e,f). Crucially, in later stages all antigens occupied the same clefts (e,f), demonstrating that the clefts initially containing REX1 and SBP1 accumulated the other proteins later on. This establishes that export occurred without cleft formation.Grüring C, Heiber A, Kruse F, Ungefehr J, Gilberger TW, Spielmann T. Development and host cell modifications of Plasmodium falciparum blood stages in four dimensions. Nat Commun. 2011 2:165.
See original on MMP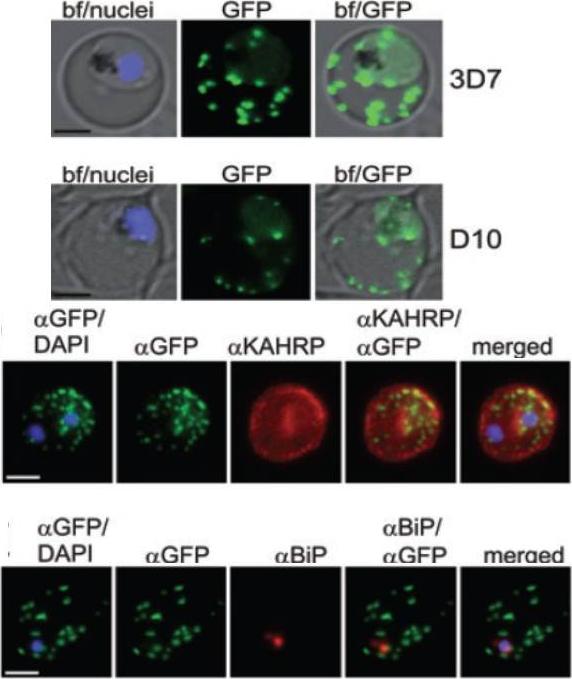
A and B. GFP fluorescence in live 3D7 (A) or D10 (B) parasites expressing wild-type REX2GFP from an episomal vector. GFP fluorescence appears as dots in the host cell cytoplasm typical for Maurer’s cleft localization. Nuclei are stained with DAPI (blue); merges with bright field images (bf) are shown. D. IFA of acetone-fixed infected RBCs expressing REX2GFP and stained with anti-GFP (green) and anti-KAHRP (red) antibodies. Overlays are shown as indicated above each panel. The last panel shows an overlay of all three images (merged). E. IFA of acetone-fixed infected RBCs expressing REX2GFP and stained with anti-GFP (green) and anti-BiP (red) antibodies. Overlays of panels are shown as in (D). Size bars are 2 mm.Haase S, Herrmann S, Grüring C, Heiber A, Jansen PW, Langer C, Treeck M, Cabrera A, Bruns C, Struck NS, Kono M, Engelberg K, Ruch U, Stunnenberg HG, Gilberger TW, Spielmann T. Sequence requirements for the export of the Plasmodium falciparum Maurer's clefts protein REX2. Mol Microbiol. 2009 71:1003-17. Copyright John Wiley & Sons Ltd. 2010.
See original on MMP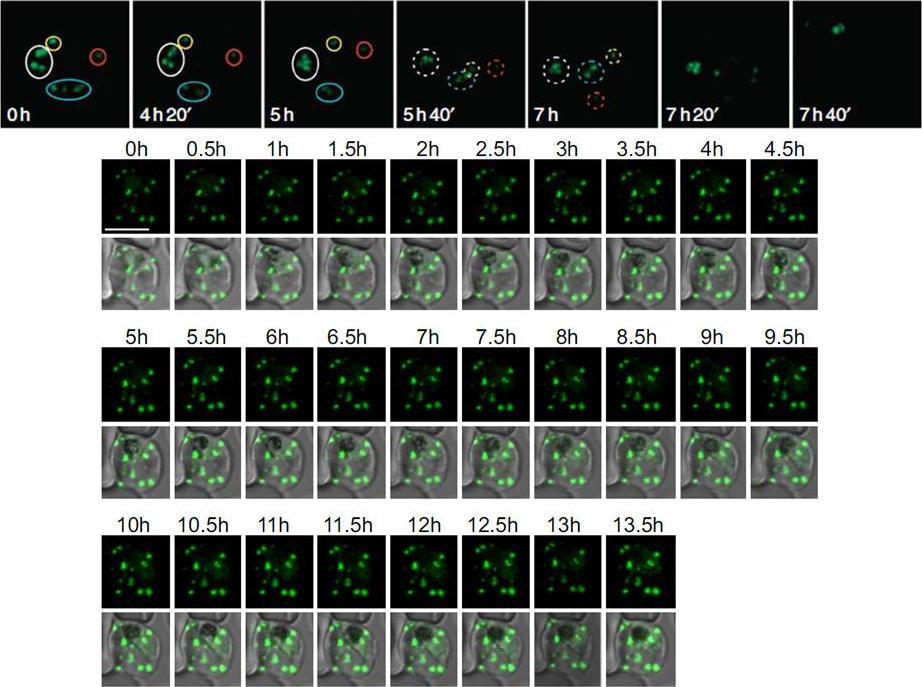
Upper panel: Time of merozoite formation and three states of Maurer’s clefts. Selected time points of 4D imaging (20 min interval) of a REX2-GFP-expressing schizont. Upper rows: maximum intensity projection of 3D reconstructions of GFP fluorescence labelling Maurer’s clefts; bottom rows: single DIC z-section of the corresponding time point. Different groups of clefts are marked with colour-coded circles to show their changing position. The broken lines encircle clefts that cannot be assigned with certainty. In the last time point before rupture, no assignment could be made. Scale bars, 5 μm. REX2-GFP was only detectable from the young trophozoite stage onwards. Unexpectedly, follow-up of these cells through the remainder of the cycle showed that the position and number of Maurer’s clefts in individual cells remained constant until shortly before rupture. Time lapse analysis of the Maurer's clefts in an erythrocyte infected with a parasite expressing REX2-GFPcrt.Grüring C, Heiber A, Kruse F, Ungefehr J, Gilberger TW, Spielmann T. Development and host cell modifications of Plasmodium falciparum blood stages in four dimensions. Nat Commun. 2011 2:165.
See original on MMP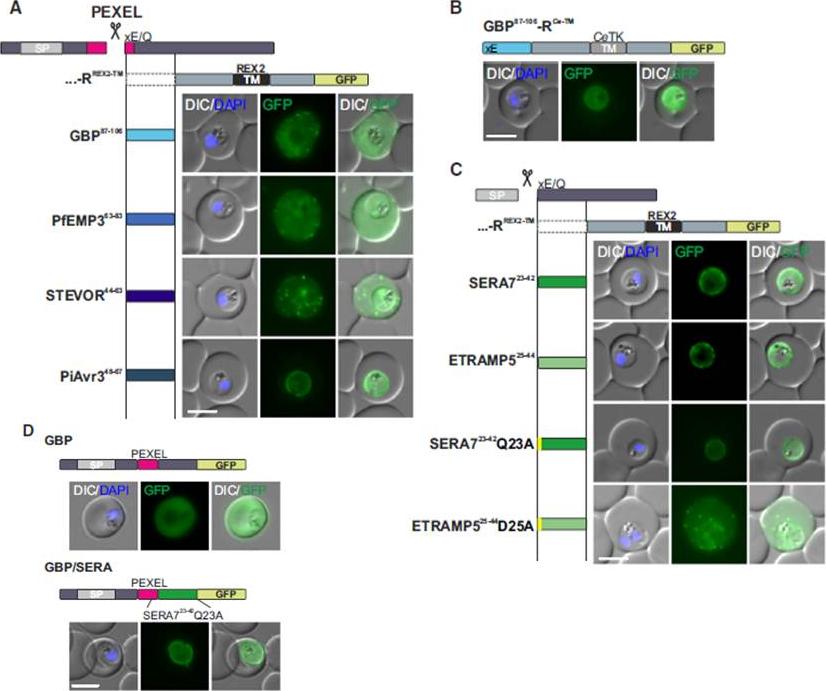
Mature PEXEL N Termini Promote Export of RREX2-TM (A–C) Images of live P. falciparum parasites expressing RREX2-TM fused with the mature N termini of PEXEL proteins (A), GBP87–106-RCe-TM (B), or RREX2-TM fused with the mature N termini of nonexported secretory proteins (C). The position of the appended region in the original protein is shown above each panel.(D) Images of live P. falciparum parasites expressing truncated GBP fused to GFP (GBP, top) or GBP-GFP with the mature N terminus of SERA7Q23A after the PEXEL (GBP/SERA, bottom). Although these N termini contain only the last of the conserved PEXEL residues (PEXEL position 5) and the nonconserved position 4, they promoted export of the reporter into the host cell (A). GFP fluorescence was detected in the erythrocyte cytosol and the Maurer’s clefts.Grüring C, Heiber A, Kruse F, Flemming S, Franci G, Colombo SF, Fasana E,Schoeler H, Borgese N, Stunnenberg HG, Przyborski JM, Gilberger TW, Spielmann T. Uncovering common principles in protein export of malaria parasites. Cell Host Microbe. 2012 12(5):717-29.
See original on MMP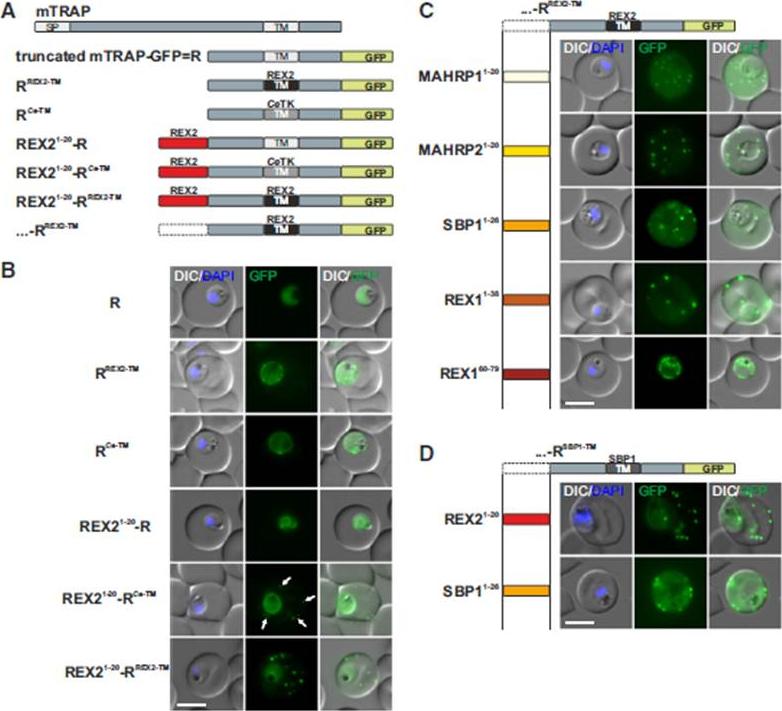
N Termini of PNEPs Are Sufficient for Export of RREX2-TM, (A) Schematic of mTRAP fusion constructs. (B) Representative images of live P. falciparum parasites expressing the constructs shown in (A). DIC, differential interference contrast; nuclei were stained with DAPI. Arrows indicate limited staining reminiscent of Maurer’s clefts. (C and D) Images of live P. falciparum parasites expressing RREX2-TM (C) and RSBP1-TM (D) fused with the PNEP N termini indicated. Panels are as in (B). Size bars represent 5 mm. The mTRAP backbone, different TMs, and the SP (signal peptide) are shown in different shades of gray; N-terminally appended regions of PNEPs are shown in shades from yellow to dark red. Ce-TM, C. elegans TK TM. The unmodified truncated mTRAP fused to GFP (‘‘R’’ for ‘‘reporter’’) does not contain any export-relevant sequences. This protein was found evenly distributed in the parasite cytosol (B). When the mTRAP TM in R was replaced with that of the PNEP REX2 or a previously used heterologous TM of a C.elegans tyrosine kinase (TK) (a TM that does not promote export of REX2 and does not change topology;), the R then entered the secretory pathway and was found in the parasite periphery (PPM, PV, or PVM) but was not exported.Grüring C, Heiber A, Kruse F, Flemming S, Franci G, Colombo SF, Fasana E,Schoeler H, Borgese N, Stunnenberg HG, Przyborski JM, Gilberger TW, Spielmann T. Uncovering common principles in protein export of malaria parasites. Cell Host Microbe. 2012 12(5):717-29.
See original on MMP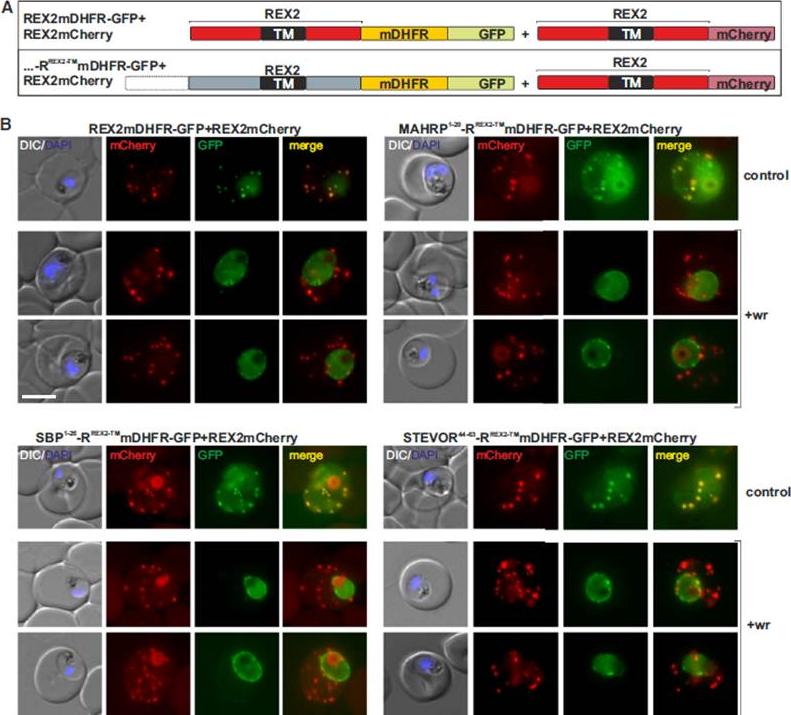
Blocking Unfolding Arrests Export of PNEPs. (A) Schematic of the constructs of the cell lines shown in (B). (B) Parasites expressing REX2mCherry (red) together with the mDHFR-GFP-tagged constructs (green) indicated above each panel. Two images are shown per cell line in the WR99210 (+wr)-treated samples to demonstrate that in different cells the parasite peripheral staining of the blocked protein displayed either a smooth or a more focal pattern. Merge, overlay of the red and green signals. The size bar represents 5 mm.Grüring C, Heiber A, Kruse F, Flemming S, Franci G, Colombo SF, Fasana E, Schoeler H, Borgese N, Stunnenberg HG, Przyborski JM, Gilberger TW, Spielmann T. Uncovering common principles in protein export of malaria parasites. Cell Host Microbe. 2012 12(5):717-29.
See original on MMP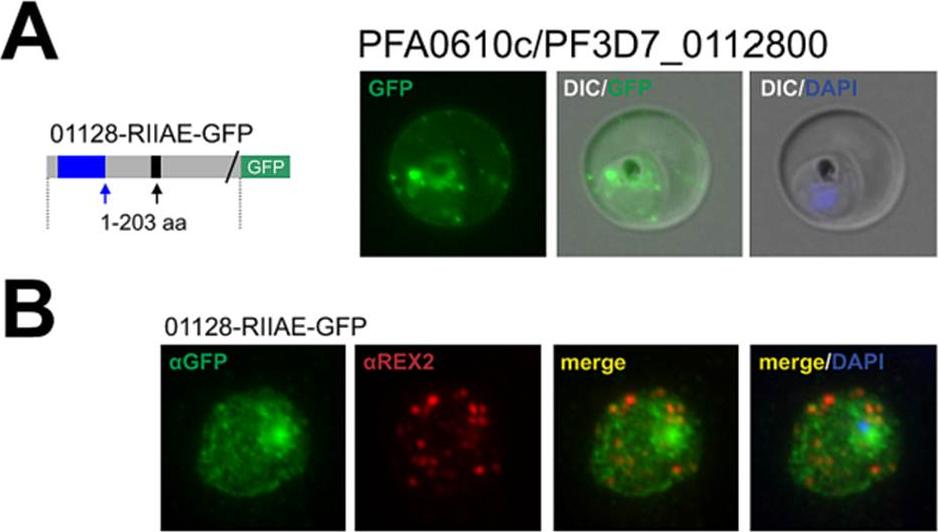
The hyp11 protein encoded by PFA0610c is an exported protein with a non-canonical PEXEL/HT. A. Fluorescence microscopy. The schematic structure of the GFP-tagged proteins is depicted on the left of representative images of iRBCs.The ER-signal is shaded blue, the (non-) canonical PEXEL/HT is shaded black and GFP is shaded green. Putative cleavage sites are indicated by an arrow (blue: SP cleavage site; black: Plasmepsin V cleavage site. B. Immunofluorescence analyses. Antibodies against GFP and the Maurer’s clefts protein were used. Membranes were stained with Bodipy-TR-C5ceramide. The fluorescence of GFP-tagged PFA0610c (01128-RIIAE-GFP), a protein with a R.I.E variant, appeared to be located close to the RBC membrane (A). 01128-RIIAE-GFP did not colocalize with REX2 (B).This indicates that PFA0610cis associated with the RBC membrane. Schulze J, Kwiatkowski M, Borner J, Schlüter H, Bruchhaus I, Burmester T, Spielmann T, Pick C. The Plasmodium falciparum exportome contains non-canonical PEXEL/HT proteins. Mol Microbiol. 2015 Apr 8. [Epub ahead of print]
See original on MMP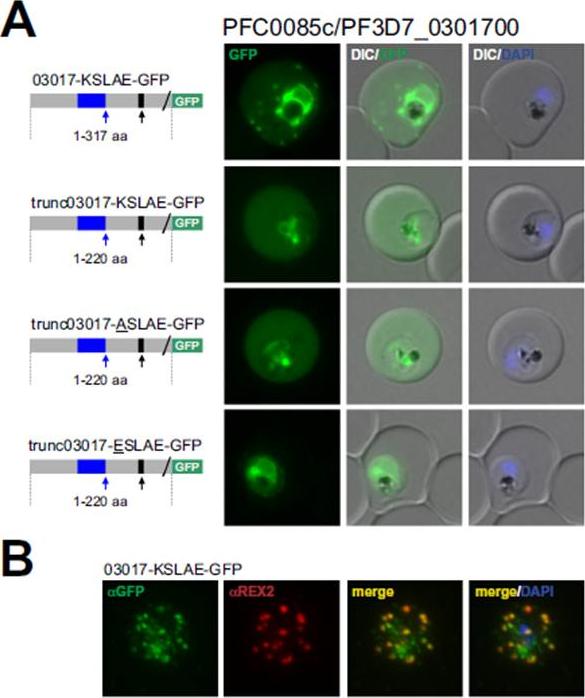
PFC0085c encodes an exported protein with a non-canonical PEXEL/HT. A. Fluorescence microscopy. The schematic structure of the GFP-tagged proteins is depicted on the left of representative images of iRBCs. B. Immunofluorescence analyses. Antibodies against GFP and the Maurer’s clefts protein REX2 were used. The ER-signal is shaded blue, the (non-) canonical PEXEL/HT is shaded black and GFP is shaded green. Putative cleavage sites are indicated by an arrow (blue: SP cleavage site; black: Plasmepsin V cleavage site. Fluorescence of GFP-tagged PFC0085c (03017-KSLAE-GFP), a protein with a K.L.E variant, indicated a Maurer’s clefts localization in addition to a uniformly distributed pool in the iRBC cytoplasm (A). This was confirmed by immunofluorescence analyses, demonstrating that 03017-KSLAE-GFP colocalized with REX2 (B).Schulze J, Kwiatkowski M, Borner J, Schlüter H, Bruchhaus I, Burmester T, Spielmann T, Pick C. The Plasmodium falciparum exportome contains non-canonical PEXEL/HT proteins. Mol Microbiol. 2015 Apr 8. [Epub ahead of print]
See original on MMP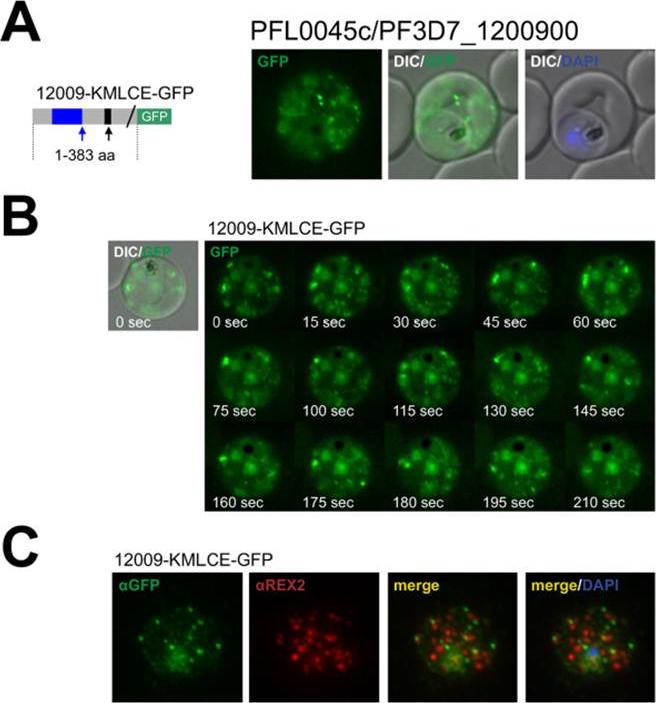
The PHISTc protein encoded by PFL0045c/PF3D7_1200900 is an exported protein with a non-canonical PEXEL/HT. A. Fluorescence microscopy. The schematic structure of the GFP-tagged proteins is depicted on the left of representative images of iRBCs The ER-signal is shaded blue, the (non-) canonical PEXEL/HT is shaded black and GFP is shaded green. Putative cleavage sites are indicated by an arrow (blue: SP cleavage site; black: Plasmepsin V cleavage site. B. Fluorescence microscopy. A representative time lapse series is given. C. Immunofluorescence analyses. Antibodies against GFP and the Maurer’s clefts protein REX2 were used. Fluorescence of GFPtagged PFL0045c (12009-KMLCEGFP), a protein with a K.L.E variant, was restricted to numerous small dots in the iRBC cytoplasm (A) that moved rapidly (B). Congruently, 12009-KMLCE-GFP did not colocalize with REX2 (C), indicating that PFL0045c is localized to structures that are similar to J-dotsSchulze J, Kwiatkowski M, Borner J, Schlüter H, Bruchhaus I, Burmester T, Spielmann T, Pick C. The Plasmodium falciparum exportome contains non-canonical PEXEL/HT proteins. Mol Microbiol. 2015 Apr 8. [Epub ahead of print] PMID:
See original on MMP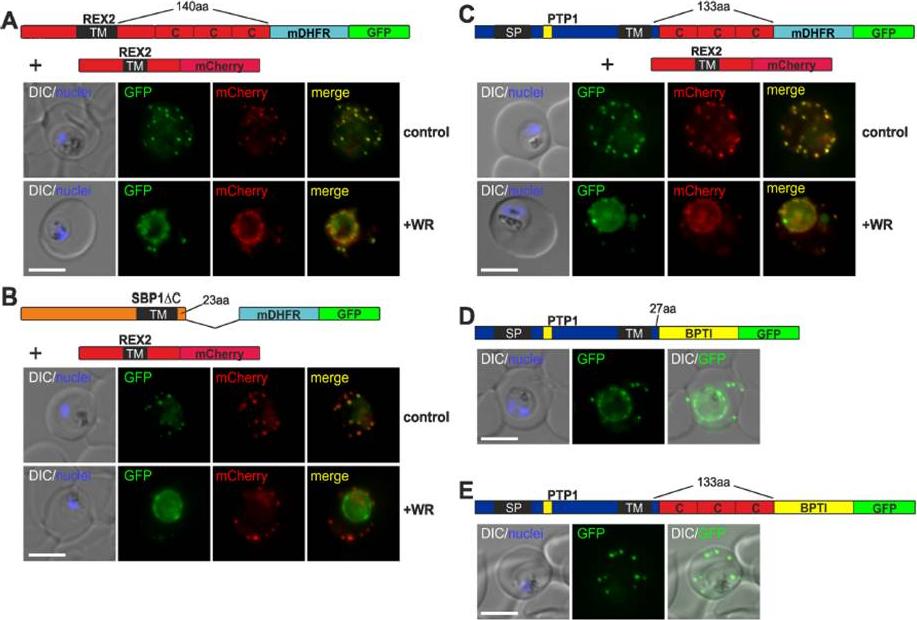
The distance between blocking domain and TM is important for co-blocking and BPTI sensitivity. (A-E) Representative images of live P. falciparum parasites expressing the constructs shown schematically above each panel (numbers refer to the length of the amino acids sequence between TM and blocking domain). (A-C) Parasites grown in the presence (+WR) or absence of WR (control). DIC, differential interference contrast. Size bars: 5 μmMesén-Ramírez P, Reinsch F, Blancke Soares A, Bergmann B, Ullrich AK, Tenzer S, Spielmann T. Stable Translocation Intermediates Jam Global Protein Export in Plasmodium falciparum Parasites and Link the PTEX Component EXP2 with Translocation Activity. PLoS Pathog. 2016 May 11;12(5):e1005618. “
See original on MMP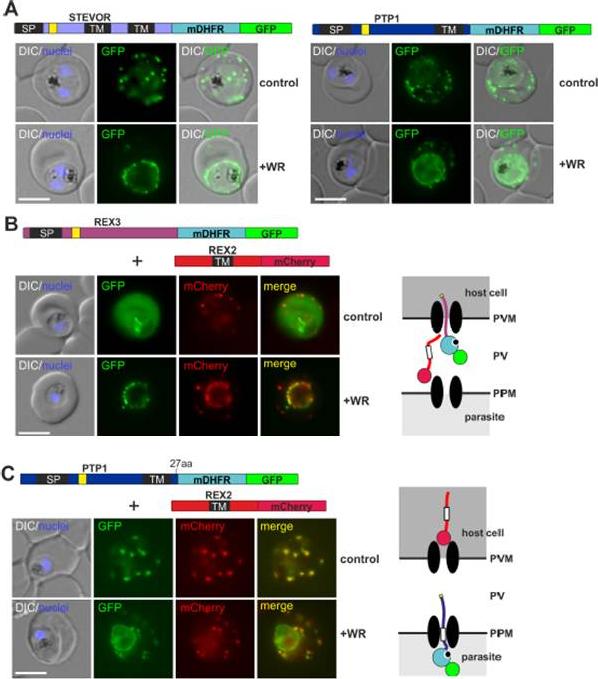
PEXEL TM proteins require translocation for export and some PEXEL proteins can also jam the translocon. (A-C) Representative images of live P. falciparum parasites expressing the constructs shown schematically above each panel (numbers refer to the length of the amino acids sequence between TM and blocking domain). DIC, differential interference contrast. Size bars: 5 μm. A schematic of the co-block (B) and failure to coblock (C) are shown to the right.Mesén-Ramírez P, Reinsch F, Blancke Soares A, Bergmann B, Ullrich AK, Tenzer S, Spielmann T. Stable Translocation Intermediates Jam Global Protein Export in Plasmodium falciparum Parasites and Link the PTEX Component EXP2 with Translocation Activity. PLoS Pathog. 2016 May 11;12(5):e1005618
See original on MMP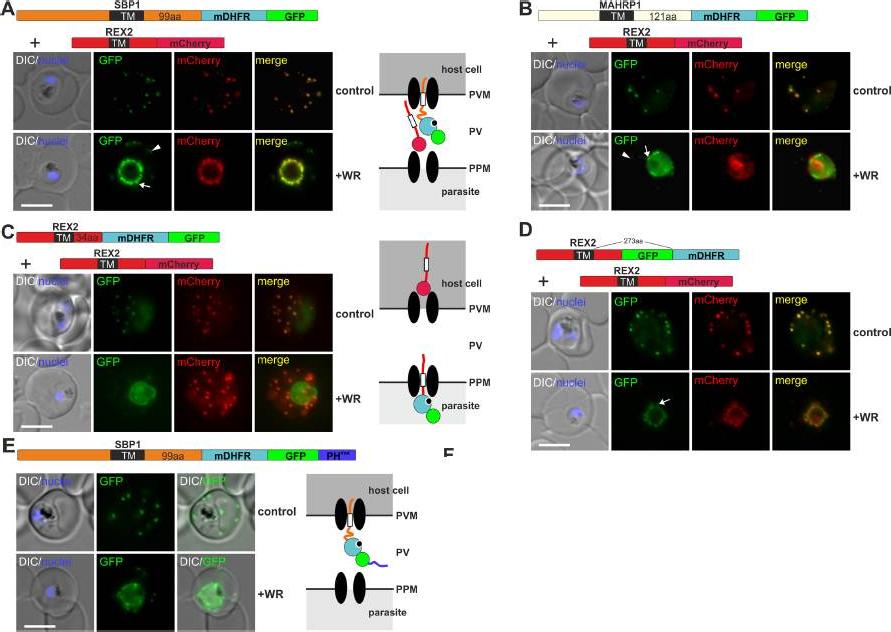
Mouse DHFR fusions can clog the PVM translocon and co-block the export of other proteins. (A-E) Representative images of live P. falciparum parasites grown in the presence (+WR) or absence of WR (control) and expressing the constructs shown schematically above each panel. DIC, differential interference contrast. Arrowheads indicate faint signals at the Maurer’s clefts; arrows show mobile protrusions (note that they do not overlap in the red and the green signal due to movement between capture of the images). Size bars: 5 μm. Schematics to the right show the location of the fusion protein containing the folded WR-bound mDHFR domain (light blue circle with smaller black circle in binding pocket) and the co-expressed REX2 (red line) fused to mCherry (red circle); white box, TM; green circle, GFP; blue line, protease sensitive mutated PH domain.Mesén-Ramírez P, Reinsch F, Blancke Soares A, Bergmann B, Ullrich AK, Tenzer S, Spielmann T. Stable Translocation Intermediates Jam Global Protein Export in Plasmodium falciparum Parasites and Link the PTEX Component EXP2 withTranslocation Activity. PLoS Pathog. 2016 May 11;12(5):e1005618.
See original on MMP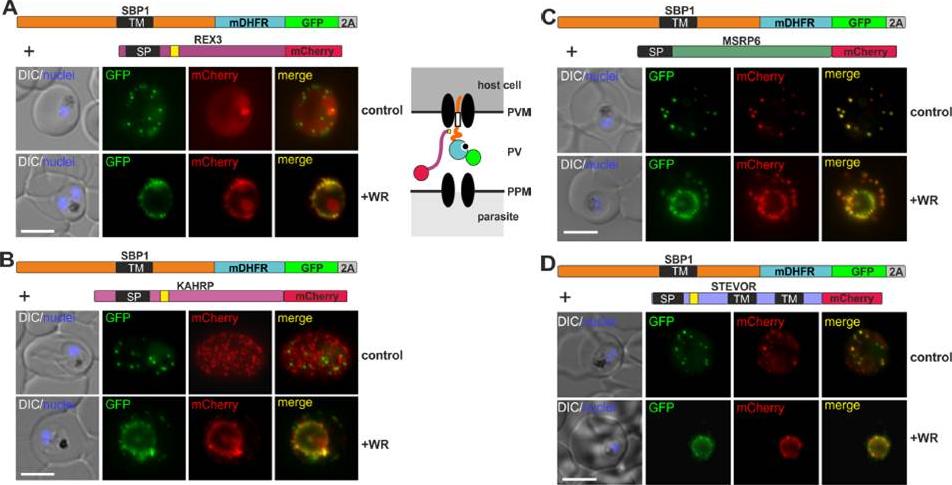
Different types of proteins pass through the same translocon. (A-D) Representative images of live P. falciparum parasites grown in the presence (+WR) or absence of WR (control), expressing the constructs shown schematically above each panel. The skip peptide is indicated by a grey box labelled 2A. The two proteins expressed from the same open reading frame are shown skipped. DIC, differential interference contrast. Size bars: 5 μm. A schematic of the co-block is shown for A. The yellow box indicates the mature PEXEL.Mesén-Ramírez P, Reinsch F, Blancke Soares A, Bergmann B, Ullrich AK, Tenzer S, Spielmann T. Stable Translocation Intermediates Jam Global Protein Export in Plasmodium falciparum Parasites and Link the PTEX Component EXP2 with Translocation Activity. PLoS Pathog. 2016 May 11;12(5):e1005618.
See original on MMP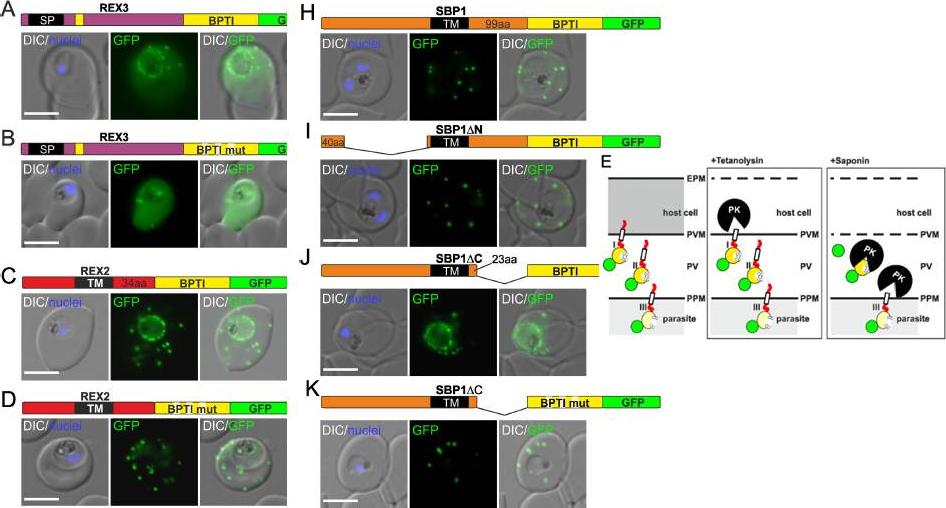
Fusion with the redox sensitive BPTI reveals a second translocation step for TM proteins. (A-D and F-L) Representative images of live P. falciparum parasites expressing the constructs shown schematically above each panel. Hydrophobic regions (SP, signal peptide; TM, transmembrane domain) are in black, the PEXEL motif in yellow. Numbers refer to amino acids (aa). Red boxes labelled C, additional REX2 C-termini. Interrupted yellow box, mutated BPTI (BPTImut). DIC, differential interference contrast. Size bars: 5 μm. (E) Schematic for the protease K (PK) protection assay. Left, intact infected RBC with 3 possibilities (I, II, III) for the location of the fusion construct: I, protein is integral to PVM; II, protein is freely accessible in the PV; III, protein is integral to PPM. Middle, after permeabilisation of the erythrocyte plasma membrane (EPM) with tetanolysin the N-terminus of the construct will be digested if it is in the PVM (I), but remains intact in situation II and III. Right, after permeabilisation of the PVM with saponin, the constructs will be digested if it is in the PVM (I) or the PV (II) but if in the PPM (III), an N-terminally truncated fragment will be generated. Red, exported protein; white box, TM; yellow, BPTI with double cysteine bonds; green, GFP. Mesén-Ramírez P, Reinsch F, Blancke Soares A, Bergmann B, Ullrich AK, Tenzer S, Spielmann T. Stable Translocation Intermediates Jam Global Protein Export in Plasmodium falciparum Parasites and Link the PTEX Component EXP2 withTranslocation Activity. PLoS Pathog. 2016 May 11;12(5):e1005618.
See original on MMPMore information
| PlasmoDB | PF3D7_0936000 |
| GeneDB | PF3D7_0936000 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | PFI1740c |
| Orthologs | |
| Google Scholar | Search for all mentions of this gene |