PF3D7_0831800 histidine-rich protein II (HRPII)
Disruptability [+]
| Species | Disruptability | Reference | Submitter |
|---|---|---|---|
| P. falciparum 3D7 |
Possible |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen |
Mutant phenotypes [+]
| Species | Stage | Phenotype | Reference | Submitter |
|---|---|---|---|---|
| P. falciparum 3D7 | Asexual |
Possible |
25052298 (Spontaneous loss, not experimental genetics)
Viable parasites occur with the deletion in natural populations |
Theo Sanderson, Wellcome Trust Sanger Institute |
Imaging data (from Malaria Metabolic Pathways)
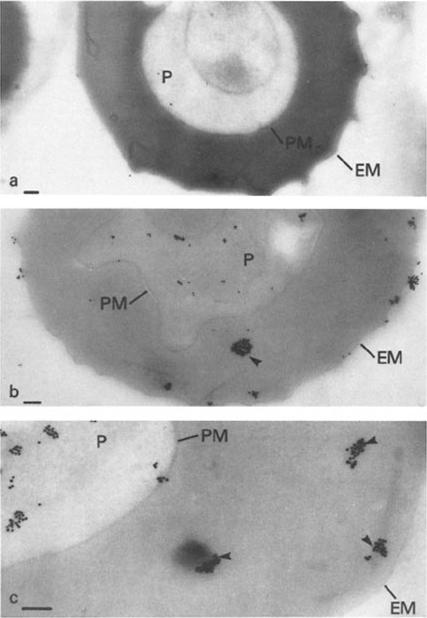
Immunoelectron microscopy of mature trophozoites of K + Malayan Camp P. falciparum with (a) a control IgM mAb (1A1D5) or (b and c) mAb 87. In a, the ascites was diluted to 1:400, and in b and c, the ascites was diluted to 1:6,000 for reaction with the frozen section. Addition of secondary antibody (rabbit anti-mouse Ig) prior to Protein A-gold had no effect on the results (a and c show the results without secondary antibody). Identical results to a were obtained if primary antibody was omitted and Protein A-gold added either directly or after incubation with secondary antibody. P, parasite; PM, parasite plasma membrane; EM, erythrocyte plasma membrane. Dense packets of Protein A-gold particles reacting with the cytoplasm of the host erythrocyte are indicated by arrows in b and c. Bars, 0.5 mm.Pf HRP II localized to several cell compartments including the parasite cytoplasm, as concentrated packets in the host erythrocyte cytoplasm and at the IRBC membrane. The results provide evidence for an intracellular route of transport for a secreted malarial protein from the parasite through several membranes and the host cell cytoplasm.Howard RJ, Uni S, Aikawa M, Aley SB, Leech JH, Lew AM, Wellems TE, Rener J, Taylor DW. Secretion of a malarial histidine-rich protein (Pf HRP II) from Plasmodium falciparum-infected erythrocytes. J Cell Biol. 1986 103:1269-77.
See original on MMP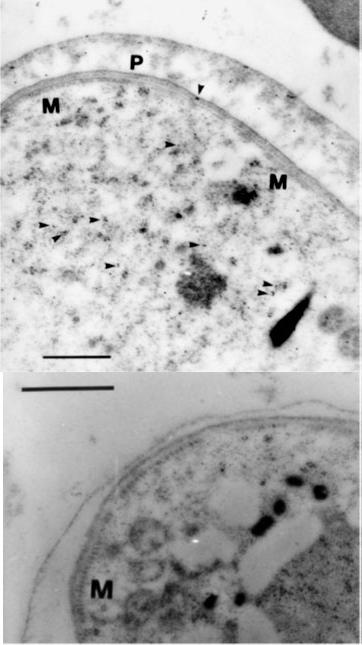
Immunogold electron microscopy for ultrastructural localization of HRPII in the gametocyte. Gametocytes were identified by a corset of microtubules (M) and a three-membrane pellicular complex (P). Arrowheads mark gold particles. (B) Gametocyte labeling with secondary gold-conjugated antibody only. Bar, 500 nm.When the gametocyte matures, the distribution of HRPII to the host cell cytoplasm is limited and may be affected by the formation of the trilaminate membrane in stage IIA gametocytes and certainly a general reduction in protein level. A reduction in HRPII trafficking correlates with cessation of hemoglobin breakdown and subsequent heme processing as gametocytes mature. Authors speculate that HRPII in the infected RBC of late gametocytes is residual protein from synthesis, translocation, and function in earlier stages.Hayward RE, Sullivan DJ, Day KP. Plasmodium falciparum: histidine-rich protein II is expressed during gametocyte development. Exp Parasitol. 2000 96:139-46. A
See original on MMP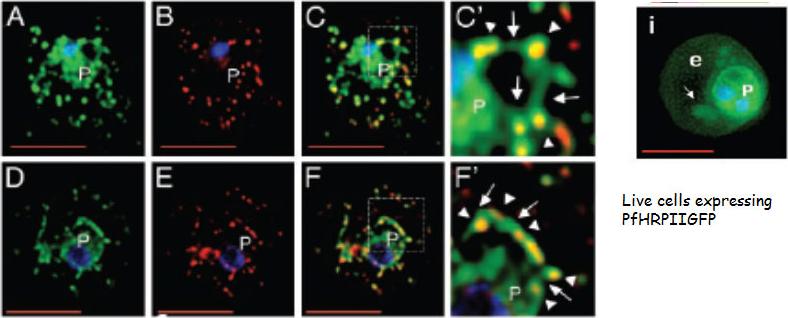
The Maurer’s clefts are intermediates in PfHRPII transport and are targeted by its histidine-rich sequences. Single optical sections of trophozoite infected cells expressing SSGFPHis154–327 (A–C) or PfHRPIIGFP (D–F) are shown. Cells were fixed, permeabilized, subjected to indirect immunofluorescence assays and probed with antibodies to GFP (green) and a Maurer’s cleft resident protein PfSBP (red). Magnification of the boxes in C and F are shown as C’ and F’, respectively. The extent of colocalization is shown in yellow. In C’ and F’, arrows indicate GFP-labeled tubules or vesicles connecting clefts (arrowheads) to each other or the vacuolar parasite (P). Nuclei were stained with Hoechst (blue). (Scale bar, 5 mm.)Lopez-Estraño C, Bhattacharjee S, Harrison T, Haldar K. Cooperative domains define a unique host cell-targeting signal in Plasmodium falciparum-infected erythrocytes. Proc Natl Acad Sci U S A. 2003 100:12402-7. Copyright (2009) National Academy of Sciences, U.S.A
See original on MMP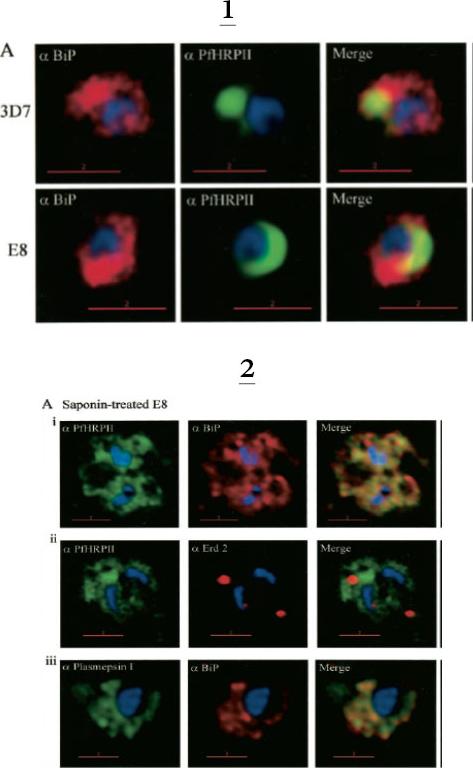
1. Comparative analysis of secretory recruitment of HRPII, export of parasite proteins in E8- and 3D7-infected erythrocytes, and distribution of total PfHRPII, PfHRPIImyc, plasmepsin I, and BiP in parasites and isolate food vacuoles. A, ring-infected red cells from E8 and 3D7-strains were treated with brefeldin A, stained for PfBiP (red), PfHRPII (green), and Hoechst dye (blue) in indirect immunofluorescence assays, and imaged using DeltaVision. Single optical sections showing a merge of red and green as yellow are shown. In the presence of BFA, all cell-associated HRPIIs detected in both E8 and 3D7 reside within regions that stain with the ER marker PfBiP, confirming that HRPII is recruited to the secretory pathway.2. Membrane association of parasite-HRPII in E8 and ACPGFP cells. A, 30–33-h trophozoite-infected red cells from 3D7 and E8 strains were attached to poly-L-lysine-coated cover slips and then permeabilized with 0.01% saponin to release the erythrocytic contents. The adherent cells were then fixed with formaldehyde and probed for the distribution of the indicated proteins by indirect immunofluorescence microscopy. HRPII and plasmepsin I show substantial colocalization with PfBiP in E8 cells but not markers of the Golgi (ERD2).Akompong T, Kadekoppala M, Harrison T, Oksman A, Goldberg DE, Fujioka H, Samuel BU, Sullivan D, Haldar K. Trans expression of a Plasmodium falciparum histidine-rich protein II (HRPII) reveals sorting of soluble proteins in the periphery of the host erythrocyte and disrupts transport to the malarial food vacuole. J Biol Chem. 2002 277:28923-33.
See original on MMP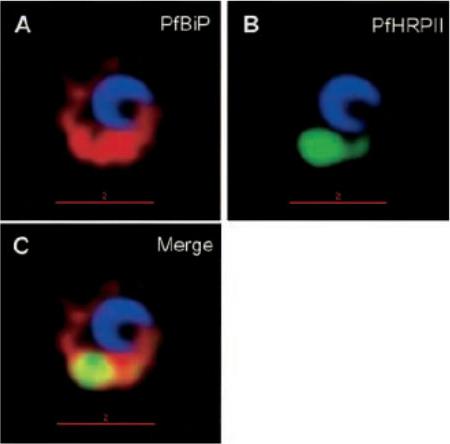
Protein accumulation in the ER in P. falciparum-infected cells treated with brefeldin A. Infected red cells at early times of asexual growth, were incubated with 5 mgml-1 brefeldin A for 24 h and subsequently stained for PfHRPII (green), PfBiP (red), Hoechst (blue for nuclei), in indirect immunofluorescence assays and imaged by DeltaVision deconvolution microscopy. Yellow indicates extent of merge between green and red. The location to the ER may reflect altered (albeit reversible) membrane organization induced by the drug, rather than a specialized organelle. In addition, since the P. falciparum (primary) ER is not well characterized, the context of a secondary ER remains unclear.Haldar K, Mohandas N, Samuel BU, Harrison T, Hiller NL, Akompong T, Cheresh P. Protein and lipid trafficking induced in erythrocytes infected by malaria parasites. Cell Microbiol. 2002 4:383-95.
See original on MMP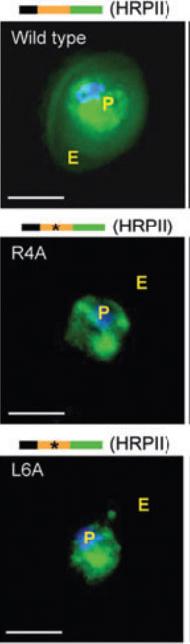
Fluorescent micrographs of live cells expressing green fluorescent protein (GFP) chimeras of the parasite vacuolar translocation sequence containing the wild-type HT motif or point mutations at R4A or L6A, where 4 and 6 indicate the position in the signal. Export of GFP to the erythrocyte (E) is abrogated by both mutations. GFP fluorescence was detected by digitized fluorescence microscopy. Parasite (P) nucleus is Hoechst stained (blue). Schematics above micrographs indicate constructs containing an endoplasmic reticulum–type signal sequence (black) needed for protein delivery to the parasitophorous vacuolar membrane, the vacuolar translocation sequence (orange) of the indicated protein, and the C-terminal GFP protein (green). Stars indicate single amino acid substitutions. Scale bar, 5 μm. HRPII, histidine-rich protein II.Haldar K, Murphy SC, Milner DA, Taylor TE. Malaria: mechanisms of erythrocytic infection and pathological correlates of severe disease. Annu Rev Pathol. 2007; 2:217-49.
See original on MMP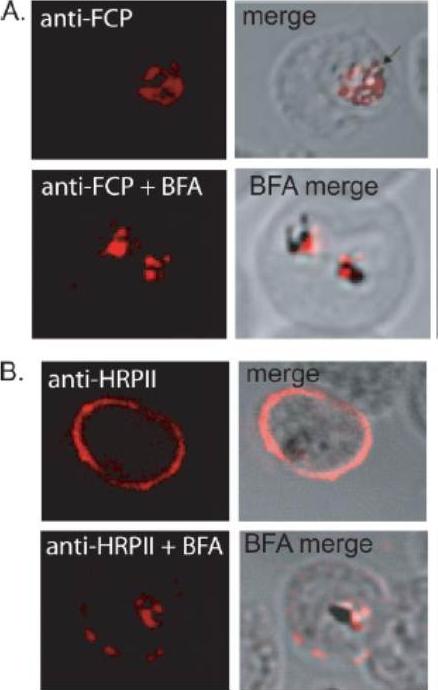
FCP localizes to the FV but does not traffic via the classical or alternate secretory pathways. A, immunofluorescence using anti-FCP revealed a discrete localization to the parasite FV. Ring stage parasites were treated either with 5 mg/ml BFA (anti-FCPP+BFA) or solvent as control (anti-FCP) for 12 h before immunofluorescence microscopy. BFA treatment did not alter FV localization of FCP. B, as a control experiment, parasites treated with BFA (anti-HRPIIBFA) or without BFA (anti-HRPII) were stained using antisera against a known secreted protein, HRPII. HRPII was localized predominantly to the cytoplasm of the host infected erythrocyte (anti-HRPII). BFA treatment resulted in a significant loss in erythrocyte localization of HRPII and a concomitant increase in the parasite cytoplasm (anti-HRPII + BFA).McIntosh MT, Vaid A, Hosgood HD, Vijay J, Bhattacharya A, Sahani MH, Baevova P, Joiner KA, Sharma P. Traffic to the malaria parasite food vacuole: a novel pathway involving a phosphatidylinositol 3-phosphate-binding protein. J Biol Chem. 2007 282:11499-508.
See original on MMP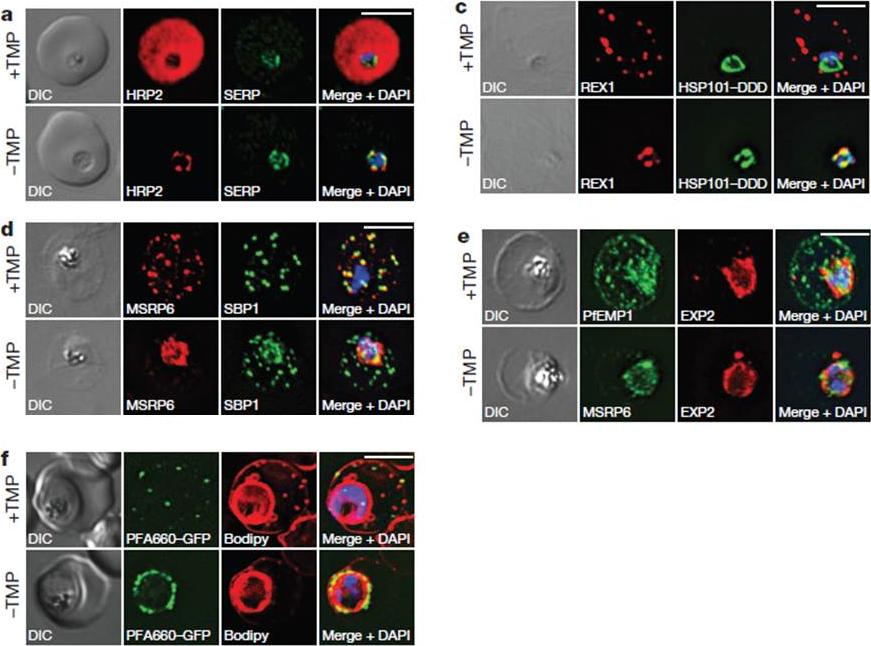
HSP101 is required for export of PEXEL and PNEP proteins. a, c, Immunofluorescence assay (IFA) of ring-stage 13F10 parasites with orwithout TMP (DDD-stabilizing small molecule trimethoprim). TMP was removed in late schizont stage and parasites were allowed to reinvade and grow 18–24 h before fixation with paraformaldehyde (a) or acetone (c). a, IFA of the exported PEXEL-containing protein HRP2. c, IFAs of the PNEP REX1, which colocalizes with HSP101DDD at the PVM in the absence of TMP. d, e, IFA of trophozoite-stage 13F10 parasites with or withoutTMP. TMP was removed in late ring stage and parasites were allowed to develop 12–24 h before fixation with acetone. f, Live fluorescence imaging of 13F10 parasites expressing a PFA660–GFP fusion and labelled with Bodipy TR Ceramide to demarcate the PVM (other membranes are also labelled). TMP treatment as in d, e. All scale bars, 5 mm. Images in c–f are representative of two independent experiments.Beck JR, Muralidharan V, Oksman A, Goldberg DE. PTEX component HSP101 mediates export of diverse malaria effectors into host erythrocytes. Nature. 2014 Jul 16. [Epub ahead of print]
See original on MMP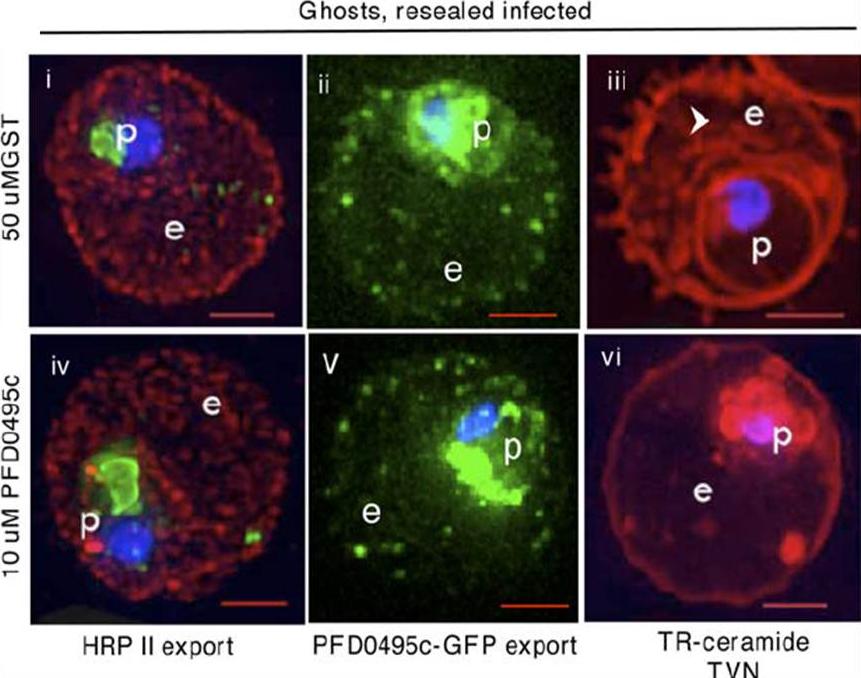
PFD0495c is important for proper development of the TVN. Trophozoite stage parasites in ghosts resealed with 50 mM GST (i, ii, iii), or 10 mM GST. PFD0495cCterm (Biv, v, vi), were infected with: (i, iv) 3D7 P. falciparum and probed with anti-HRPII (red), anti-MSP1 (green, to mark the parasite) and Hoechst 33342 (blue to stain parasite DNA); (ii, v) clone 1 expressing PFD0495c-GFP and visualized live for green fluorescence; (iii, vi) 3D7 P. falciparum stained with the membrane permeable probe TR-ceramide and visualized live.Tamez PA, Bhattacharjee S, van Ooij C, Hiller NL, Llinás M, Balu B, Adams JH, Haldar K. An erythrocyte vesicle protein exported by the malaria parasite promotes tubovesicular lipid import from the host cell surface. PLoS Pathog. 2008 Aug 8;4(8):e1000118.
See original on MMP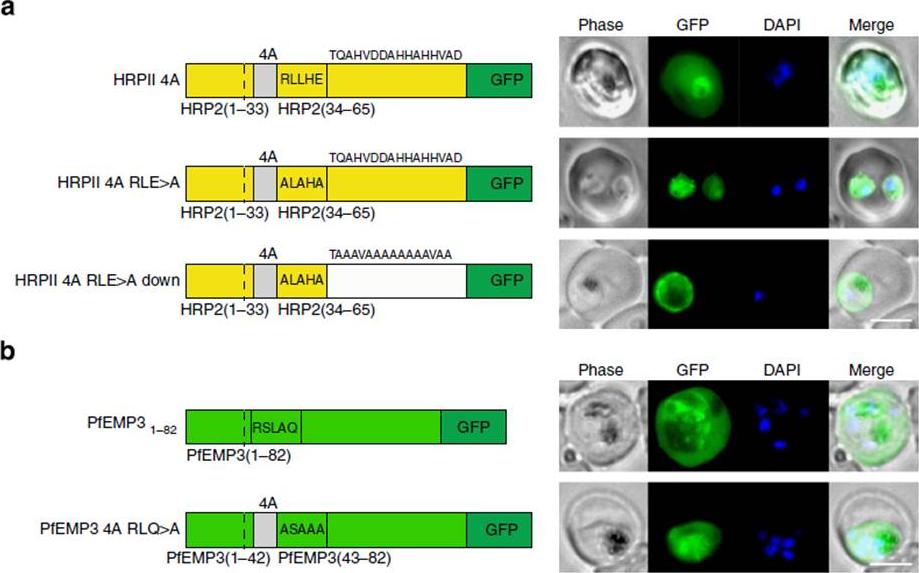
PEXEL-independent export of PEXEL proteins does not occur in P. falciparum. (a) HRPII 4A is exported to the P. falciparum-infected erythrocyte. HRPII 4A RLE4A and HRPII 4A RLE4A Down are not exported. Scale bar, 5 mm. (b) PfEMP31–82 is exported by P. falciparum to the erythrocyte, but PfEMP3 4A RLQ4A is not. Scale bar, 5 mm.Boddey JA, O'Neill MT, Lopaticki S, Carvalho TG, Hodder AN, Nebl T, Wawra S, van West P, Ebrahimzadeh Z, Richard D, Flemming S, Spielmann T, Przyborski J, Babon JJ, Cowman AF. Export of malaria proteins requires co-translational processing of the PEXEL motif independent of phosphatidylinositol-3-phosphate binding. Nat Commun. 2016 Feb 1;7:10470.
See original on MMP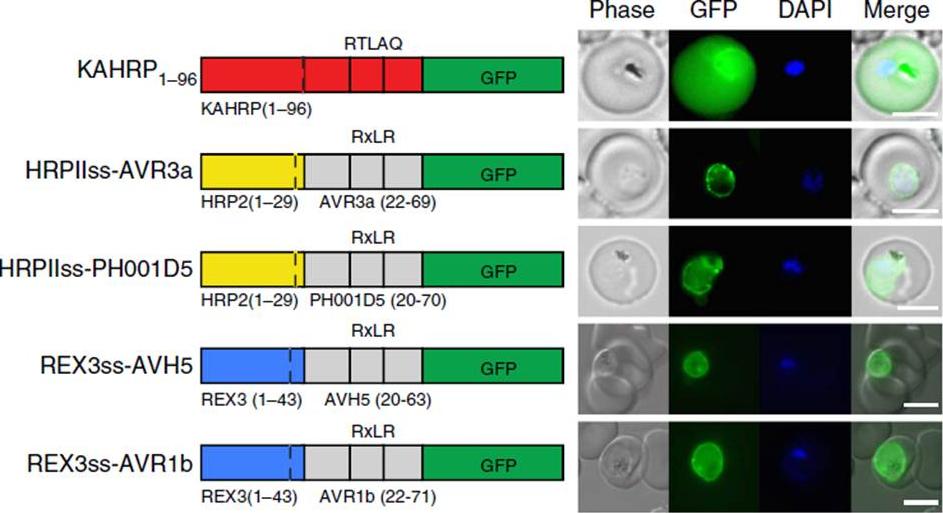
Oomycete RxLR does not mediate export in P. falciparum. (a) KAHRP1–96 is exported to the P. falciparum-infected erythrocyte; however, HRPIIss-AVR3a, HRPIIss-PH001D5, REX3ss-AVH5 and REX3ss-AVR1b are not exported but accumulate in the parasite and PV. Scale bar, 5 mm.Boddey JA, O'Neill MT, Lopaticki S, Carvalho TG, Hodder AN, Nebl T, Wawra S, van West P, Ebrahimzadeh Z, Richard D, Flemming S, Spielmann T, Przyborski J, Babon JJ, Cowman AF. Export of malaria proteins requires co-translational processing of the PEXEL motif independent of phosphatidylinositol-3-phosphate binding. Nat Commun. 2016 Feb 1;7:10470.
See original on MMPMore information
| PlasmoDB | PF3D7_0831800 |
| GeneDB | PF3D7_0831800 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | MAL7P1.231 |
| Orthologs | |
| Google Scholar | Search for all mentions of this gene |