PF3D7_0631400 Pfmc-2TM Maurer's cleft two transmembrane protein (MC-2TM)
Disruptability [+]
| Species | Disruptability | Reference | Submitter |
|---|---|---|---|
| P. falciparum 3D7 |
Refractory |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen |
Mutant phenotypes [+]
None reported yet. Please press the '+' button above to add one.Imaging data (from Malaria Metabolic Pathways)
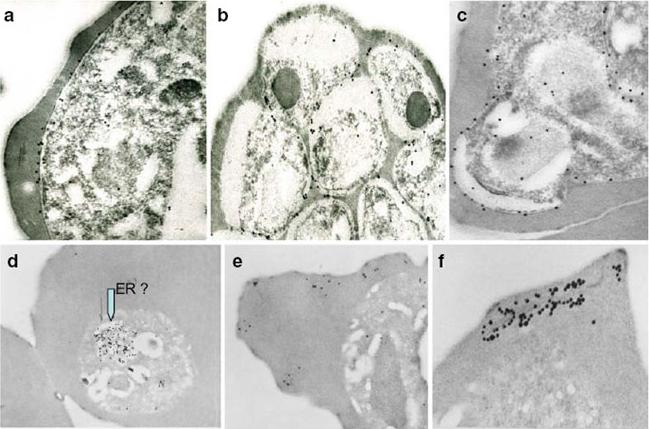
Immunoelectron micrographs of young trophozoite-, late trophozoite-, young schizont-, and late schizont-infected erythrocytes reactive with rabbit antisera against PfMC-2TM peptides, 703, 705, and 713 (identical sequences in all PfMC-2TM-coding genes and Mab SP1A6 against the 130 kDa Maurer’s cleft protein. a–c Peptide-specific antisera are reactive with the PV/PVM and weakly around clefts. d–f Mab SP1A6 is reactive with structures within the parasite in young trophozoites, probably the endoplasmic reticulum (d), late trophozoites in erythrocyte cytoplasm under knobs (e), and in young schizonts around longitudinal clefts in the erythrocyte cytoplasm (f).Tsarukyanova I, Drazba JA, Fujioka H, Yadav SP, Sam-Yellowe TY. Proteins of the Plasmodium falciparum two transmembrane Maurer's cleft protein family, PfMC-2TM, and the 130 kDa Maurer's cleft protein define different domains of the infected erythrocyte intramembranous network. Parasitol Res. 2009 104:875-91. Copyright Springer 2011.
See original on MMP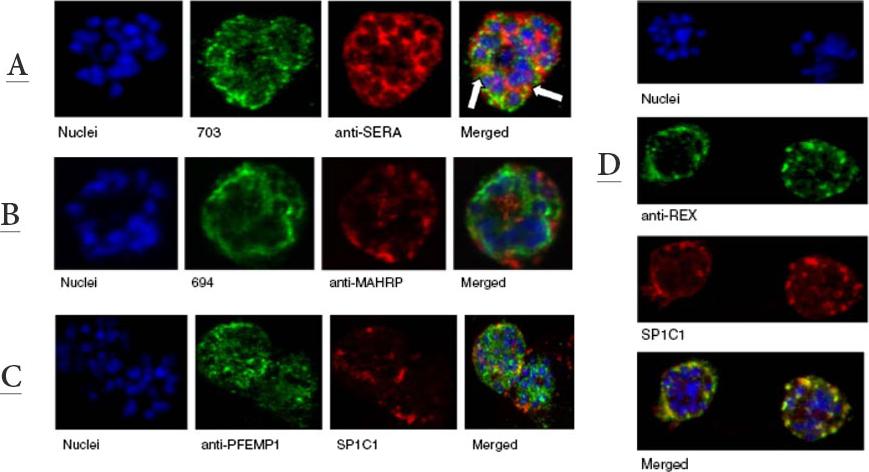
A. Immunofluorescence assay of rabbit antisera against PfMC-2TM peptides colocalized with anti-SERA 1 antibodies. Arrows indicate areas of colocalization between SERA 1 andPfMC-2TMB. Mouse antisera against MAHRP1 MAL13P1.413 was incubated with antisera against PfMC-2TM peptides.C. Immunofluorescence assay of rabbit antisera against PfEMP1 with Mabs SP1C1. PfEMP1 colocalized with PfMC-2TM, D. Immunofluorescence assay of rabbit antisera against REX1 PFI1735c colocalized with Mabs SP1C1. Optical sections were collected at a step size of 0.5 μm from the top to the bottom of the optical plane. Continuous overlap of the antibodies was observed.Tsarukyanova I, Drazba JA, Fujioka H, Yadav SP, Sam-Yellowe TY. Proteins of the Plasmodium falciparum two transmembrane Maurer's cleft protein family, PfMC-2TM, and the 130 kDa Maurer's cleft protein define different domains of the infected erythrocyte intramembranous network. Parasitol Res. 2009 104:875-91. Copyright Springer 2011.
See original on MMP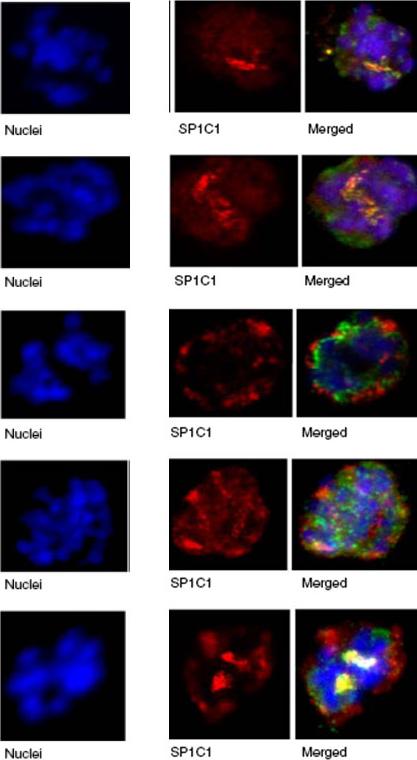
Immunofluorescence assay of rabbit antisera against PfMC-2TM peptides colocalized with Mab SP1C1. Immunofluorescence assays (IFA) in this figure and Figs. 4, 5, 6, 7, 8, and 9 were performed using the same stage of parasites. Trophozoite- and schizont-infected erythrocytes were incubated with mouse and rabbit primary antibodies, followed by secondary antibodies directed to both species, conjugated to different colored fluorochromes; Alexa 488 and Alexa 568 (Molecular Probes) for detection of primary antibodies. Antibody staining was considered colocalized if there was overlap (yellow/orange) in the staining pattern obtained with the two antibodies on or within the same structure or within domains of the intra-membranous network.Tsarukyanova I, Drazba JA, Fujioka H, Yadav SP, Sam-Yellowe TY. Proteins of the Plasmodium falciparum two transmembrane Maurer's cleft protein family, PfMC-2TM, and the 130 kDa Maurer's cleft protein define different domains of the infected erythrocyte intramembranous network. Parasitol Res. 2009 104:875-91. Copyright Springer 2011.
See original on MMP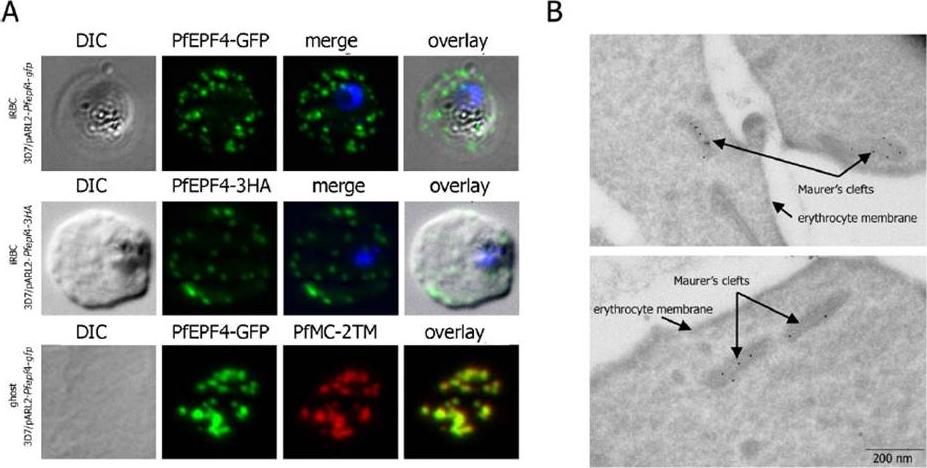
Localization and topology of the PfEPF4 proteins. A. Fluorescent patterns of iRBCs infected by 3D7/pARL2-Pfepf4-gfp (live imaging) and 3D7/pARL2-Pfepf4-3HA (immunodetection using anti-HA antibodies) and of resealed ghosts from 3D7/pARL2-Pfepf4-gfp iRBCs (GFP fluorescence, in green, and immunodetection using anti-PfMC-2TM antibodies, in red). RBCs and ghost preparations were incubated with DAPI for nucleus labelling. B. Immunoelectron microscopy using anti-GFP antibodies. PfEPF4 (A) display a pattern of fluorescent dots in the iRBC cytoplasm. PfEPF4-GFP colocalized with a Maurer’s cleft transmembrane protein, PfSBP1 or PfMC-2TM2 when resealed ghosts from iRBCs were analysed by immunofluorescence. The localization of PfEPF4 at the Maurer’s clefts was confirmed by immunoelectron-microscopy.Mbengue A, Audiger N, Vialla E, Dubremetz JF, Braun-Breton C. Novel Plasmodium falciparum Maurer's clefts protein families implicated in the release of infectious merozoites. Mol Microbiol. 2013 88(2):425-42
See original on MMP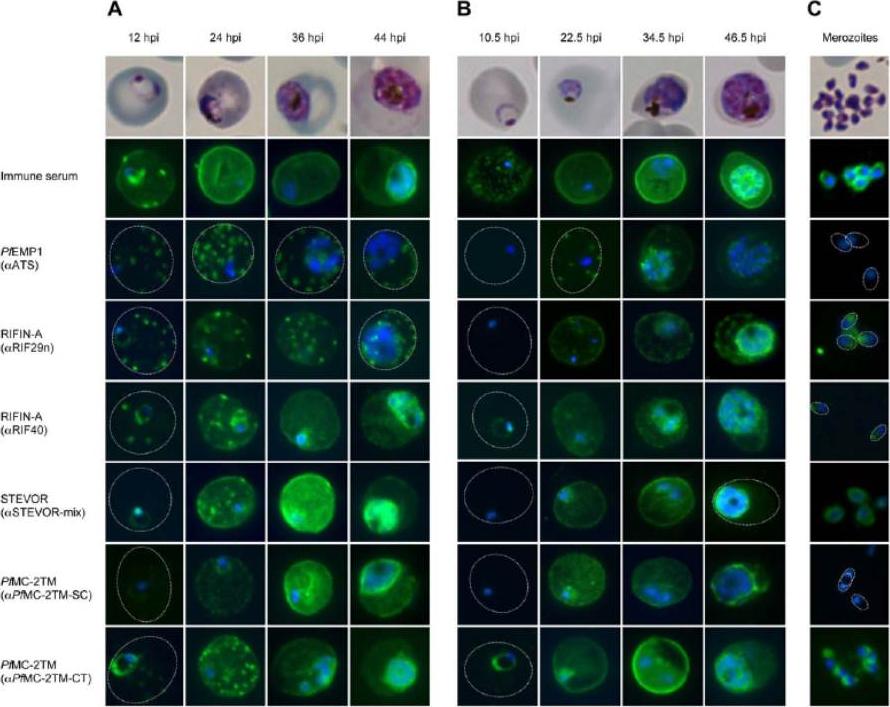
Localization of VSAs during the intraerythrocytic developmental cycle. A-C: Representative immunofluorescence images of the indicated VSAs in different parasite developmental stages of clinical isolate #4 (A), 3D7 parasites (B), and free merozoites from isolate #1 (C). First row: Giemsa staining of the corresponding parasitic stage. Second row: Positive control serum obtained from a semi-immune patient. Third row: PfEMP1-specific antibody, showing the presence of the protein in Maurer’s clefts over the entire time course (A, B). Third to eighth rows: 2TM proteins were exported into the host cell (12–36 hpi) during the trophozoite stage but remained inside the parasite in the schizont stage (48 hpi). Proteins of the RIFIN-A family frequently localized to Maurer’s clefts, particularly when using the a-RIF29n antiserum, and the erythrocyte membrane; STEVOR and PfMC-2TM localized predominantly to the erythrocyte membrane (A, B). RIFIN and STEVOR proteins were also observed at the apical tip or at the merozoite membrane, respectively. Isolate #1 also exhibited PfMC-2TM-specific fluorescence in free merozoites when using the a-PfMC-2TM-CT antiserum (C). All antibodies were visualized with Alexa488-conjugated secondary antibody (green), and nuclei were stained with DAPI (blue).Bachmann A, Petter M, Tilly AK, Biller L, Uliczka KA, Duffy MF, Tannich E, Bruchhaus I. Temporal Expression and Localization Patterns of Variant surface Antigens in Clinical Plasmodium falciparum Isolates during Erythrocyte Schizogony. PLoS One. 2012;7(11): e49540. PMID:
See original on MMP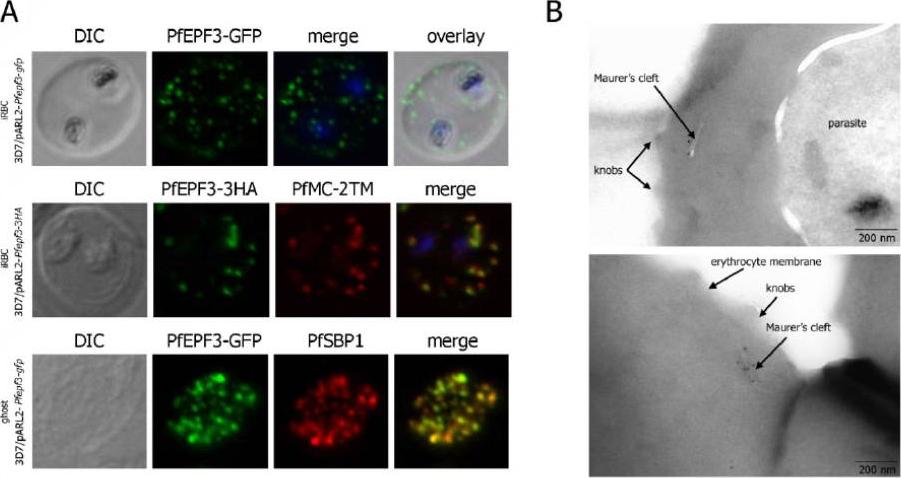
Localization and topology of the PfEPF3 proteins. A. Fluorescent patterns of iRBCs infected by 3D7/pARL2-Pfepf3-gfp (live imaging) and 3D7/pARL2-Pfepf3-3HA (immunodetection using anti-HA antibodies, in green, and anti-PfMC-2TM antibodies, in red) and of resealed ghosts from 3D7/pARL2-Pfepf3-gfp iRBCs (GFP fluorescence, in green, and immunodetection using anti-SBP1 antibodies, in red). RBCs and ghost preparations were incubated with DAPI for nucleus labelling. B. Immunoelectron microscopy using anti-GFP antibodies.Mbengue A, Audiger N, Vialla E, Dubremetz JF, Braun-Breton C. Novel Plasmodium falciparum Maurer's clefts protein families implicated in the release of infectious merozoites. Mol Microbiol. 2013 88(2):425-42 PMID: .
See original on MMP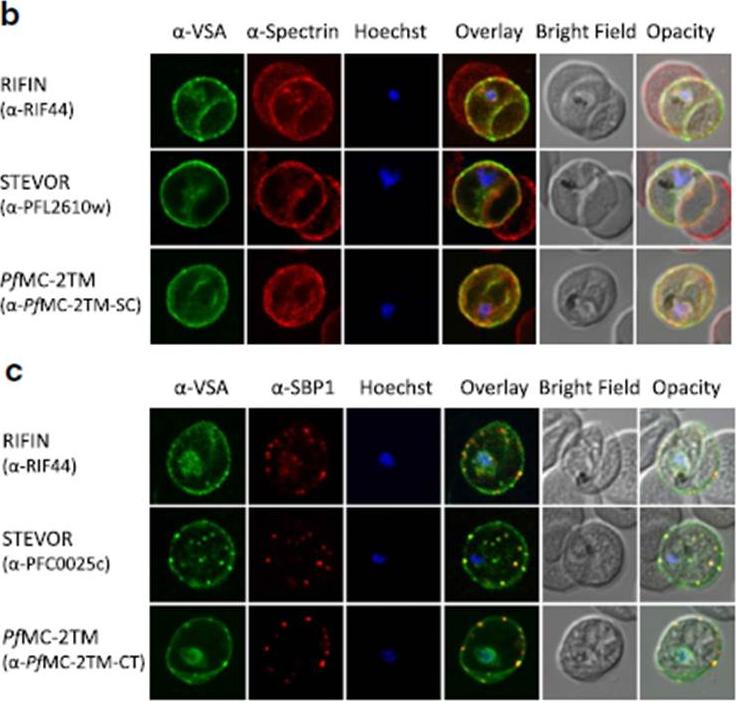
b Co-localization of α-RIF44, STEVOR α-PFL2610w and α-PfMC-2TM-SC (green) with human spectrin (red). c Co-localization of α-RIF44, STEVOR α-PFC0025c and α-PfMC-2TM-CT (green) with SBP1 (red).Bachmann A, Scholz JA, Janßen M, Klinkert MQ, Tannich E, Bruchhaus I, Petter M. A comparative study of the localization and membrane topology of members of the RIFIN, STEVOR and PfMC-2TM protein families in Plasmodium falciparum-infected erythrocytes. Malar J. 2015 Jul 14:274.
See original on MMP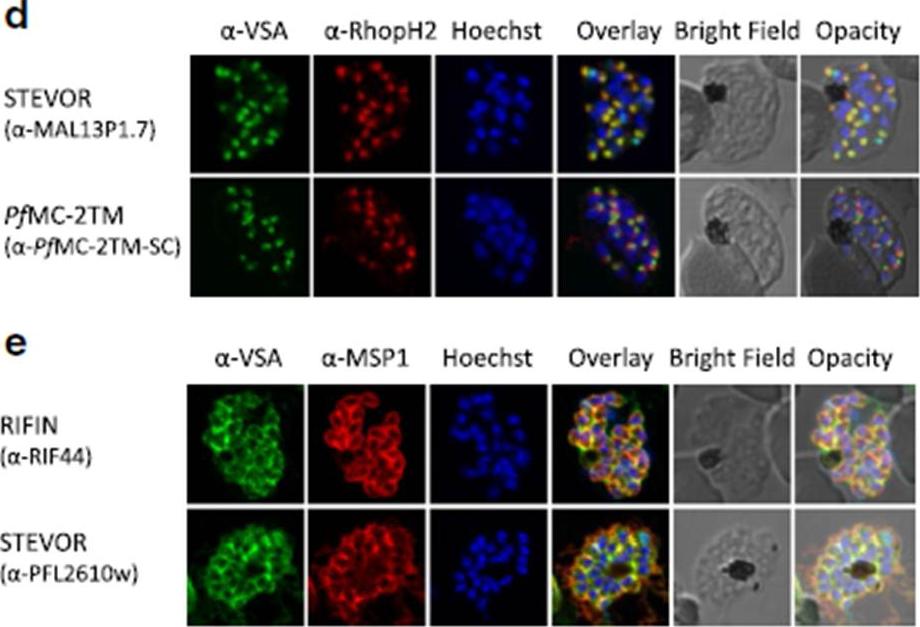
d Co-localization of STEVOR α-MAL13P1.7 or α-PfMC-2TM-SC (green) with the rhoptry marker RhopH2 (red). e Co-localization of α-RIF44 and STEVOR α-PFL2610w (green) with the merozoite surface protein MSP1 (red). Variant surface antigens (VSA).Bachmann A, Scholz JA, Janßen M, Klinkert MQ, Tannich E, Bruchhaus I, Petter M. A comparative study of the localization and membrane topology of members of the RIFIN, STEVOR and PfMC-2TM protein families in Plasmodium falciparum-infected erythrocytes. Malar J. 2015 Jul 14:274.
See original on MMP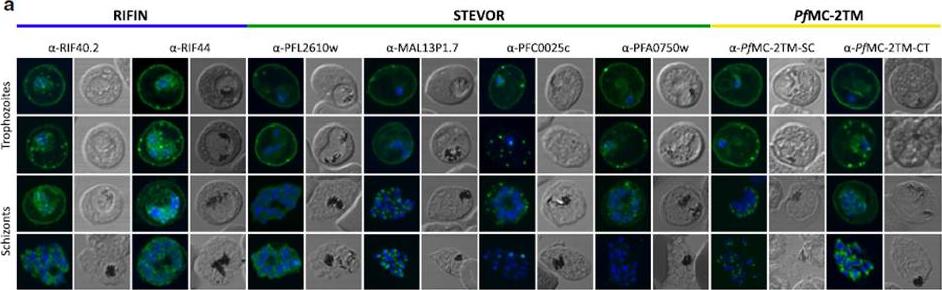
Localization of small VSA in infected erythrocytes using confocal immunofluorescence analysis. a Asexual parasites of the 3D7 parasite clone at the trophozoite and schizont stages were fixed with methanol and small VSA localization was visualized using antibodies directed against RIFIN (α-RIF40.2, α-RIF44), STEVOR (α-PFL2610w, α-MAL13P1.7, α-PFC0025c, α-PFA0750w) and PfMC-2TM (α-PfMC-2TM-SC, α-PfMC-2TM-CT) proteins (green). Nuclei were stained with Hoechst33342 (blue).Bachmann A, Scholz JA, Janßen M, Klinkert MQ, Tannich E, Bruchhaus I, Petter M. A comparative study of the localization and membrane topology of members of the RIFIN, STEVOR and PfMC-2TM protein families in Plasmodium falciparum-infected erythrocytes. Malar J. 2015 Jul 14:274.
See original on MMP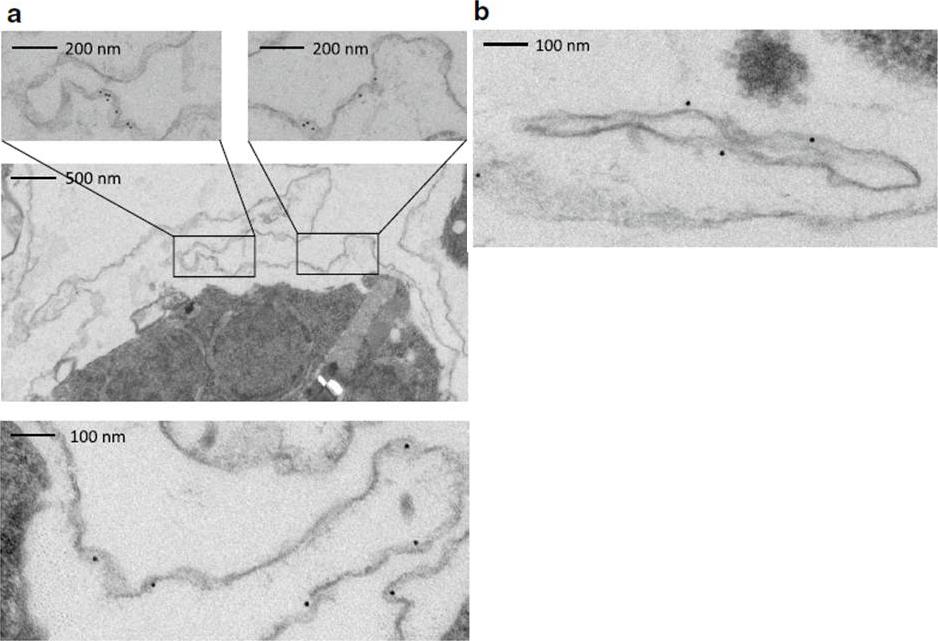
Immunoelectron microscopy of saponin permeabilized IE to confirm PfMC-2TM presence at the erythrocyte membrane. a, b A preembedding staining protocol was applied to analyse PfMC-2TM membrane association by immunoelectron microscopy. Trophozoite IE were permeabilized with saponin and incubated with the immune serum rabbit α-PfMC-2TM-CT (I) or the respective pre-immune serum (PI). Recognized proteins are visualized with 10 nm gold particles. Different sections are shown depicting PfMC-2TM association with the erythrocyte membrane (a) and with Maurer’s clefts (b).Bachmann A, Scholz JA, Janßen M, Klinkert MQ, Tannich E, Bruchhaus I, Petter M. A comparative study of the localization and membrane topology of members of the RIFIN, STEVOR and PfMC-2TM protein families in Plasmodium falciparum-infected erythrocytes. Malar J. 2015 Jul 14:274.
See original on MMPMore information
| PlasmoDB | PF3D7_0631400 |
| GeneDB | PF3D7_0631400 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | 2270.t00309, MAL6P1.15, PFF1525c |
| Orthologs | |
| Google Scholar | Search for all mentions of this gene |