PF3D7_0935900 ring-exported protein 1 (REX1)
Disruptability [+]
| Species | Disruptability | Reference | Submitter |
|---|---|---|---|
| P. falciparum 3D7 |
Possible |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen |
| P. falciparum 3D7 |
Possible |
21696460 \"We have generated mutants in which the Maurer\'s cleft protein, the ring exported protein-1 (REX1) is truncated or deleted. Removal of the C-terminal domain of REX1 compromises Maurer\'s cleft architecture and PfEMP1-mediated cytoadherance but permits some trafficking of PfEMP1 to the erythrocyte surface. Deletion of the coiled-coil region of REX1 ablates PfEMP1 surface display, trapping PfEMP1 at the Maurer\'s clefts.\" |
Theo Sanderson, Francis Crick Institute |
Mutant phenotypes [+]
None reported yet. Please press the '+' button above to add one.Imaging data (from Malaria Metabolic Pathways)
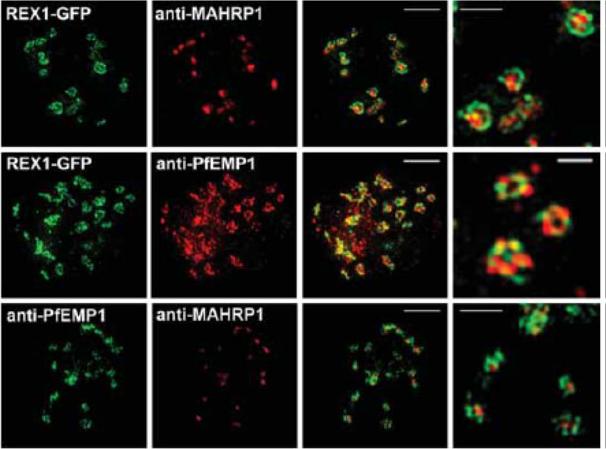
3D-SIM analysis of the organization of Maurer's cleft resident proteins. REX1-GFP transfectants were permeabilized with EqtII and labeled with antibodies recognizing GFP, PfEMP1 (ATS) or MAHRP1 and 3D stacks were generated by 3D-SIM. Higher magnification images at right. Scale bars = 2 μm, first 3 columns; 500 nm, last column.McMillan PJ, Millet C, Batinovic S, Maiorca M, Hanssen E, Kenny S, Muhle RA, Melcher M, Fidock DA, Smith JD, Dixon MW, Tilley L. Spatial and temporal mapping of the PfEMP1 export pathway in Plasmodium falciparum. Cell Microbiol. 2013 15(8):1401-18
See original on MMP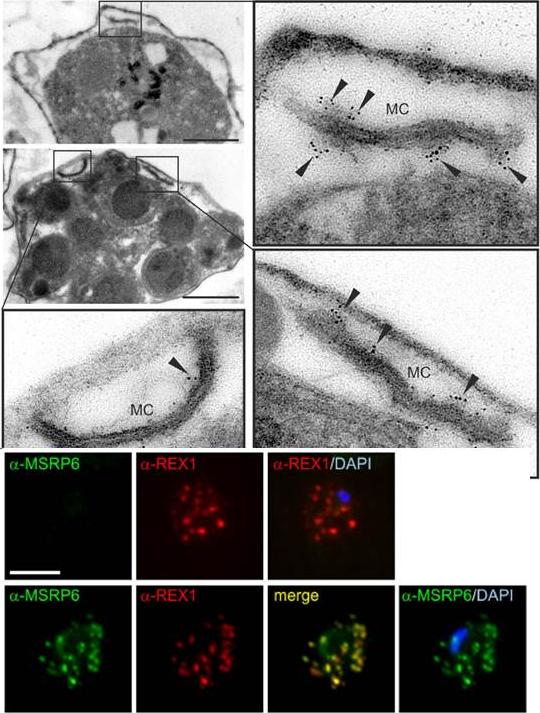
MSRP6 is a Maurer’s clefts protein expressed in trophozoites and schizonts. Pre-embedding immuno-EM of infected RBC, where the host cell cytosol has been released with tetanolysin, reacted with anti-MSRP6 antisera. Small panels show overviews with boxes highlighting individual Maurer’s clefts for which enlargements are shown. Gold label is indicated with arrowheads. Size bars: 1 mm. MSRP6 was detected in ‘cloudy’ structures at the outside of Maurer’s clefts. Lower panel: Co-localisation IFA using anti-REX1 and anti-MSRP6 antibodies with MSRP6 knock out parasites (top) and parental 3D7 parasites (bottom). Size bar: 5 mm. MSRP6 antiserum recognized foci in infected host cells representing Maurer’s clefts.Heiber A, Kruse F, Pick C, Grüring C, Flemming S, Oberli A, Schoeler H, Retzlaff S, Mesén-Ramírez P, Hiss JA, Kadekoppala M, Hecht L, Holder AA, Gilberger TW, Spielmann T. Identification of New PNEPs Indicates a Substantial Non-PEXEL Exportome and Underpins Common Features in Plasmodium falciparum Protein Export. PLoS Pathog. 2013 9(8):e1003546
See original on MMP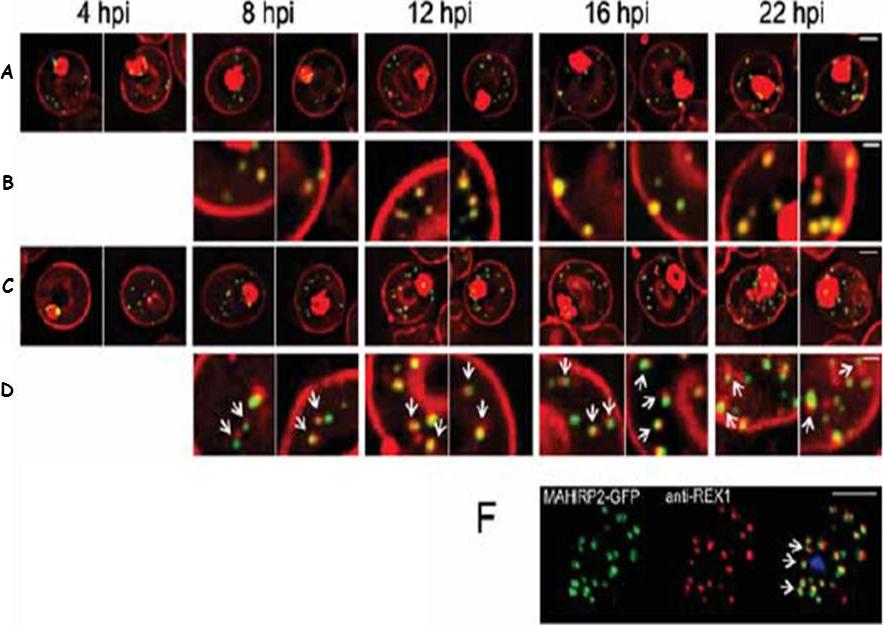
Live cell time course showing assembly of exomembrane components. Transfectants expressing GFP chimeras of (A, B) REX1 and (C, D) MAHRP2 were co-labeled with BODIPYceramide. Infected RBCs were synchronized to a ~2 h window and samples were collected at 4 h intervals. BODIPY-ceramide- and REX1-GFP-labeled Maurer's clefts and MAHRP2-GFP-labled structures are evident in the RBC cytoplasm from ~4 h post invasion. MAHRP2-GFP-labled structures are associated with the BODIPY-ceramide-labeled Maurer's clefts (D, white arrows). (F) MAHRP2-GFP transfectants were permeabilized with EqtII and labeled with antibodies recognizing REX1. Scale bars = 3 μm (A, C), 1 μm (B, D) and 4 μm (F). McMillan PJ, Millet C, Batinovic S, Maiorca M, Hanssen E, Kenny S, Muhle RA, Melcher M, Fidock DA, Smith JD, Dixon MW, Tilley L. Spatial and temporal mapping of the PfEMP1 export pathway in Plasmodium falciparum. Cell Microbiol. 2013 15(8):1401-18.
See original on MMP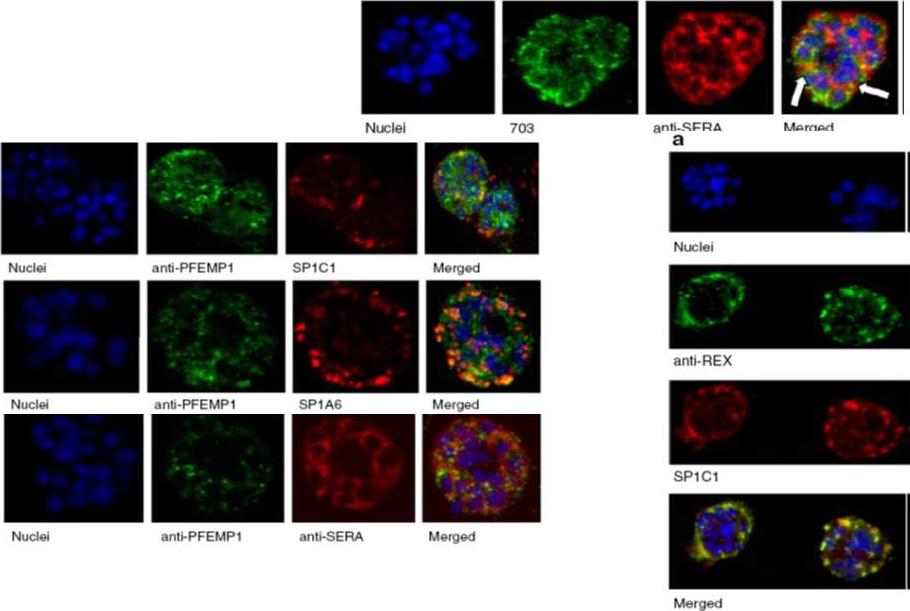
Upper row: Immunofluorescence assay of rabbit antisera against PfMC-2TM peptides colocalized with anti-SERA antibodies. Arrows indicate areas of colocalization between SERA and PfMC-2TM. Left Panel: Immunofluorescence assay of rabbit antisera against PfEMP1 with Mabs SP1C1 (a), Mab SP1A6 (b), and anti-SERA (c). PfEMP1 colocalized with PfMC-2TM, the 130 kDa Maurer’s cleft protein and SERA. Right panel: Immunofluorescence assay of rabbit antisera against REX PFI1735c colocalized with Mabs SP1C1. Tsarukyanova I, Drazba JA, Fujioka H, Yadav SP, Sam-Yellowe TY. Proteins of the Plasmodium falciparum two transmembrane Maurer's cleft protein family, PfMC-2TM, and the 130 kDa Maurer's cleft protein define different domains of the infected erythrocyte intramembranous network. Parasitol Res. 2009 104(4):875-91.
See original on MMP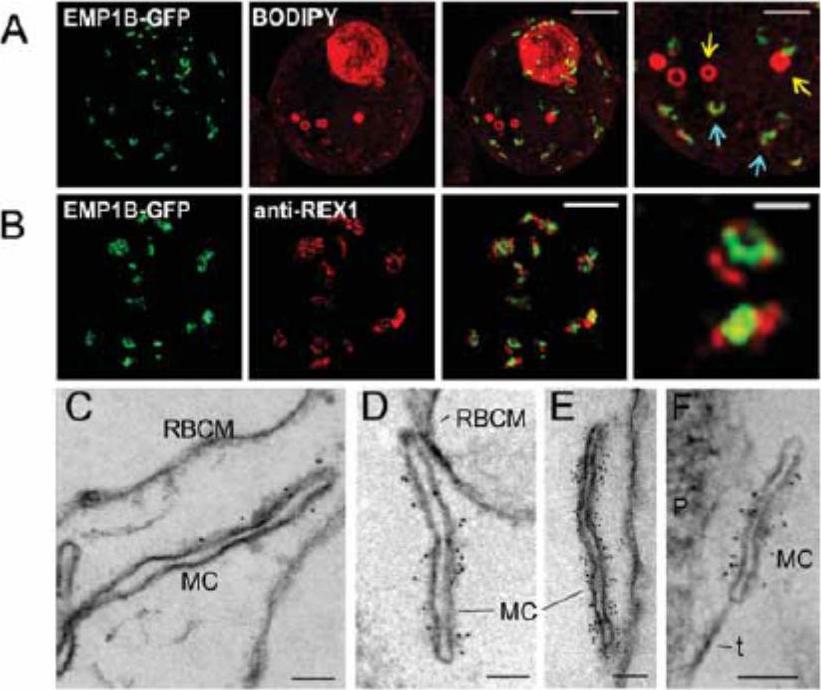
Characterization of PfEMP1B-GFP at Maurer's clefts. (A) Transfectants (ITK strain) expressing the PfEMP1B-GFP chimera were co-labeled with BODIPY-ceramide and imaged using 3DSIM. Higher magnification images are displayed in the right column. PfEMP1B-GFP is present in horseshoe-shaped Maurer's clefts (aqua arrows). Spherical structures with no GFP labeling are marked with yellow arrows. (B) EqtII-permeabilized PfEMP1B-GFP transfectants were probed with antibodies recognizing GFP and REX1 and imaged by 3D-SIM. (C-F) EqtII-permeabilized 3D7 strain or PfEMP1B-GFP transfectants were labeled with anti-PfEMP1 ATS (C) or anti-GFP (D-F) and 6 nm Protein A-gold, and prepared for EM. A tether (t) and the parasite (P) are indicated. Scale bars = 2 μm (A, first three columns, B), 1 μm (A, last column), 500 nm (B, last column), 100 nm (C-F).McMillan PJ, Millet C, Batinovic S, Maiorca M, Hanssen E, Kenny S, Muhle RA, Melcher M, Fidock DA, Smith JD, Dixon MW, Tilley L. Spatial and temporal mapping of the PfEMP1 export pathway in Plasmodium falciparum. Cell Microbiol. 2013 15(8):1401-18.
See original on MMP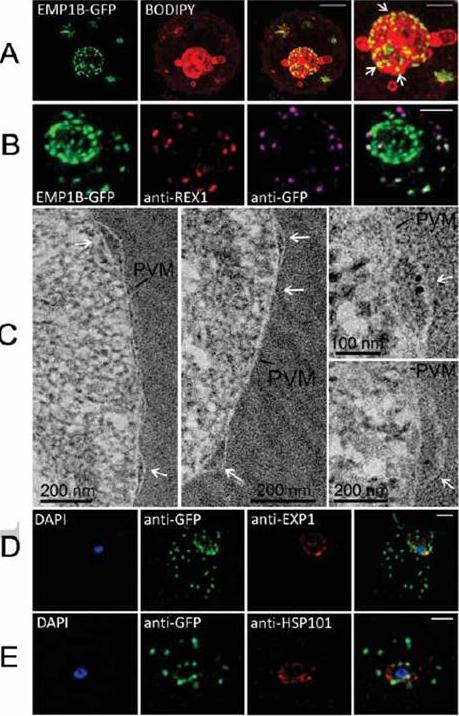
Characterization of an intermediate compartment in PfEMP1 export. (A) Transfectants (ITK strain) expressing the PfEMP1B-GFP chimera were co-labeled with BODIPY-ceramide and imaged using 3D-SIM. See higher magnification image at right. (B) PfEMP1B-GFP transfectants (endogenous GFP fluorescence; green) were permeabilized with EqtII and antibody-accessible epitopes were labeled with anti-GFP (magenta) and anti-REX1 (red). (C) PfEMP1B-GFP transfectants were fixed and resin-embedded and sections were labeled with anti-GFP and protein-A-gold (6 nm left panels, 10 nm right panels). Labeling is observed at bulges in the PV (arrows). (D) Smears of PfEMP1B-GFP transfectant-infected RBCs were fixed with acetone: methanol and stained with antibodies recognizing GFP and Exp1 (D) or Hsp101 (E). Scale bars = 2 μm (A, first three columns), 1 μm (A, last column), 2 μm (B), as marked (C) and 2 μm (D, E).McMillan PJ, Millet C, Batinovic S, Maiorca M, Hanssen E, Kenny S, Muhle RA, Melcher M, Fidock DA, Smith JD, Dixon MW, Tilley L. Spatial and temporal mapping of the PfEMP1 export pathway in Plasmodium falciparum. Cell Microbiol. 15(8):1401-18
See original on MMP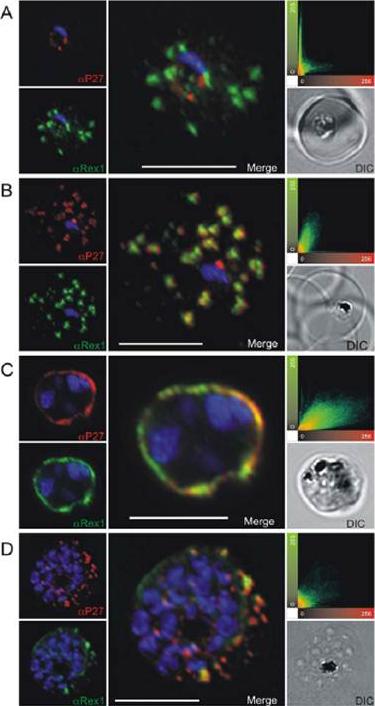
Co-localization of Tex1 with Rex1. P27-specific polyclonal mouse sera (in red) was used to detect TEX1. Rex1 polyclonal rabbit sera (in green). (A) Ring stage parasites; (B) trophozoite stages; (C) schizont stages. Scatter plots show the degree of co-localization of the Tex1 with Rex1 signal. Nuclear DNA was stained with DAPI (blue), Transmission image (DIC), Scale bar: 5 mm. Based on the co-localization of Tex1 and Rex1, Tex1 is localized to the Maurer’s clefts.Kulangara C, Luedin S, Dietz O, Rusch S, Frank G, Mueller D, Moser M, Kajava AV, Corradin G, Beck HP, Felger I. Cell biological characterization of the malaria vaccine candidate trophozoite exported protein 1. PLoS One. 2012;7(10):e46112.
See original on MMP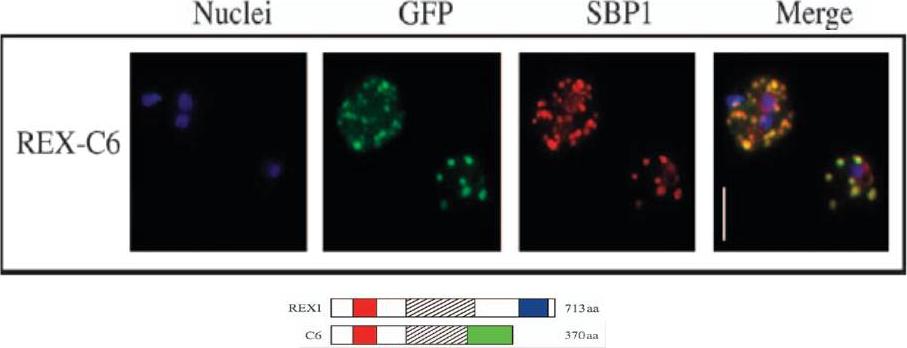
Transgenic parasites were fixed using acetone and detected with anti-GFP and anti-SBP1 antibodies. The panel shows REX-C6 colocalizations, GFP fluorescence of the chimeric protein in green and SBP1 fluorescence in red. The merge panel on the right shows complete colocalization of the two cleft proteins, confirming the presence of REX–GFP at the Maurer’s clefts.Dixon MW, Hawthorne PL, Spielmann T, Anderson KL, Trenholme KR, Gardiner DL. Targeting of the ring exported protein 1 to the Maurer's clefts is mediated by a two-phase process. Traffic. 2008 9:1316-26.
See original on MMP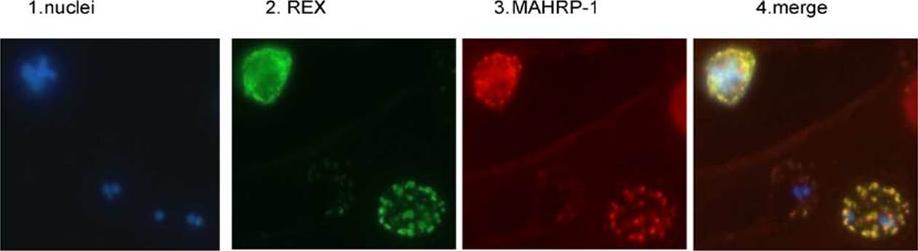
REX is located in the Maurer’s clefts. Panel 1 depicts nuclear staining (blue) of a field of 3D7 parasites. Panel 2 depicts the same field stained with REX antiserum (green). Panel 3 is the same field stained with MAHRP-1 antiserum (red). Panel 4 is a merge of the first three panels.Hawthorne PL, Trenholme KR, Skinner-Adams TS, Spielmann T, Fischer K, Dixon MW, Ortega MR, Anderson KL, Kemp DJ, Gardiner DL. A novel Plasmodium falciparum ring stage protein, REX, is located in Maurer's clefts. Mol Biochem Parasitol. 2004 136:181-9. Copyright Elsevier 2009. PMID:
See original on MMP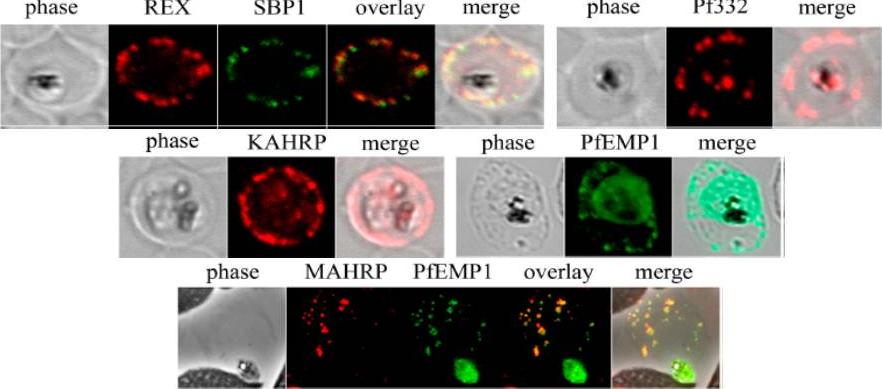
Analysis of expression and localization of Maurer’s cleft–associated proteins in IRBCs. Confocal immunofluorescence analysis of resident (REX-PFI1735c, SBP1-PFE0065w, and Pf332-PF11_0507) or transiently associated (KAHRP-PFB0100c and PfEMP1) Maurer’s cleft proteins. Colocalization of the resident Maurer’s cleft marker MAHRP-MAL13P1.413 with PfEMP1 in streptolysin O–pretreated IRBCs demonstrates that PfEMP1 is associated with Maurer’s clefts.Cooke BM, Buckingham DW, Glenister FK, Fernandez KM, Bannister LH, Marti M, Mohandas N, Coppel RL. A Maurer's cleft-associated protein is essential for expression of the major malaria virulence antigen on the surface of infected red blood cells. J Cell Biol. 2006 172:899-908.
See original on MMP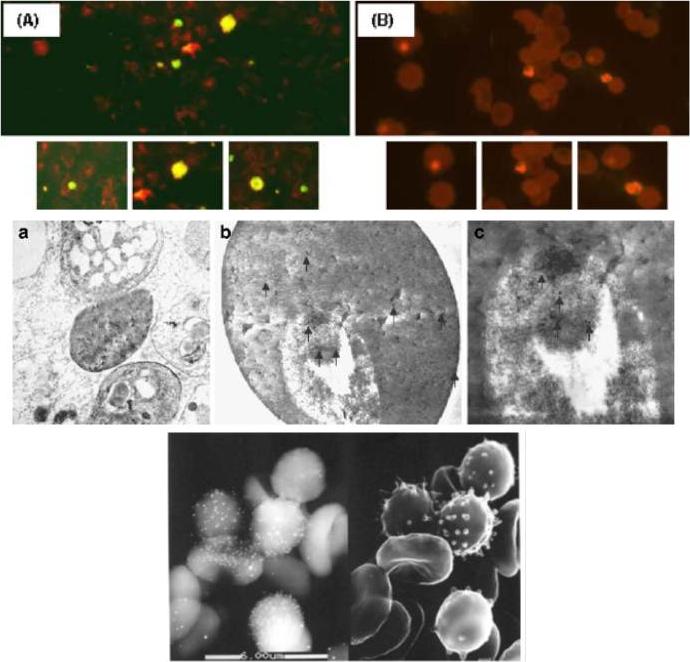
Localization of the anti-Pf62 reactivity on asexual parasite stages by indirect immuno-fluorescence. Smears of parasite-infected red blood cells, strain Dd2, were reacted with rabbit anti-Pf62 polyclonal antisera. a Incubation with 1:50 dilution of antisera raised against the Pf62 protein. b Incubation with preimmune rabbit serum at the same dilution, derived from the same rabbit used to derive Pf62 antibodies prior to the first immunization. Ultrastructural localization of Pf62 protein in different stages. Transmission electron microscopy immunogold labeling (10 nm gold particles, arrows) in ultrathin sections. a General overview of parasite-infected RBCs (×6,300, original magnification). b The Pf62 protein is localized in the trophozoite and in the erythrocyte, both in the cell interior and surface. c The same trophozoite at higher magnification. Immunoscanning electron micrograph. Left The backscattered electron detector identifies the bright signals from the gold particles that localize the Pf62 protein. Right The same field using the secondary electron detector that shows the ultrastructure of the erythrocytesMoyano EM, González LM, Montero E, Cuevas L, Perez-Pastrana E, Santa-Maria Y, Benito A. Initial characterization of Pf62, a novel protein of Plasmodium falciparum identified by immunoscreening. Parasitol Res. 2009 104:1389-97.
See original on MMP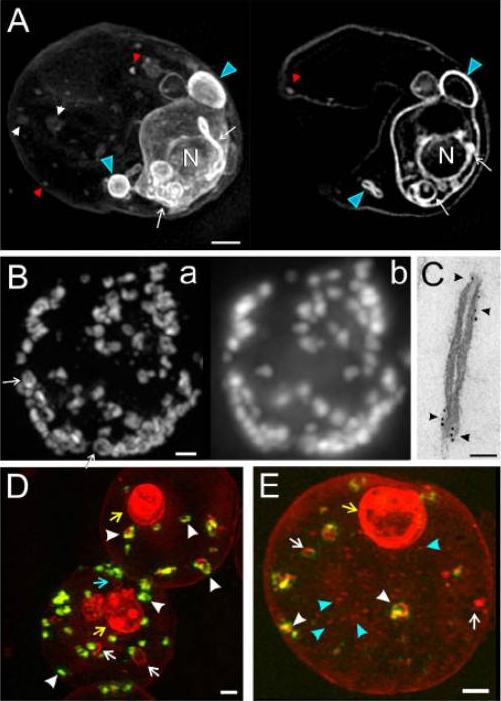
Three-dimensional structured illumination microscopy and immuno-electron microscopy (EM) of Plasmodium falciparum-infected red blood cells (RBCs). (A) 3D7 -infected RBCs (trophozoite stage) were labelled with the membrane probe, BODIPY-ceramide, and examined without fixation. Three Z-stacks (0.125 mm section spacing) were recorded. A projection image and a representative virtual section are presented. Features of the parasite’s endomembrane system (white arrows) and parasitophorous vacuole (PV) membrane/ tubulovesicular network (TVN) (blue arrowheads) are strongly labelled while the membranous features in the RBC cytoplasm (white/red arrowheads) are less strongly labelled. (B and C) RBCs infected with trophozoite stage 3D7 Ring Exported Protein-1-GFP (3D7_REX1-GFP) transfectants were permeabilized with Equinatoxin II, and labelled with anti-GFP antiserum followed by (B) AlexaRed-anti-IgG or (C) 6 nm gold protein A. (B) Z-stacks presented as the ‘‘raw” projection (b) or used to compute a structured illumination microscopy (SIM) image (a). A restricted pattern is revealed in some of the Maurer’s clefts (arrows). (C) Samples were prepared for EM. (D and E) RBCs infected with early trophozoite stage 3D7_REX1-GFP transfectants were labelled with the membrane probe, BODIPY-ceramide, and fixed with paraformaldehyde. REX1-GFP is concentrated in punctuate (green fluorescence) in the RBC cytoplasm; these structures represent Maurer’s clefts. The BODIPY-ceramide (red fluorescence) is concentrated in the parasite (yellow arrows) and in extensions of the parasite into the RBC cytoplasm (blue arrow) as well as in separate structures in the RBC cytoplasm. Some of the membrane features in the RBC cytoplasm contain REX1-GFP (white arrowheads) while others do not (white arrows). Small vesicle-like structures are observed (E, blue arrowheads). Scale bars: (A, B, D, and E) 1 mm; (C) 50 nm.Hanssen E, Carlton P, Deed S, Klonis N, Sedat J, Derisi J, Tilley L. Whole cell imaging reveals novel modular features of the exomembrane system of the malaria parasite, Plasmodium falciparum. Int J Parasitol. 2010 40:123-34.Copyright Elsevier 2010.
See original on MMP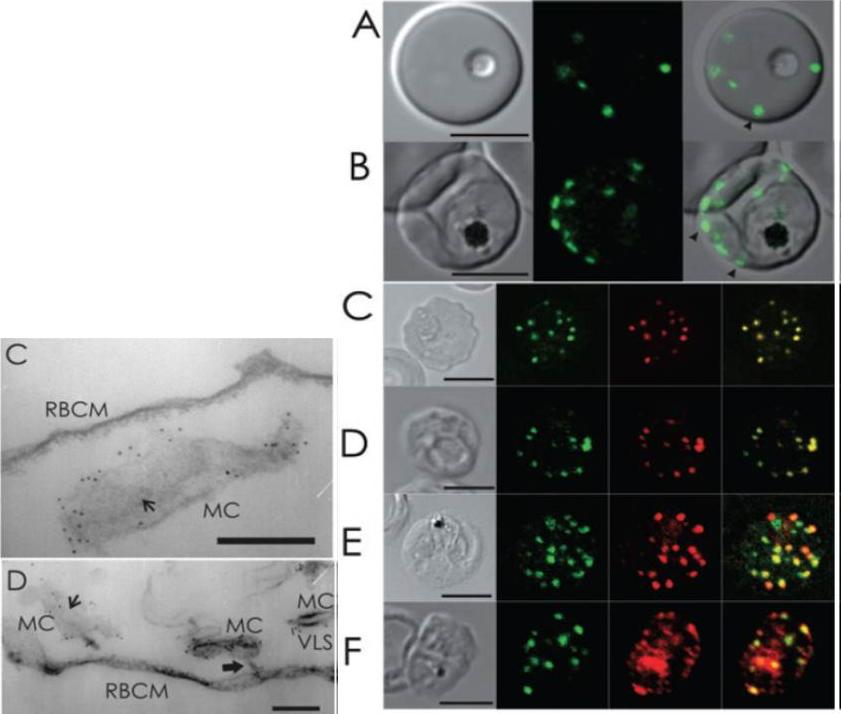
Live cell and immunofluorescence microscopy of 3D7_REX1–GFP P. falciparum transfectants. The first image in each panel is a differential interference contrast image. A and B. RBCs infected with live ring (A) and late trophozoite (B) stage parasites show puncta of fluorescence in the RBC cytoplasm, which are likely to represent Maurer’s clefts (arrowheads). C–F. Trophozoite stage-infected 3D7_REX1–GFP transfectant infected RBCs were labelled with mouse (C, E and F) or rabbit (D) anti-GFP antiserum (green) and rabbit (C, E and F) or mouse (D) antiserum recognizing REX1 (C), MAHRP-1-MAL13P1.413 (D), SBP1-PFE0065w (E) and PfEMP1 (F) (red). Overlap of the signals confirms the Maurer’s cleft locations of these proteins. PfEMP1 is also partly located at the RBC membrane and within the parasite. The intensities of the images were adjusted to optimize the fluorescence signals. Bars are 5 mm.Left: Immuno-EM of thin sections through Maurer’s clefts. Sections through EqtII-permeabilized RBCs infected with 3D7_REX1–GFP transfectants showing Maurer’s clefts (MC). Connections to the the RBC membrane (D, RBCM) are indicated with thick arrows. Flattened regions of the Maurer’s clefts observed in transverse section (C and D) are indicated with smaller arrows. The infected RBCs were labelled with anti-GFP antiserum followed by protein A-gold (6 nm conjugate). Bars are 200 nm.Hanssen E, Hawthorne P, Dixon MW, Trenholme KR, McMillan PJ, Spielmann T, Gardiner DL, Tilley L. Targeted mutagenesis of the ring-exported protein-1 of Plasmodium falciparum disrupts the architecture of Maurer's cleft organelles. Mol Microbiol. 2008 69:938-53. PMID:
See original on MMP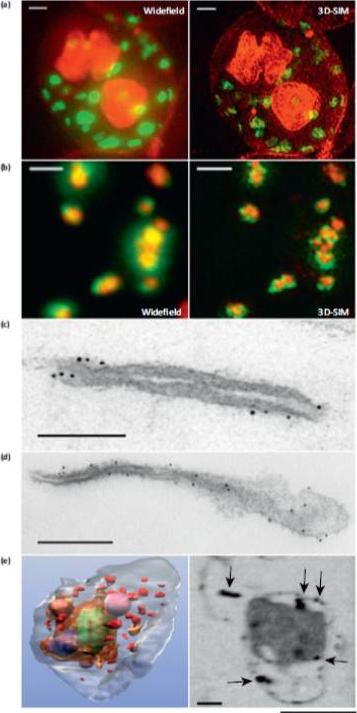
Microscopy methods for localizing Plasmodium falciparum Maurer’s cleft-associated proteins. (a) Plasmodium falciparum transfectants expressing REX1-GFP (green) costained with a membrane stain (red) and imaged using conventional widefield fluorescence microscopy and 3D structured illumination microscopy (3D-SIM). Scale bars = 1 mm. (b) Immunofluores-cence microscopy of P. falciparum REX1-GFP transfectants labeled with anti-GFP (green) and anti-MAHRP1 (red) antibodies. The sample was imaged using conventional widefield fluorescence microscopy and 3D-SIM. Scale bars = 1 mm. (c, d) Immuno-electron microscopy. Red blood cells (RBCs) infected with trophozoite stage REX1-GFP transfectants [(c) scale bar = 100 nm)] or MAHRP1-GFP trans-fectants [(d) (scale bar = 200 nm)] were permeabilized with Equinatoxin II and labeled with anti-GFP antiserum followed by 6 nm gold-labeled protein A. (e) Left hand panel: rendered cryo-X-ray tomogram of an Equinatoxin II permeabilized trophozoite-infected RBC. The surface of the trophozoite is rendered in brown, the nucleus in green, the digestive vacuole in purple. Hemoglobin containing vesicles in the RBC cytoplasm are rendered in magenta and Maurer’s clefts in red. Right hand panel: cryo-X-ray micrograph of an infected RBC. MAHRP1-GFP transfectant-infected RBCs were permeabilized and labeled with mouse anti-GFP antiserum followed by 6 nm gold-labeled protein A. The size of the gold particles was increased by silver enhancement. Dark deposits in the RBC cytoplasm (arrowheads) reveal labeling of the Maurer’s clefts. Scale bars = 500 nm.Woodcroft BJ, McMillan PJ, Dekiwadia C, Tilley L, Ralph SA. Determination of protein subcellular localization in apicomplexan parasites. Trends Parasitol. 2012 Dec;28(12):546-54.
See original on MMP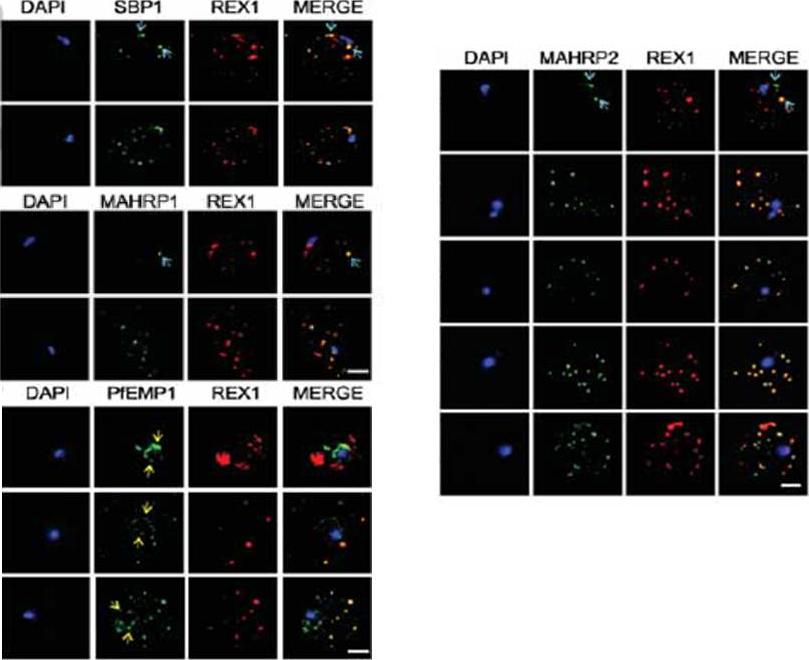
Immunofluorescence microscopy showing staggered delivery of different exomembrane components. (A-D) Infected RBCs were synchronized to a 1 h window, samples were collected at 2 h intervals and smears were fixed with acetone and stained with antibodies recognizing REX1, MAHRP1, SBP1, MAHRP2 and PfEMP1 (ATS) and co-stained with DAPI. Images are presented at time points before and after delivery to the RBC cytoplasm. Scale bars = 3 μm. McMillan PJ, Millet C, Batinovic S, Maiorca M, Hanssen E, Kenny S, Muhle RA, Melcher M, Fidock DA, Smith JD, Dixon MW, Tilley L. Spatial and temporal mapping of the PfEMP1 export pathway in Plasmodium falciparum. Cell Microbiol. 2013 15(8):1401-18
See original on MMP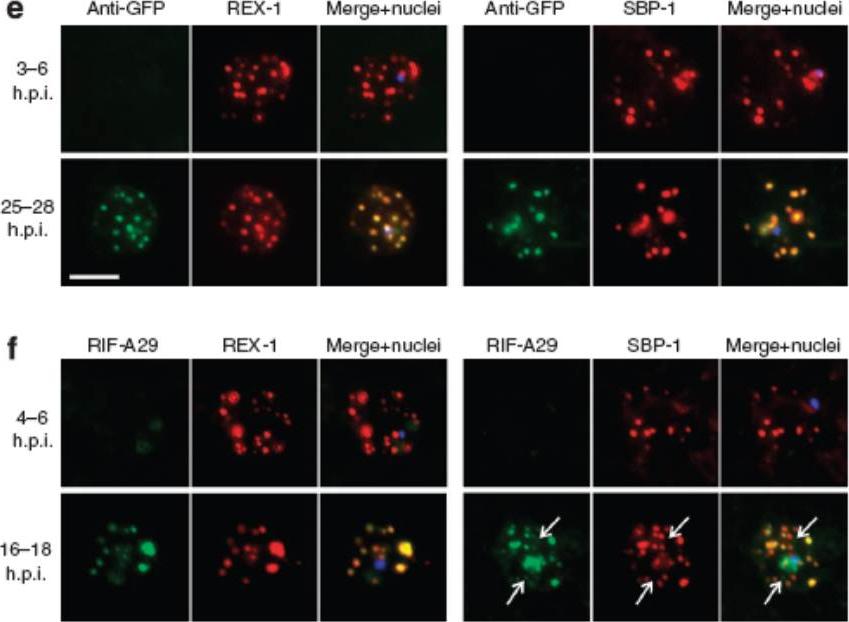
Export of membrane proteins is independent of cleft formation. IFA showing that in REX2-GFPcrt-expressing parasites (e) and 3D7 (f), early clefts contain REX1 and but not REX2-GFPcrt (e, upper panels) or RIFIN A29 (RIF-A29, f, upper panels). On progress of the culture to the trophozoite stage, the clefts containing REX1 and now also contain REX2-GFPcrt (e, lower panel) and RIFIN A29 (f, lower panel). Dots positive for RIFIN A29 only, found in ~50% of cells, are indicated by white arrows. Scale bars, 5 μm; h.p.i., hours post invasion. the clefts in early stages contained only the early markers (e,f). In contrast, later stages were positive for all four antigens (e,f). Crucially, in later stages all antigens occupied the same clefts (e,f), demonstrating that the clefts initially containing REX1 and SBP1 accumulated the other proteins later on. This establishes that export occurred without cleft formation.Grüring C, Heiber A, Kruse F, Ungefehr J, Gilberger TW, Spielmann T. Development and host cell modifications of Plasmodium falciparum blood stages in four dimensions. Nat Commun. 2011 2:165.
See original on MMP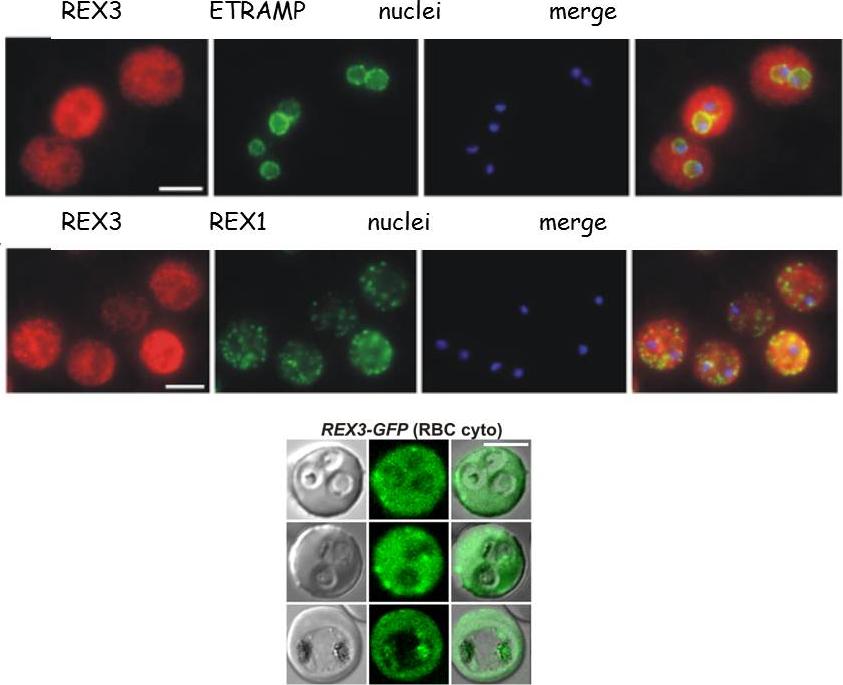
Upper row: Location of REX3 in IRBCs fixed with 4% formaldehyde/0.005% glutaraldehyde. IFAs with 3D7 parasites expressing a myc-tagged ETRAMP4 showed that REX3 (red; Texas Red) occurs as a uniform stain outside of the PVM (ETRAMP4myc in the PVM detected by a rabbit anti-myc antibody; green, Cy2). Second row: REX3 (red; Texas Red) did not colocalize with the Maurer’s cleft protein REX1 (green; Cy2) in 3D7 parasites. Bars, 5 mm.Lower panel: Confocal fluorescence microscopy images of transfected 3D7 P. falciparum-infected RBCs. REX3 is expressed in its early ring stages (top row) and is directed to the bulk compartment of the host cell cytoplasm. Trophozoites and schizonts (middle and bottom rows) display a uniform fluorescence pattern with some puncta. Tilley L, McFadden G, Cowman A, Klonis N. Illuminating Plasmodium falciparum-infected red blood cells. Trends Parasitol. 2007 23:268-77.
See original on MMP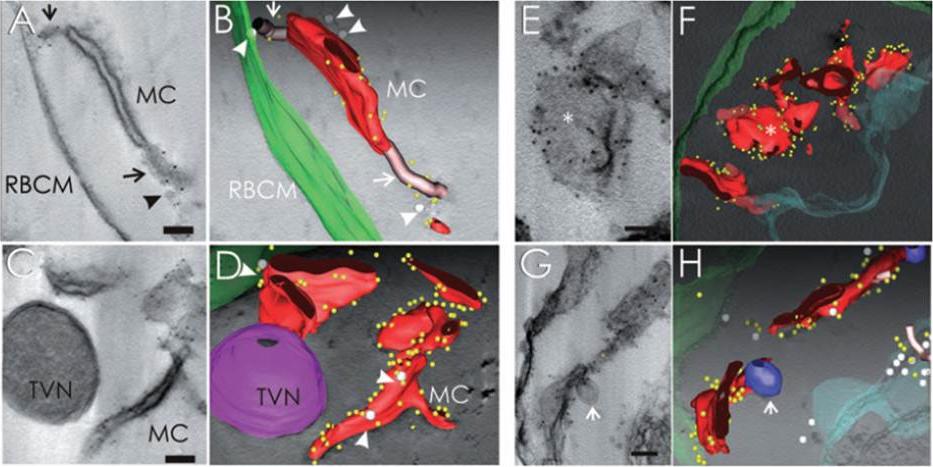
Immunoelectron tomography of permeabilized 3D7- and 3D7_REX1–GFP transfectant-infected RBCs showing the subcellular location of REX1. A–D. EqtII-permeabilized 3D7-infected RBC labelled with anti-REX1 antiserum followed by protein A-gold (6 nm conjugate). A. Virtual 20 nm section through a tomogram showing a Maurer’s cleft body flanked by two tether-like structures. C. A whole tomogram was projected and averaged with ImageJ using minimum intensity. The segmentation was performed semi-automatically using the IMOD-auto routine and the contours were assigned manually (B and D, red, Maurer’s cleft; green, RBC membrane; yellow, gold particles). The gold particles are concentrated at the edges of the Maurer’s cleft bodies. Vesicle-like structures (grey spheres, arrowheads) are observed associated with the Maurer’s clefts and RBC membrane (B and D). Regions with extended stalk-like profiles, which may be parts of tethers linking to the RBC membrane, are depicted as pink tubes (A and B, arrows). These regions are also labelled with gold particles. C and D. A double membrane-bound haemoglobin-containing structure which may be part of the TVN is indicated in magenta. This structure is not labelled with anti-REX1. A REX1-labelled Maurer’s cleft appears to be connected to the TVN. E–H. EqtII-permeabilized 3D7_REX1–GFP transfectants were labelled with anti-GFP and prepared for semi-thin sectioning (300 nm). E and G. Re-projections of the whole thickness of tomograms in planes parallel (E) and perpendicular (G) to the transverse surfaces of Maurer’s clefts. The whole tomograms were used to generate the models and drive the rendering process (F and H: red, Maurer’s cleft; green, RBC membrane; aqua, PV membrane; blue, double membrane-bound compartment/TVN; grey spheres, vesicle-like structures; pink tubes, tether-like structures; yellow, gold particles). The asterisk marks a flatter region of a Maurer’s cleft body that is depleted of gold particles. Bars are 100 nm.Hanssen E, Hawthorne P, Dixon MW, Trenholme KR, McMillan PJ, Spielmann T, Gardiner DL, Tilley L. Targeted mutagenesis of the ring-exported protein-1 of Plasmodium falciparum disrupts the architecture of Maurer's cleft organelles. Mol Microbiol. 2008 69:938-53.
See original on MMP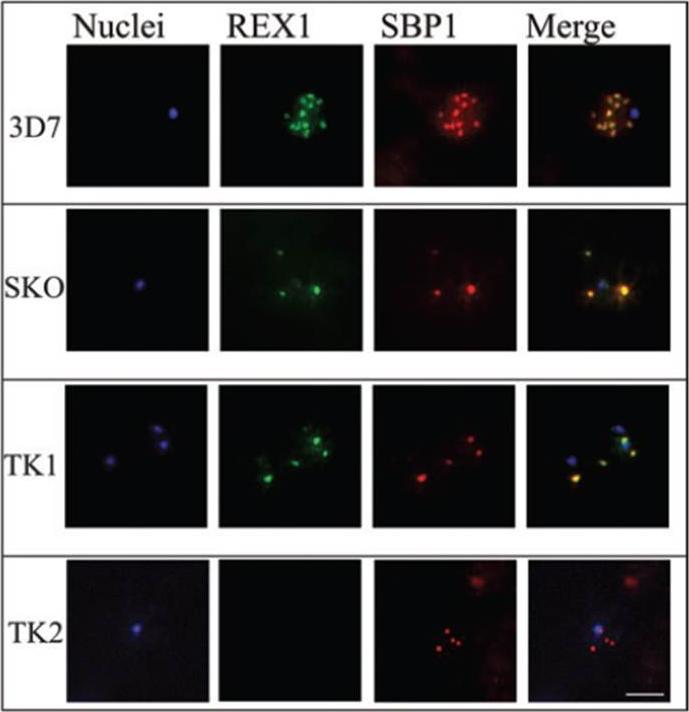
Immunofluorescence microscopy of paraformaldehyde-fixed RBCs infected with 3D7, SKO, TK1 or TK2 parasites probed with anti-REX166-169 antiserum and anti-SBP1. The nuclei were visualized with Hoechst staining. Bar is 5 mm. Maurer’s clefts are observed as punctate structures in the RBC cytoplasm in 3D7 parasites labelled with the known Maurer’s cleft protein, SBP1 or with the antiserum against REX166-169 (top row). By contrast, RBCs infected with D10 parasites are not labelled with either REX1 antiserum (data not shown). In the SKO (second row), the antiserum against REX166-169 also recognized the truncated REX1 protein at the Maurer’s clefts. However, an antiserum against the C-terminal region does not recognize the truncated REX1 protein. The Maurer’s clefts location was confirmed by dual labelling with anti-SBP1 antiserum (second row).Hanssen E, Hawthorne P, Dixon MW, Trenholme KR, McMillan PJ, Spielmann T, Gardiner DL, Tilley L. Targeted mutagenesis of the ring-exported protein-1 of Plasmodium falciparum disrupts the architecture of Maurer's cleft organelles. Mol Microbiol. 2008 69(4):938-53
See original on MMP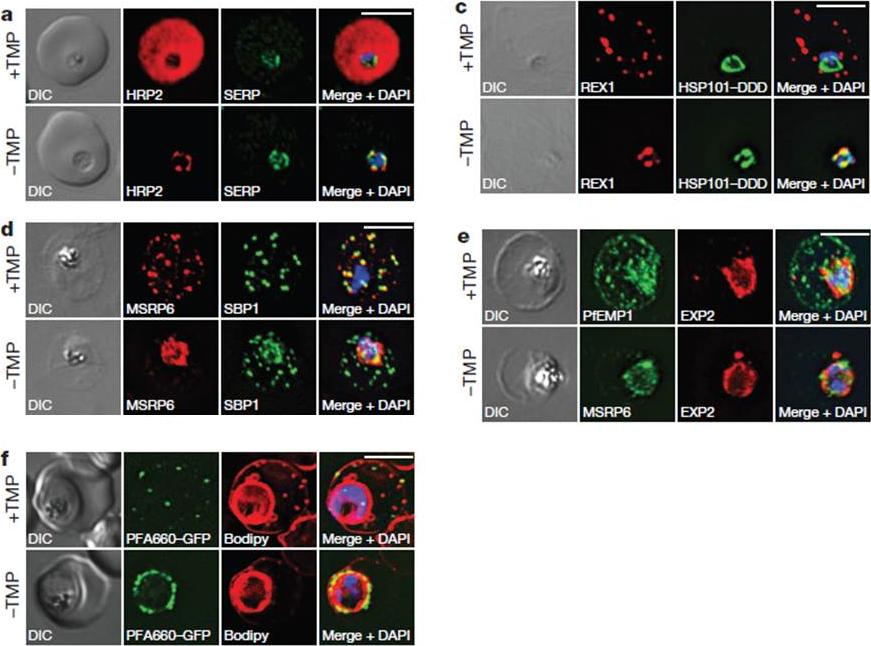
HSP101 is required for export of PEXEL and PNEP proteins. a, c, Immunofluorescence assay (IFA) of ring-stage 13F10 parasites with orwithout TMP (DDD-stabilizing small molecule trimethoprim). TMP was removed in late schizont stage and parasites were allowed to reinvade and grow 18–24 h before fixation with paraformaldehyde (a) or acetone (c). a, IFA of the exported PEXEL-containing protein HRP2. c, IFAs of the PNEP REX1, which colocalizes with HSP101DDD at the PVM in the absence of TMP. d, e, IFA of trophozoite-stage 13F10 parasites with or withoutTMP. TMP was removed in late ring stage and parasites were allowed to develop 12–24 h before fixation with acetone. f, Live fluorescence imaging of 13F10 parasites expressing a PFA660–GFP fusion and labelled with Bodipy TR Ceramide to demarcate the PVM (other membranes are also labelled). TMP treatment as in d, e. All scale bars, 5 mm. Images in c–f are representative of two independent experiments.Beck JR, Muralidharan V, Oksman A, Goldberg DE. PTEX component HSP101 mediates export of diverse malaria effectors into host erythrocytes. Nature. 2014 Jul 16. [Epub ahead of print]
See original on MMP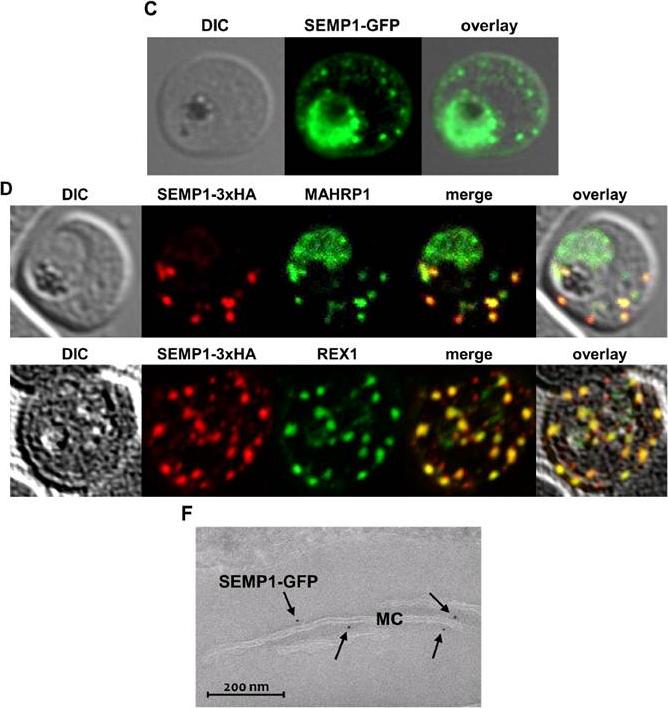
C: Live cell imaging of 3D7 parasites expressing SEMP1 with C-terminal tagged GFP. D: Immunofluorescence assays of MeOHfixed RBCs infected with 3D7 expressing SEMP1 with a C-terminal 3xHA tag, co-labelled with rat a-HA and either rabbit a-MAHRP1 or rabbit a-REX1 antibodies. D: Scatter plot of co-localization of SEMP1 and REX1 in SEMP1-3xHA parasites. E: Electron microscopy (EM) of RBCs infected with 3D7 expressing SEMP1 with a C-terminal GFP tag labelled with rabbit a-GFP antibodies and decorated with 5 nm gold conjugated Protein A. GFP-tagged protein was exported (c). SEMP1-3xHA co-localized with MAHRP1 and REX1 to the MCs. Electron microscopy using antibodies against GFP confirmed the localization of SEMP1-GFP at the MCs (F). Dietz O, Rusch S, Brand F, Mundwiler-Pachlatko E, Gaida A, Voss T, Beck HP. Characterization of the Small Exported Plasmodium falciparum Membrane Protein SEMP1. PLoS One. 2014 9(7):e103272.
See original on MMP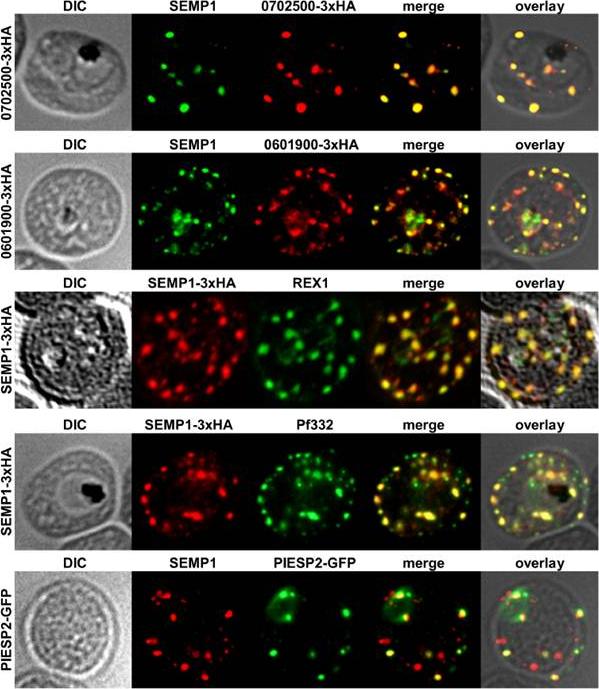
Localization of potential SEMP1 interacting proteins. Immunofluorescence assays of MeOH-fixed RBCs infected with 3D7 expressing EMP1/PF3D7_0702500 (PF07_0008)/PF3D7_0601900 (PFF0090w) with a C-terminal 3xHA tag and 3D7 expressing PIESP2 with a C-terminal GFP-tag, co-labelled with mouse a-SEMP1 and rat a-HA (PF3D7_0702500-3xHA & PF3D7_0601900-3xHA) /a-GFP (PIESP2-GFP). For co-labelling of SEMP1-3xHA with REX1 and Pf332, rat a-HA and rabbit a-REX1 / mouse a-Pf332 antibodies were used. All potential interaction partners co-localized at least partially with SEMP1, but in particular with Pf332 or PIESP2 antibodies only a fraction of SEMP1 positive MCs were labelled with either antibody suggesting that distinct subpopulations of MCs exist. Dietz O, Rusch S, Brand F, Mundwiler-Pachlatko E, Gaida A, Voss T, Beck HP. Characterization of the Small Exported Plasmodium falciparum Membrane Protein SEMP1. PLoS One. 2014 9(7):e103272.
See original on MMP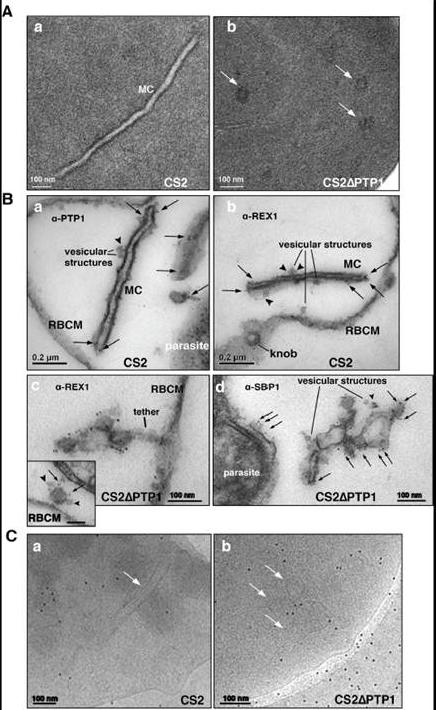
Comparison of transmission electron microscopical studies on CS2 versus CS2ΔPTP1 cell line.(A) Conventional chemical Fixation: a) CS2 parental line. MC (MC) display electron dense membranes and an electron lucent lumen; b) CS2ΔPTP1 cell line shows globular structures and no long lamellar MC structures (arrows). (B) Equinatoxin II treated cells with pre-embedding labelling: a) α-PfPTP1 antibodies or b) α-REX1 antibodies label the slender MC membranes (arrows) and vesicles (arrowheads) in the CS2 parasite line; c) α-REX1 or d) α-SBP1 antibodies label globular structures (arrows) and vesicular structures (arrowheads) in the CS2ΔPTP1 cell line. RBCM – red blood cell membrane. (C) The frozen hydrated samples show MC (arrow) in which the lumen looks similar to the RBC cytoplasm as described earlier. The CS2ΔPTP1 cell line shows aggregated globular structures (arrows). In CS2 typical single lamellae for MC were observed with an electron dense coat and translucent lumen (Aa). In contrast, CS2ΔPTP1 displayed electron dense areas randomly distributed through the RBC cytoplasm (Ab). The α-PfPTP1 antibodies line the MC lamella similar to REX1 localisation (B). Vesicular structures were observed that appeared to bud off MC and these had a dense lumen and less distinct membranes. These may be vesicles involved in trafficking between PVM and MC and/or MC to the RBC membrane (i.e. J-Dots). These structures were decorated withPfPTP1 and REX1 antibodies (Ba,b). In CS2ΔPTP1 the architecture of MC was disrupted showing large aggregated globular structures (Bc,d). SBP1 and REX1 were located on these structures. Single lamellae of MC were observed in CS2 (Ca) whereas globular structures were observed for CS2ΔPTP1 (Cb).Rug M, Cyrklaff M, Mikkonen A, Lemgruber L, Kuelzer S, Sanchez CP, Thompson J, Hanssen E, O'Neill M, Langer C, Lanzer M, Frischknecht F, Maier AG, Cowman AF. Export of virulence proteins by malaria-infected erythrocytes involves remodelling of host actin cytoskeleton. Blood. 2014 Aug 19 [Epub ahead of print]
See original on MMP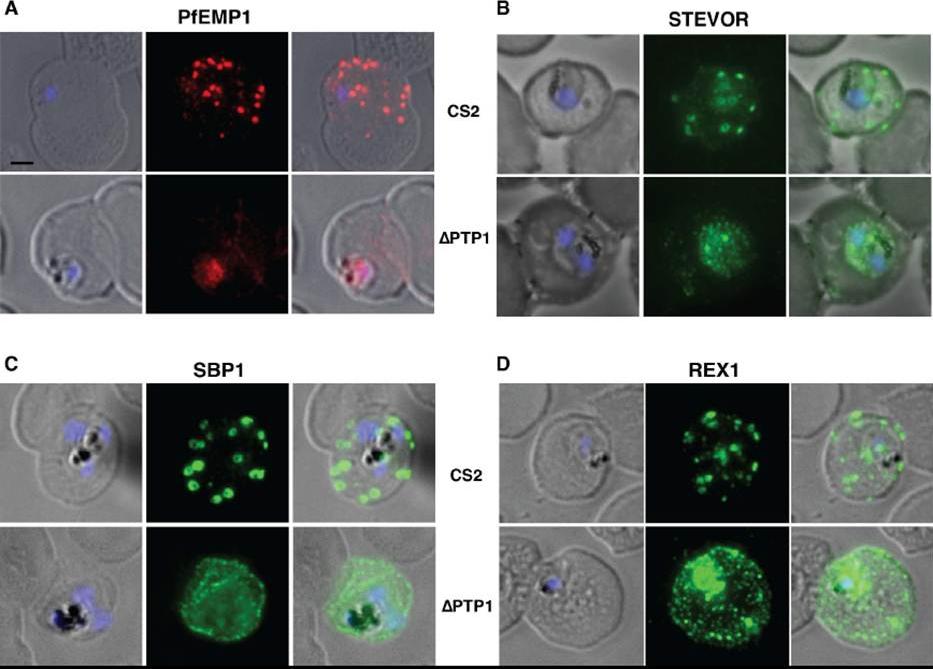
Immunofluorescence assay on CS2 (upper panels) versus CS2ΔPTP1 parasites (lower panels). Immunofluorescence assay probed with a-PfEMP1 antibody (A), a-STEVOR antibody (B), a -SBP 1 antibody (C) and a -REX 1 antibody (D). Scale bar: 1 μm; same size of ROI in each image. In CS2ΔPTP1 the architecture of MC was disrupted showing large aggregated globular structures. SBP1 and REX1 were located on these structures reflecting the results obtained by IFA.Rug M, Cyrklaff M, Mikkonen A, Lemgruber L, Kuelzer S, Sanchez CP, Thompson J, Hanssen E, O'Neill M, Langer C, Lanzer M, Frischknecht F, Maier AG, Cowman AF. Export of virulence proteins by malaria-infected erythrocytes involves remodelling of host actin cytoskeleton. Blood. 2014 Aug 19 [Epub ahead of print]
See original on MMP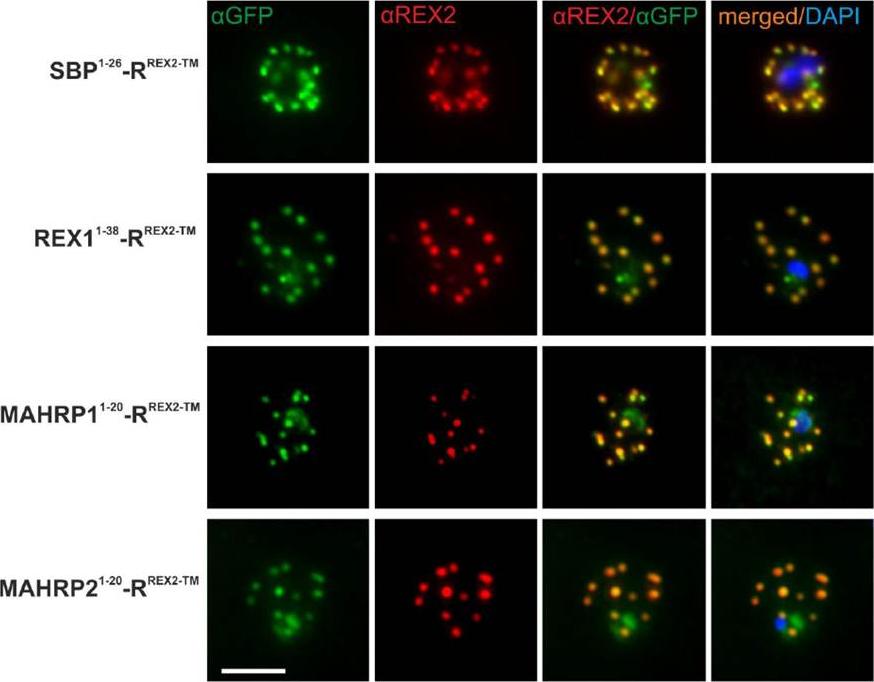
Immunofluorescence Assays Confirm Maurer’s Clefts Localization of PNEP N Termini mTRAP Reporter Fusions. Acetone fixed parasites expressing the reporter indicated on the left were probed with anti-GFP and a serum reacting with the C-terminus of REX2. Under these fixing conditions soluble proteins in the host cell are lost, leading to absence of the soluble pool of the reporter. DIC, differential interference contrast; DAPI, nuclei. Size bar: 5m.Grüring C, Heiber A, Kruse F, Ungefehr J, Gilberger TW, Spielmann T. Development and host cell modifications of Plasmodium falciparum blood stages in four dimensions. Nat Commun. 2011 2:165. PMID:
See original on MMP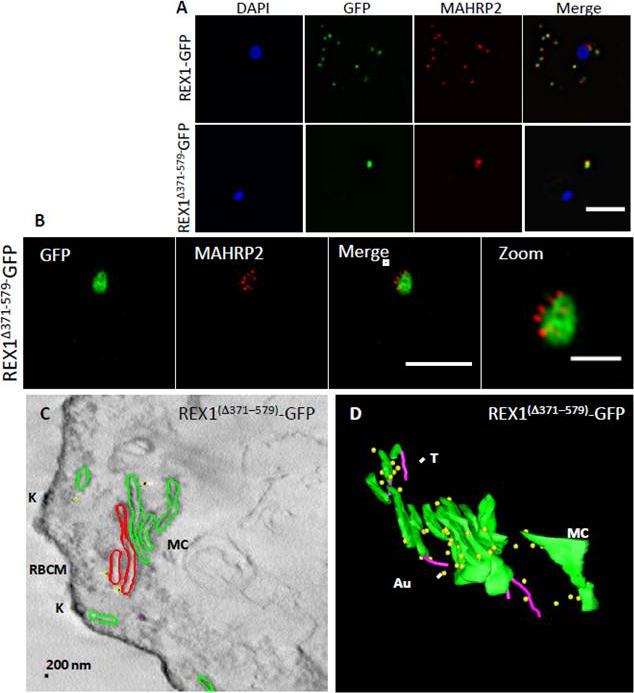
Analysis of the Maurer’s clefts ultrastructure and distribution of tethers ofREX1(Δ371–579)-GFP parasites. REX1-GFP and REX1(Δ371–579)-GFP transfectants were fixed with paraformaldehyde/ glutaraldehyde, permeabilized with Triton X-100, and probed with anti-GFP (green) and anti-MAHRP2 (red). Samples imaged using (A) widefield deconvolution microscopy or (B) 3D-SIM. Scale bars = 3 μm; zoom bar = 1 μm. C. STEM tomogram (600 nm section) of an EqII-permeabilized REX1(Δ371–579)-GFP-infected RBC showing the stacked Maurer’s clefts (MC) layers, and knobs (K) on the RBC membrane (RBCM). The lamella indicated in red share a membrane continuum. D. Rendered STEM tomogram of REX1(Δ371–579)-GFP-infected RBC labelled with anti-GFP antibodies and protein A gold showing Maurer’s clefts (MC, green), tethers (T, magenta) and gold particles (Au, yellow). In REX1-GFP parasites, immunofluorescence reveals MAHRP2 labelling (Fig. 6A) closely adjacent to the REX1-GFP labelled Maurer's clefts. For the REX1Δ371-579-GFP parasites, the MAHRP2 labelling partly overlaps with and partly sits outside the single puncta of GFP labelling. McHugh E, Batinovic S, Hanssen E, McMillan PJ, Kenny S, Griffin MD, Crawford S, Trenholme KR, Gardiner DL, Dixon MW, Tilley L. A repeat sequence domain of the ring-exported protein-1 of Plasmodium falciparum controls export machinery architecture and virulence protein trafficking. Mol Microbiol. 2015 Aug 24. [Epub ahead of print]
See original on MMP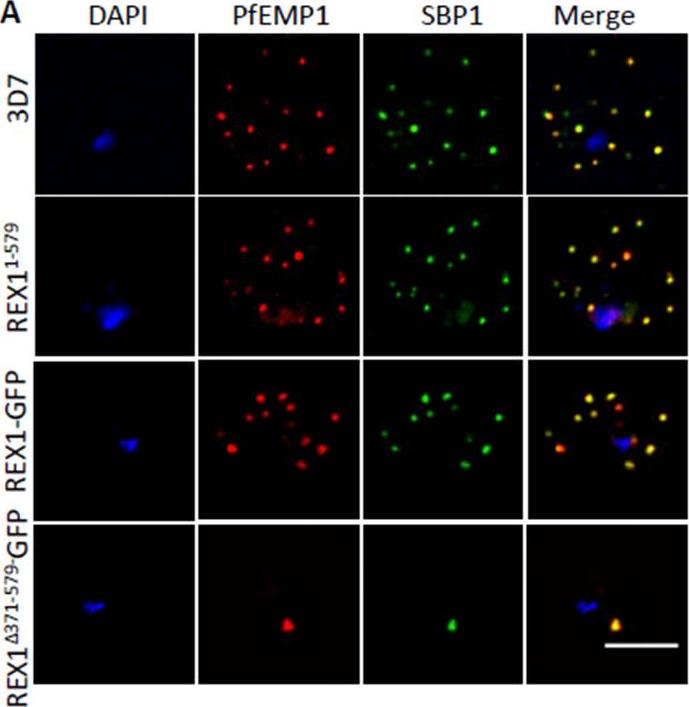
PfEMP1 surface-exposure and cytoadherence of REX1 transfectants. A. Immunofluorescence microscopy of acetone-fixed RBCs infected with 3D7 and REX1 transfectants. Maurer’s clefts were identified by immunolabeling with anti-SBP1 (green). Anti-PfEMP1 antibodies (red) revealed colocation of the virulence protein with Maurer’s clefts. The nuclei are stained with DAPI (blue). Scale bar = 3 μm. Accumulation of PfEMP1 at the Maurer’s clefts in the 3D7 wildtype parasites, was observed as confirmed by co-labelling with the Maurer’s cleft marker, SBP1. PfEMP1 is also delivered to the Maurer's clefts in the REX11-579, REX1-GFP and REX1Δ371-579-GFP mutant parasite lines. Thus despite the aberrant morphology there is no obvious defect in trafficking of PfEMP1 from the parasite to the stacked Maurer’s clefts.McHugh E, Batinovic S, Hanssen E, McMillan PJ, Kenny S, Griffin MD, Crawford S, Trenholme KR, Gardiner DL, Dixon MW, Tilley L. A repeat sequence domain of the ring-exported protein-1 of Plasmodium falciparum controls export machinery architecture and virulence protein trafficking. Mol Microbiol. 2015 Aug 24. [Epub ahead of print]
See original on MMP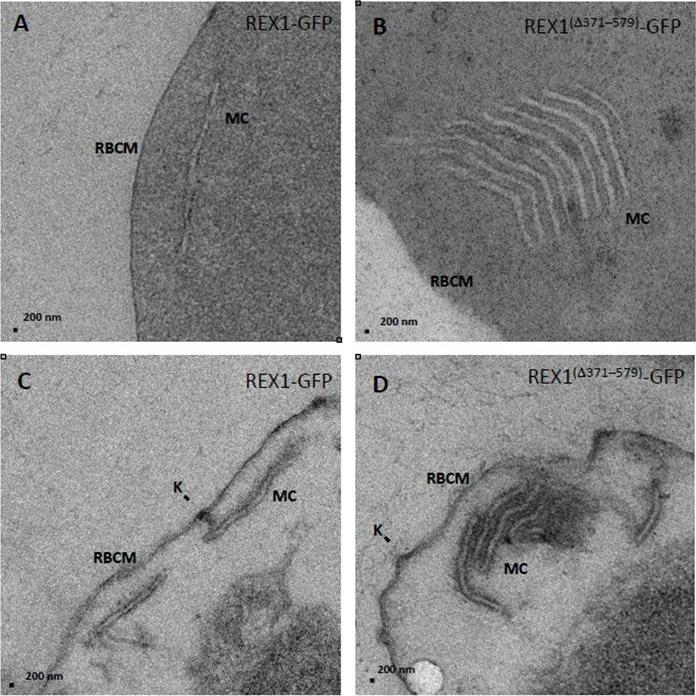
Transmission electron microscopy analysis of REX1-GFP and REX1(Δ371–579)-GFP Maurer’s clefts. A,B. TEM images of glutaraldehyde-fixed REX1-GFP and REX1(Δ371–579)-GFPinfected RBCs (100 nm sections) revealing unstacked and highly stacked Maurer’s clefts (MC). lamellae. C,D. TEM images of EqtII-permeabilized samples showing single Maurer’s cleft lamellae and stacked lamellae (100 nm sections). Knobs (K) are indicated. The REX1-GFP parasites displayed an unstacked Maurer's cleft phenotype (A,C) equivalent to that previously reported for the 3D7 strain Dramatically, the REX1Δ371-579-GFP parasites showed very large stacks of Maurer's clefts (B).McHugh E, Batinovic S, Hanssen E, McMillan PJ, Kenny S, Griffin MD, Crawford S, Trenholme KR, Gardiner DL, Dixon MW, Tilley L. A repeat sequence domain of the ring-exported protein-1 of Plasmodium falciparum controls export machinery architecture and virulence protein trafficking. Mol Microbiol. 2015 Aug 24. [Epub ahead of print]
See original on MMP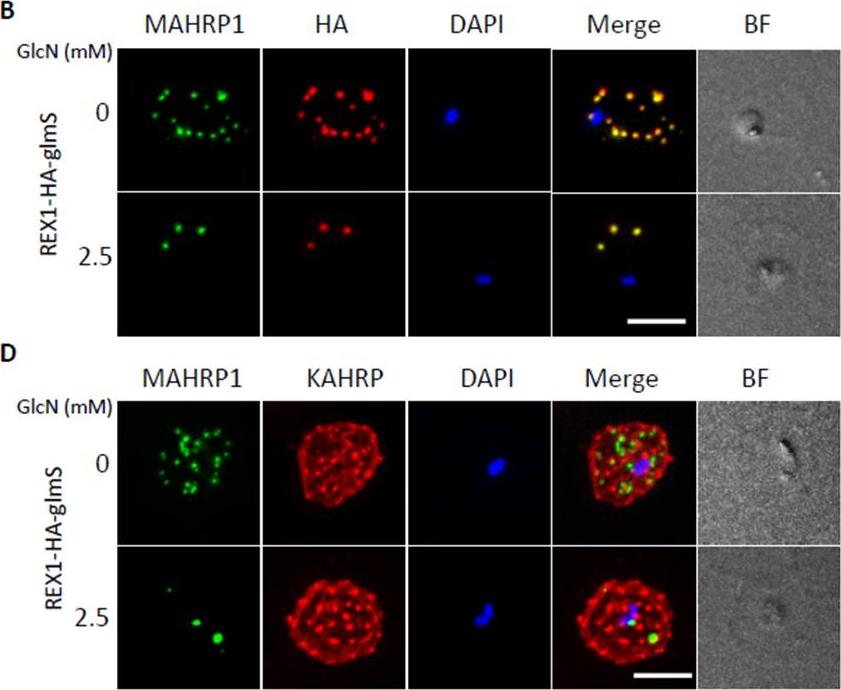
Inducible knockdown of REX1 using the glmS ribozyme system decreases the number of Maurer's cleft puncta. B,D. Immunofluorescencemicroscopy of paraformaldehyde-fixed infected RBCs probed with Maurer’s cleft marker anti-MAHRP1 (green) and anti-HA or anti-KAHRP (red). Nuclei are stained with DAPI (blue). Scale bar = 3 μm. C. Quantitation of numbers of Maurer’s clefts produced by REX1_KD transfectants in the presence and absence of 2.5 mM GlcN. The mean number of Maurer’s clefts produced per singly nucleated infected RBC was determined by counting SBP1-labelled puncta in at least 10 cells. Error bars = S.D. These puncta correspond to individual Maurer’s cleft cisternae that have a distributed organization within the RBC cytoplasmMcHugh E, Batinovic S, Hanssen E, McMillan PJ, Kenny S, Griffin MD, Crawford S, Trenholme KR, Gardiner DL, Dixon MW, Tilley L. A repeat sequence domain of the ring-exported protein-1 of Plasmodium falciparum controls export machineryarchitecture and virulence protein trafficking. Mol Microbiol. 2015 Aug 24. [Epub ahead of print]
See original on MMP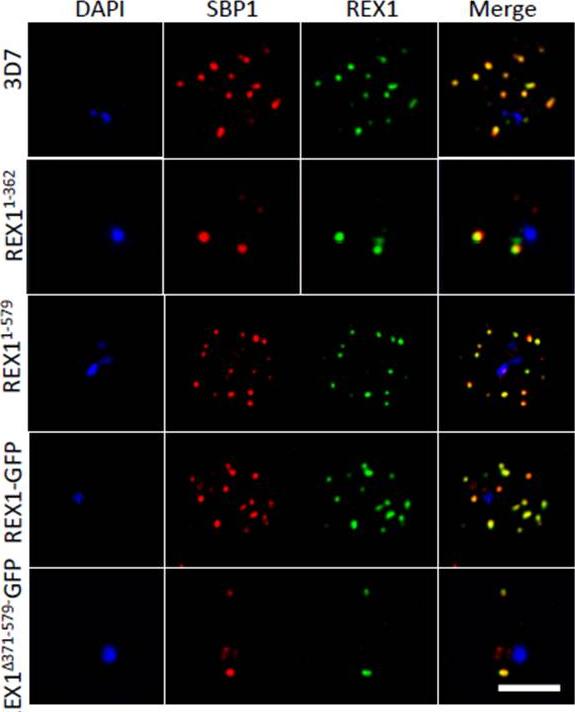
Deletion of the repeat region of REX1 decreases the number of Maurer's cleft puncta. A. Immunofluorescence microscopy of acetone-fixed infected RBCs probed with anti-SBP1 (red) and anti-REX1 (green). Nuclei are stained with DAPI (blue). Scale bar = 3 μm. Full-length REX1-GFP is correctly localized at the Maurer's clefts and that these transfectants exhibit an average number of puncta equivalent to that of the 3D7 parent. By contrast, the REX1Δ371-579-GFP parasites exhibited a striking Maurer’s cleft phenotype, with only 2 + 0.3 punctate structures observed in the infected RBC cytoplasm.McHugh E, Batinovic S, Hanssen E, McMillan PJ, Kenny S, Griffin MD, Crawford S, Trenholme KR, Gardiner DL, Dixon MW, Tilley L. A repeat sequence domain of the ring-exported protein-1 of Plasmodium falciparum controls export machinery architecture and virulence protein trafficking. Mol Microbiol. 2015 Aug 24. [Epub ahead of print]
See original on MMP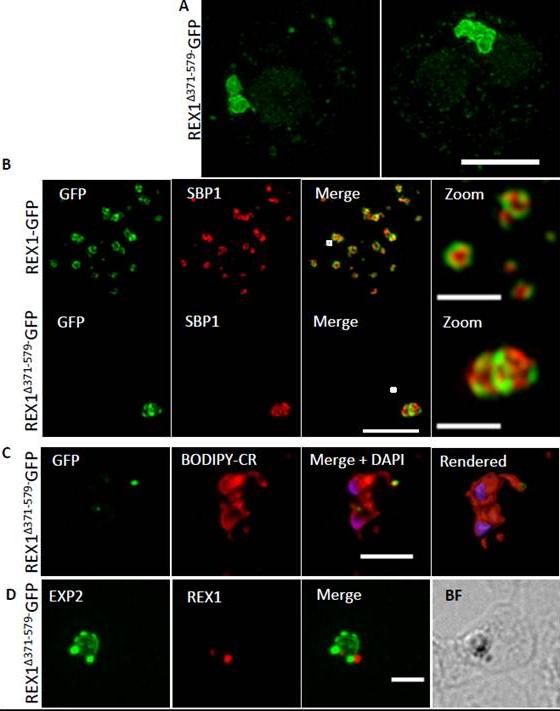
REX1(Δ371–579)-GFP parasites exhibit giant Maurer’s clefts. A. REX1(Δ371–579)-GFPinfected RBCs fixed with paraformaldehyde, permeabilized with EqtII and labelled with anti-GFP. B. REX1-GFP and REX1(Δ371–579)-GFP-infected RBCs were fixed with paraformaldehyde/glutaraldehyde and probed with anti-GFP (green) and anti-SBP1 (red). Samples were examined using 3D-SIM. 3D-SIM reveals giant Maurer's clefts with an apparently convoluted surfaceC. REX1(Δ371–579)-GFP-infected RBCs were labelled with BODIPY-ceramide, fixed with paraformaldehyde/ glutaraldehyde, labelled with DAPI and imaged using widefield deconvolution microscopy. The right hand panel shows rendering of the surface of the 3D structure using Imaris software. REX1Δ371-579-GFP appears to accumulate at discrete regions of the PVM as does EXP-2. D. REX1(Δ371–579)-GFP-infected RBCs were fixed with paraformaldehyde/ glutaraldehyde and probed with anti-EXP2 (green) and anti-REX1 (red). Samples were examined using deconvolution microscopy. Scale bars = 3 μm; zoom bar = 1 μm.McHugh E, Batinovic S, Hanssen E, McMillan PJ, Kenny S, Griffin MD, Crawford S, Trenholme KR, Gardiner DL, Dixon MW, Tilley L. A repeat sequence domain of the ring-exported protein-1 of Plasmodium falciparum controls export machinery architecture and virulence protein trafficking. Mol Microbiol. 2015 Aug 24. [Epub ahead of print] PMID:
See original on MMP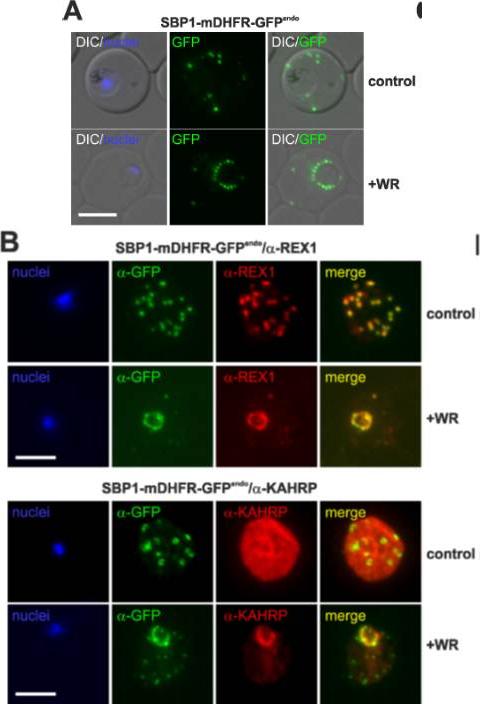
Translocation arrested substrates induce a pan export block that slows parasite development. (A) Representative live fluorescence images of the cell line expressing endogenous SBP1 fused to mDHFR-GFP (SBP1-mDHFR-GFPendo) grown with (+WR) and without WR (control). DIC, differential interference contrast. Size bar: 5 μm. (B) IFA of SBP1-mDHFR-GFPendo parasites using α-REX1 and α-KAHRP in parasites treated with (+WR) and without (control) WR. Size bars: 5 μm. Mesén-Ramírez P, Reinsch F, Blancke Soares A, Bergmann B, Ullrich AK, Tenzer S, Spielmann T. Stable Translocation Intermediates Jam Global Protein Export in Plasmodium falciparum Parasites and Link the PTEX Component EXP2 with Translocation Activity. PLoS Pathog. 2016 May 11;12(5):e1005618.
See original on MMP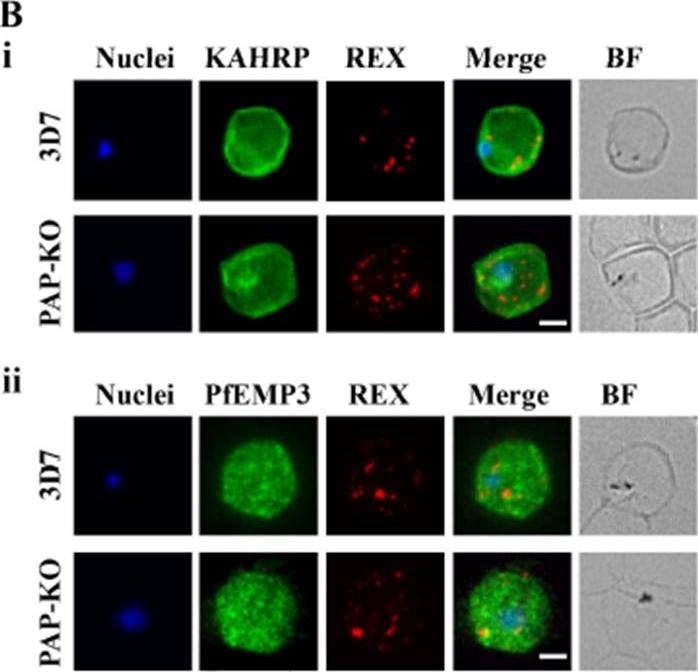
Targeted gene disruption of PfPAP changes the filterability of the infected RBCs but not the expression or localization of KAHRP, REX1, and PfEMP-3. B). Immunofluorescence using anti-KAHRP, REX1, and PfEMP-3 antibodies on 3D7 and 3D7_PAP KO show no difference in the location of these proteins. C). Electron microscopy of 3D7_PAP-KO showing electron dense structures at the surface of the infected erythrocyte indicative of the presence of knobs. Bars are 5 mm.da Silva FL, Dixon MW, Stack CM, Teuscher F, Taran E, Jones MK, Lovas E, Tilley L, Brown CL, Trenholme KR, Dalton JP, Gardiner DL, Skinner-Adams TS. A Plasmodium falciparum S33 proline aminopeptidase is associated with changes in erythrocyte deformability. Exp Parasitol. 2016 Jun 30. pii:S0014-4894(16)30135-7.
See original on MMP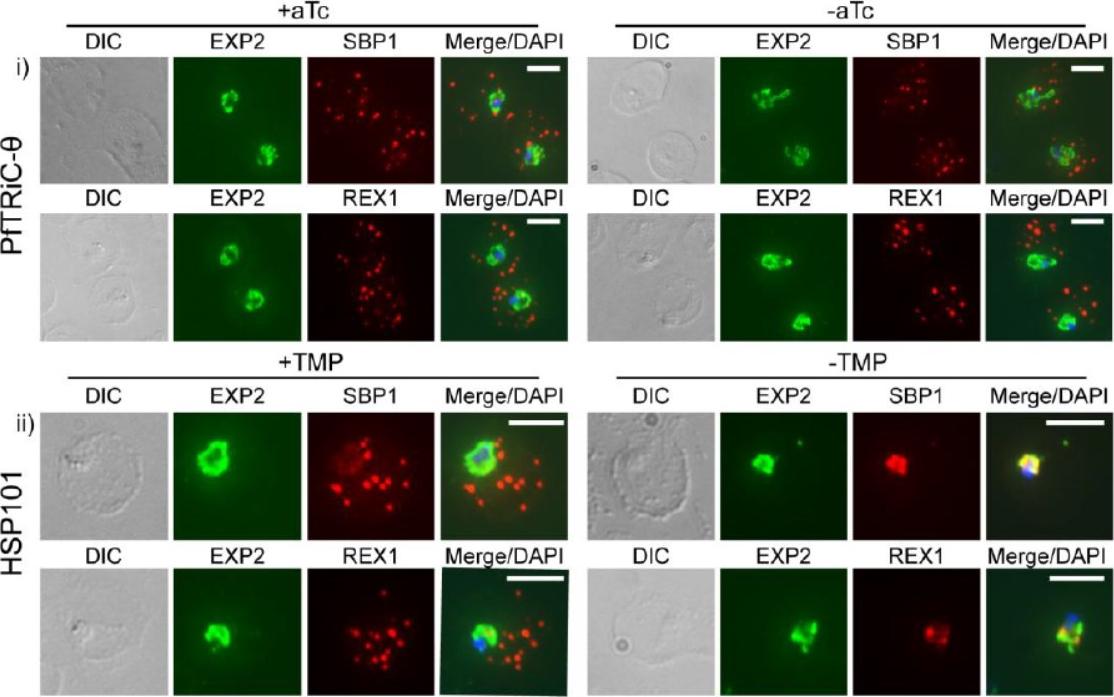
Loss of PfTRiC-θ does not alter export of PfSBP1 or PfREX1. IFA of acetone-fixed ring-stage infected RBC. i) PfTRiC-θ ± 0.5 μM aTc (anhydrotetracycline), ii) HSP101 ± 10 μM trimethoprim (TMP). DAPI stains the parasite nuclei and PfEXP2 delineates the parasite parasitophorous vacuole compartment. Scale bars = 5 μm. Data are representative of two independent assays. DIC = differential interference contrast. Loss of the PfTRiC complex does not alter export of PfSBP1 or PfREX1 (i). This was in contrast to the block in export of PfSBP1 and PfREX1 observed upon TMP removal in the previously characterized PfHSP101-DDD line (ii).Spillman NJ, Beck JR, Ganesan SM, Niles JC, Goldberg DE. The chaperonin TRiC forms an oligomeric complex in the malaria parasite cytosol. Cell Microbiol. 2017 Jan 9.
See original on MMPMore information
| PlasmoDB | PF3D7_0935900 |
| GeneDB | PF3D7_0935900 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | PFI1735c |
| Orthologs | |
| Google Scholar | Search for all mentions of this gene |