PF3D7_0207600 serine repeat antigen 5 (SERA5)
Disruptability [+]
| Species | Disruptability | Reference | Submitter |
|---|---|---|---|
| P. falciparum 3D7 |
Refractory |
17893128 | Theo Sanderson, Wellcome Trust Sanger Institute |
| P. falciparum 3D7 |
Refractory |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen |
Mutant phenotypes [+]
| Species | Stage | Phenotype | Reference | Submitter |
|---|---|---|---|---|
| P. falciparum 3D7 | Asexual |
Egress defect |
28683142 DiCre excision led to viable parasites but with a 50% reduced multiplication rate. There was an egress phenotype, with premature rupture of the host membranes and a failure of the merozoites to separate efficiently. |
Theo Sanderson, Wellcome Trust Sanger Institute |
Imaging data (from Malaria Metabolic Pathways)
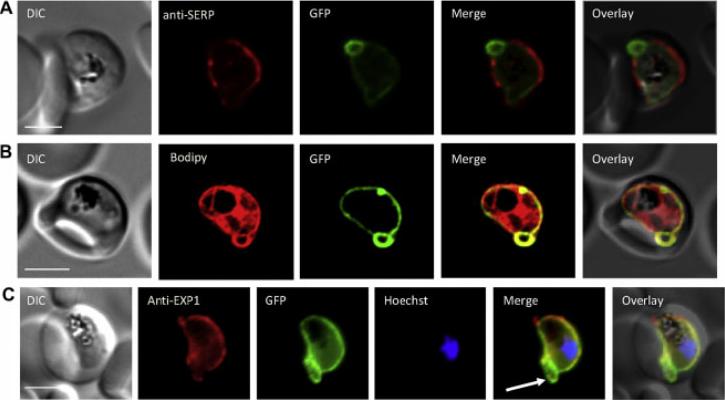
Localization of PfAK2. (A) Co-localization of PfAK2-GFP with serine-rich protein (SERA5). (B) A comparison the localization of PfAK2-GFP with Bodipy-TR-ceramide shows that the loops contain membranous material. (C) Co-localization of PfAK2-GFP with Plasmodium falciparum exported protein (Exp1) by using an anti-Exp1 antibody. Scale bar, 3 mm. PfAK2 localized to a ring-like structure around the 264 parasite, and ‘‘loops’’ apparently connected to the PVM. PfAK2-GFP co localizes with the PV resident protein SERA5 which seems to be excluded from the ‘‘loops’’ suggesting that PfAK2-GFP in actual fact associates with the membrane of the PV, rather than being found in a soluble state in the PV lumen.Ma J, Rahlfs S, Jortzik E, Heiner Schirmer R, Przyborski J, Becker K. Subcellular localization of adenylate kinases in Plasmodium falciparum. FEBS Lett. 2012 Jul 17. [Epub ahead of print]
See original on MMP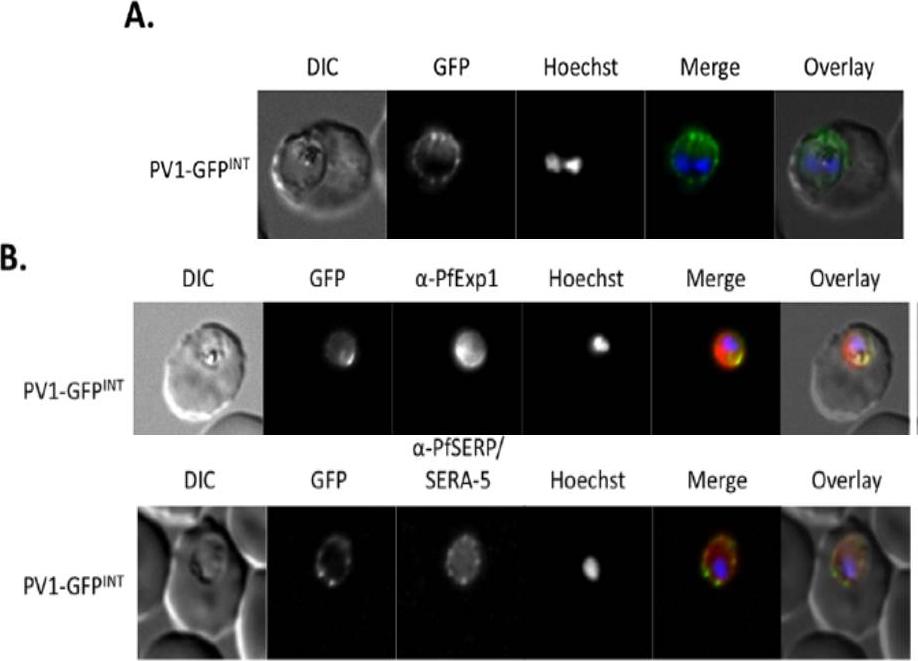
Live cell imaging. (A) Left: live cell imaging of the PV1-GFPINT transgenic line. (B) Upper panel, colocalisation of PV1-GFP chimera with PfExp1. Lower panel, colocalisation of PV1-GFP chimera with PfSERA-5. In merge and overlay: GFP, green; specific antibody, red; Hoechst, blue. Immunofluorescence co-localisation using antibodies against the PV and PVM markers SERA-5 and Exp-1 respectively verified that this fluorescent signal represented the PV.Chu T, Lingelbach K, Przyborski JM. Genetic evidence strongly support an essential role for PfPV1 in intra-erythrocytic growth of P. falciparum. PLoS One. 2011 6(3):e18396.
See original on MMP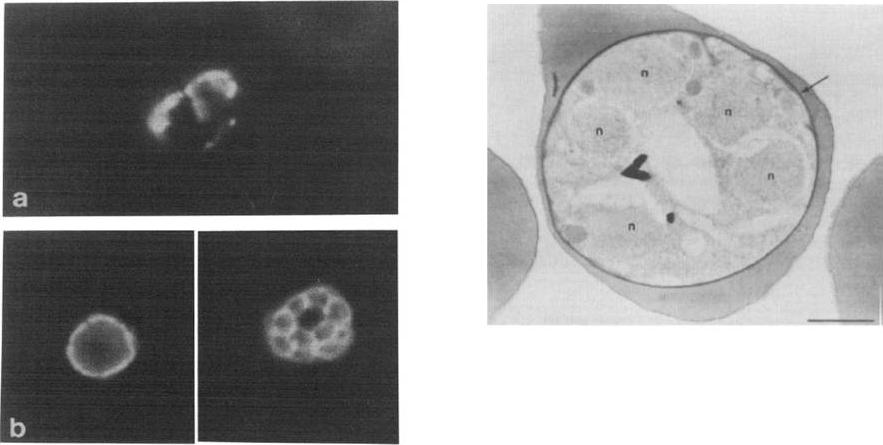
Left: Indirect immunofluorescence assay with McAb 24C6 1F1 on P. falciparum schizont infected human erythrocytes unfixed (a) or formalin fixed (b) before drying. The periphery of the parasites is well delineated m fixed parasites. Irregular peripheral patterns are obtained on unfixed material.Right: Immunoperoxidase detection of P 126 by McAb 24C61F1 in P. falciparum schizont The electron dense deposit (arrow) is located on the parasitophorous vacuole surrounding the parasite (n: parasite nuclei). Bar represents 1 mm.Delplace P, Fortier B, Tronchin G, Dubremetz JF, Vernes A. Localization, biosynthesis, processing and isolation of a major 126 kDa antigen of the parasitophorous vacuole of Plasmodium falciparum. Mol Biochem Parasitol. 1987 23:193-201. Copyright Elsevier
See original on MMP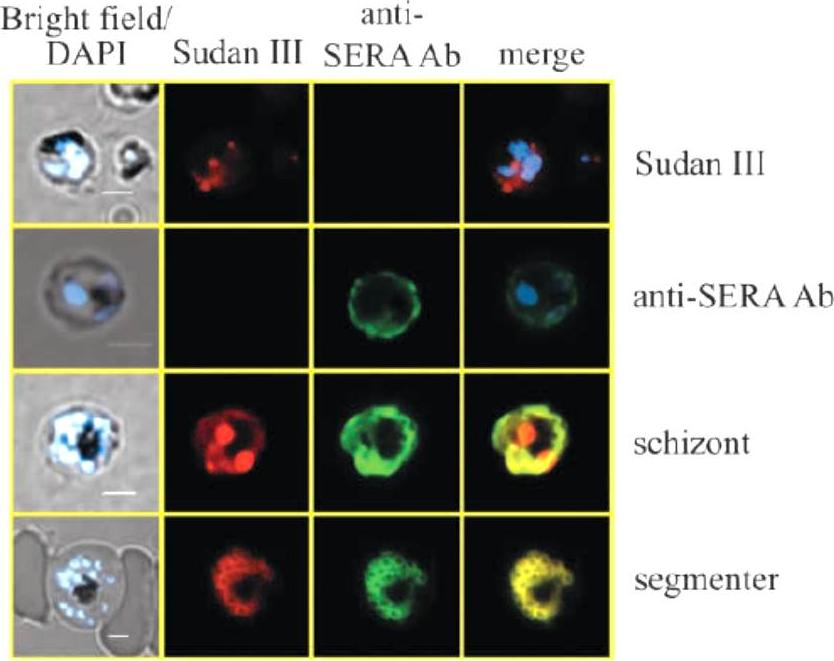
Co-localization of lipid bodies with the parasitophorous vacuole marker protein SERA. (A) Fluorescent microscopic analysis of schizont-stage P. falciparum-infected erythrocyte stained with Sudan III and anti-SERA Ab. From left to right, panels represent bright-field overlaid with DAPI, Sudan III, SERA and overlay of Sudan III and SERA. Yellow areas denote regions of overlap. Single-stained cells (top two rows) exhibit virtually no fluorescence with the opposing filter.Palacpac NM, Hiramine Y, Mi-ichi F, Torii M, Kita K, Hiramatsu R, Horii T, Mitamura T. Developmental-stage-specific triacylglycerol biosynthesis, degradation and trafficking as lipid bodies in Plasmodium falciparum-infected erythrocytes. J Cell Sci. 2004 117:1469-80.
See original on MMP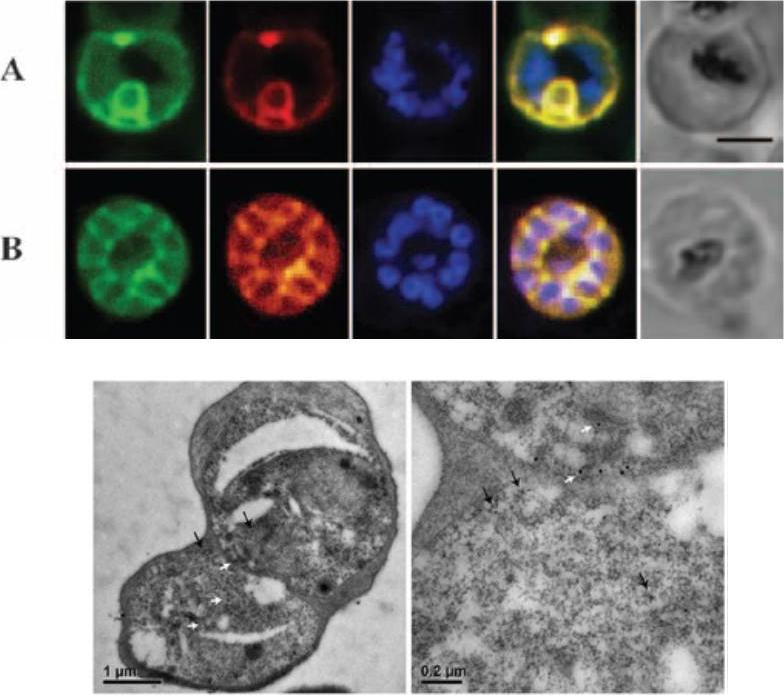
SBP1 (SERA5 targeting peptide) colocalizes with SERA5 within the parasitophorous vacuole of mature blood-stage parasites. Late trophozoites (A) or schizonts (B) were probed with biotinylated SBP1 (column 1; green) and MAb 3E8 (column 2; red). Parasite nuclei (column 3) were stained (blue) using the DNA stain Hoechst 33258. Areas of colocalization for SBP1 and SERA5 are observed as yellow areas (column 4) in the superimposed fluorescence patterns for biotinylated SBP1 and MAb 3E8. The transmission images of parasites viewed using light microscopy are shown in column 5. Bar, 2 mm. SBP1 sequence: LVCHPAVPALLCAR.Lower panel: Immuno-EM images of synchronized parasites cultured in the presence of biotinylated SBP1 (710 M). Sections were probed with MAb 2F3 (to detect SERA5) and avidin (to detect biotinylated SBP1) and visualized using 18-nm and 10-nm gold beads, respectively. SERA5 was generally restricted to the parasitophorous vacuole.Fairlie WD, Spurck TP, McCoubrie JE, Gilson PR, Miller SK, McFadden GI, Malby R, Crabb BS, Hodder AN. Inhibition of malaria parasite development by a cyclic peptide that targets the vital parasite protein SERA5. Infect Immun. 2008 76:4332-4344.
See original on MMP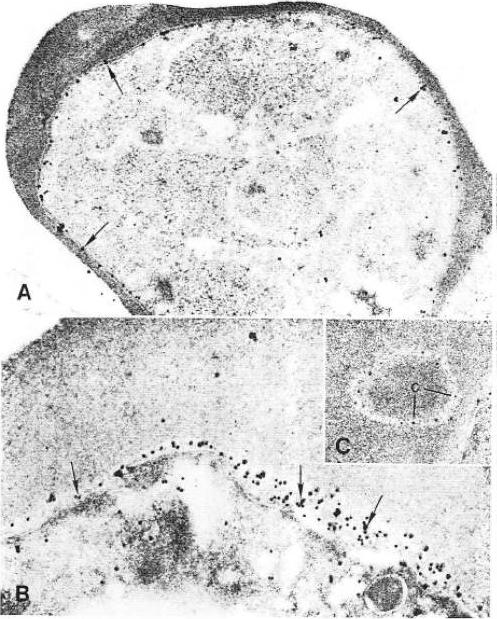
Test of Ag2 localization by immunoeleclron micrography. Electron microscopy micrographs was performed on sections whjch were reacted wiih rabbit anti-Ag2 sera and goat anti-rabbit IgG conjugated to gold particles. A: The schizont membrane is labeled with gold particles (arrow) x30.000. B: Gotd parlicles larrow) are seen on the parasitophorous nembrane and in the space of the parasitophorous vacuole x45,000. C: Gold particles are present within a cleft (arrow) x 36,000.Jakobsen PH, Grellier P, Theander TG, Behrendt N, Torii M, Aikawa M, Schrevel J, Jepsen S. Identification and localization of a soluble antigen, Ag2, of 136 kDa from Plasmodium falciparum in vitro cultures. APMIS. 1991 99:155-62. Copyright John Wiley & Sons Ltd. 2010.
See original on MMP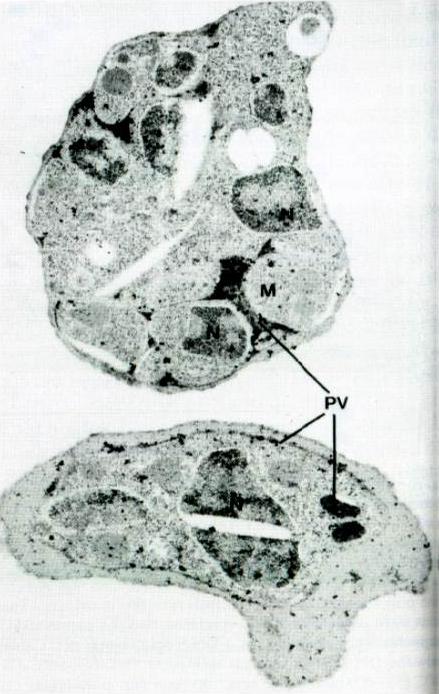
Immunoelectron micrograph of a P. falciparum schizont section reacted with an antiserum against the fusion protein expressed by pEX140-1. (N, nucleus; M, merozoite; PV, parasitophorous vacuole), x 18000. pEX140-1 codes for 33 kDa polypeptide resulting from the fusion of the SERA5 cDNA and the lgt11 plasmid. Sera5 is located mainly within the parasitophorous vacuole. Additional labelling of the parasite surface as well as the vacuolar membrane cannot be excluded.Knapp B, Hundt E, Nau U, Küpper HA. Molecular cloning, genomic structure and localization in a blood stage antigen of Plasmodium falciparum characterized by aserine stretch. Mol Biochem Parasitol. 1989 32:73-83. Copyright Elsevier 2010.
See original on MMP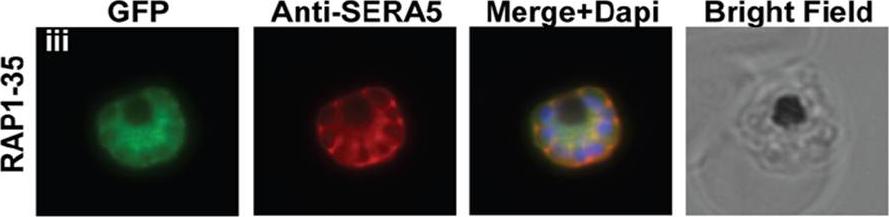
The RAP1-35 construct, GFP is trafficked to the PV. RAP1-35-GFP was targeted to the parasitophorous vacuole (PV), the default destination for the secretory pathway, where it is co-localized with SERA5.Richard D, Kats LM, Langer C, Black CG, Mitri K, Boddey JA, Cowman AF, Coppel RL. Identification of Rhoptry Trafficking Determinants and Evidence for a Novel Sorting Mechanism in the Malaria Parasite Plasmodium falciparum. PLoS Pathog. 2009 5(3):e1000328.
See original on MMP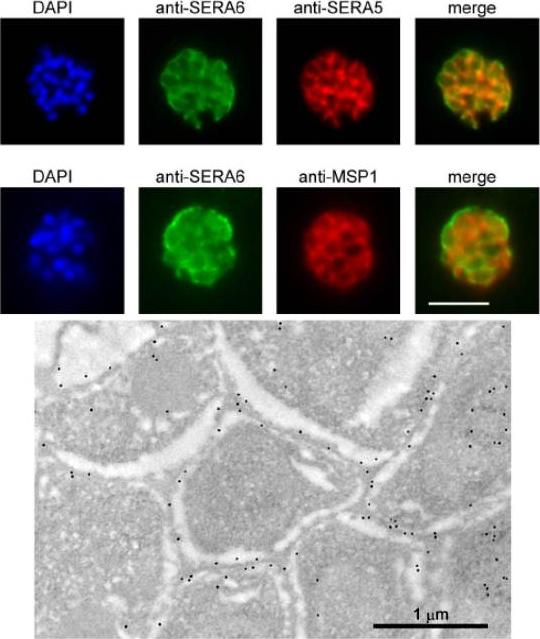
SERA6 is a soluble parasitophorous vacuole (PV) protein which may partially interact with the PVM. Upper panel: IFA demonstrates colocalization of the anti-SERA6 and anti-SERA5 signal in mature schizonts, except that the SERA6 signal additionally shows an association with the outer confines of the intracellular parasite, probably corresponding to the PVM. Scale bar, 5 μm. Lower panel: Immuno-electron microscopic localization of SERA6 in a P. falciparum schizont, using the anti-S6C1 antibodies labelled with 10 nm immunogold. The majority of the signal is clearly associated with the peripheral space between intracellular merozoites, consistent with a PV localization. Magnification, x 15000.Ruecker A, Shea M, Hackett F, Suarez C, Hirst EM, Milutinovic K, Withers-Martinez C, Blackman MJ. Proteolytic activation of the essential parasitophorous vacuole cysteine protease SERA6 accompanies malaria parasite egress from its host erythrocyte. J Biol Chem. 2012287(45):37949-63.
See original on MMP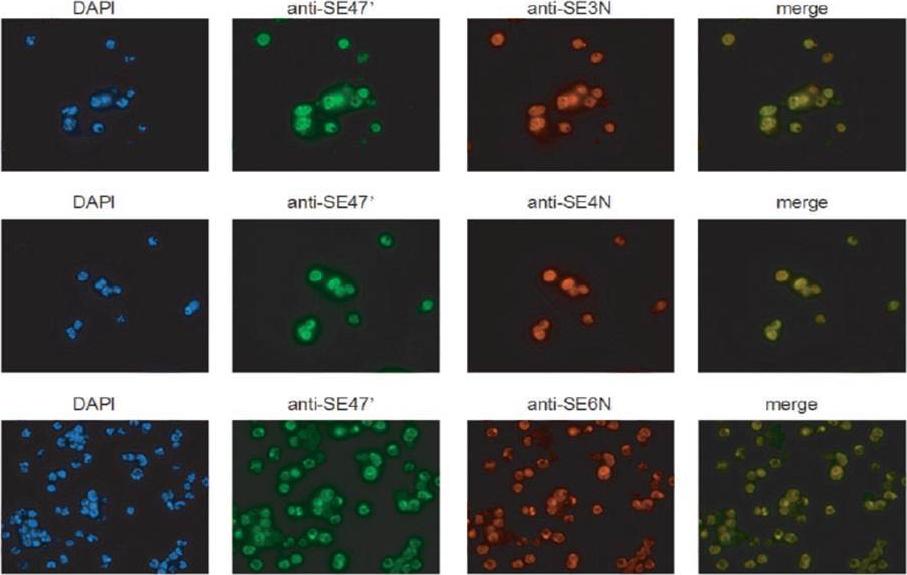
SERA3–6 genes are expressed in single parasites at similar localization. Trophozoite- and schizont (Honduras-1)-infected erythrocytes were purified by Percoll and subjected to immunofluorescence staining with rabbit anti-SE3N, -SE4N, or -SE6N antiserum and mouse anti-SE47’ (SERA5) IgG. The secondary antibodies used were Cy3-conjugated anti-rabbit IgG and fluorescein isothiocyanate-conjugated anti-mouse IgG. 4’,6’-Diamidino-2-phenylindole was also used to stain parasite nuclei.Aoki S, Li J, Itagaki S, Okech BA, Egwang TG, Matsuoka H, Palacpac NM, Mitamura T, Horii T. Serine repeat antigen (SERA5) is predominantly expressed among the SERA multigene family of Plasmodium falciparum, and the acquired antibody titers correlate with serum inhibition of the parasite growth. J Biol Chem. 2002 277:47533-40.
See original on MMP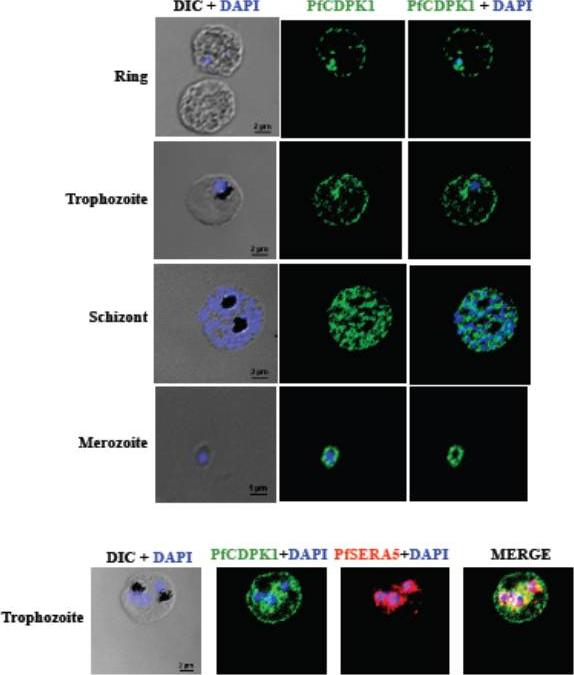
PfCDPK1 is expressed throughout erythrocytic schizogony as detected by by immunofluorescence assay (IFA). PfCDPK1 was detected in blood stages of P. falciparum by IFA using anti-PfCDPK1 mouse sera and Alexa Fluor 488-conjugated anti-mouse IgG goat sera (green). Anti-SERA5 rabbit sera and Alexa Fluor 594-conjugated anti-rabbit IgG goat sera (red) were used to localize the parasitophorous vacuole (PV) protein SERA5. Nuclear DNA was counterstained with DAPI (blue). Bright field (DIC) merged with DAPI (DIC + DAPI), fluorescence images detecting PfCDPK1 (PfCDPK1) and merged fluorescence images detecting PfCDPK1 and DAPI, PfSERA5 and DAPI or PfCDPK1, PfSERA5 and DAPI (Merge) are shown. Bar represents 2 mm (ring, trophozoite, schizont) or 1 μm (merozoite). PfCDPK1 localized to the rim of the infected erythrocyte and appeared to be associated with the erythrocyte membrane in rings and trophozoites (lower panel). In addition, PfCDPK1 was found in the parasitophorous vacuole (PV) and co-localized with SERA5, a known PV protein in trophozoites. Inmerozoites, PfCDPK1 was primarily found at the boundary of the merozoite at the merozoite plasma membraneBansal A, Singh S, More KR, Hans D, Nangalia K, Yogavel M, Sharma A, Chitnis CE. Characterization of Plasmodium falciparum calcium dependent protein kinase 1 (PfCDPK1) and its role in microneme secretion during erythrocyte invasion. J Biol Chem. 2013 288(3):1590-602.
See original on MMP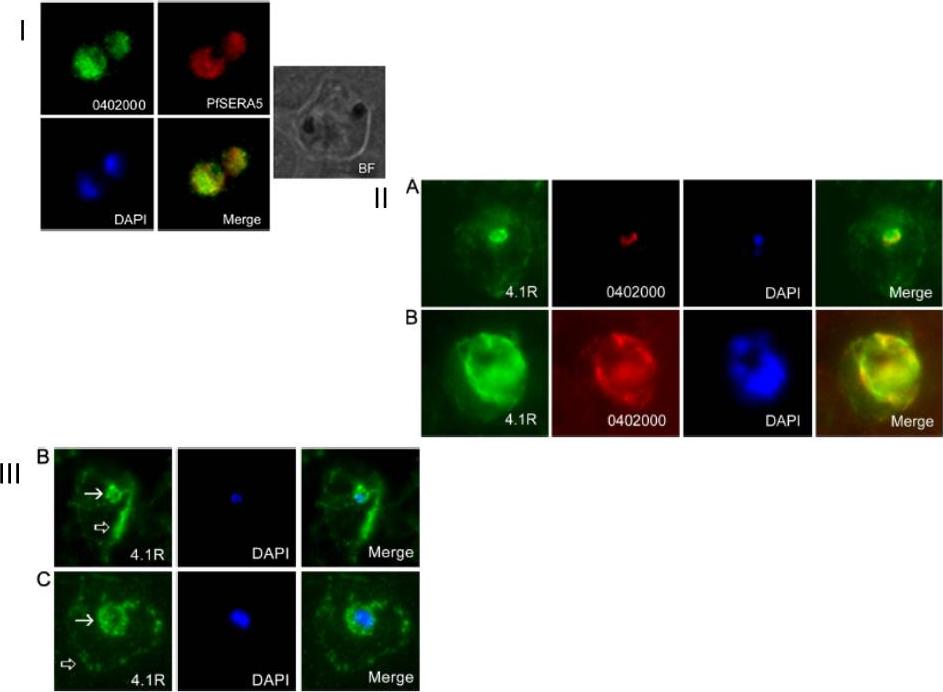
I. PFB0340c (PF3D7_0402000) localizes to the parasitophorous vacuole membrane. Fixed smears probed with rat anti-PF3D7_0402000 (0402000) and anti-PfSERA5 (SERA5) to test for co-localization (Merge). Parasite nuclei were identified with the nuclear stain DAPI, and infected erythrocytes imaged in brightfield (BF).II. PF3D7_0402000 colocalizes with a sub-population of 4.1R. Immunofluorescence microscopy of air-dried smears of 3D7 P. falciparum-infected erythrocytes probed with rabbit anti-4.1R (green), rat anti-PF3D7_0402000 (red) and the nuclear dye (DAPI). PF3D7_0402000 and 4.1R colocalize in early (A) and late (B) trophozoite stage parasites.III. A subpopulation of 4.1R is found at the PVM in Plasmodium falciparum-infected erythrocytes. Immunofluorescence microscopy of air-dried smears of 3D7 P. falciparum-infected erythrocytes probed with rabbit anti-4.1R (green). 4.1R consistently localizes near the erythrocyte plasma membrane(black arrows), but in early (B) and late (C) trophozoite-stage parasites, a subpopulation of 4.1R is found in close apposition to the parasite, asdetected by DAPI staining (blue).Parish LA, Mai DW, Jones ML, Kitson EL, Rayner JC. A member of the Plasmodium falciparum PHIST family binds to the erythrocyte cytoskeleton component band 4.1. Malar J. 2013 12(1):160.
See original on MMP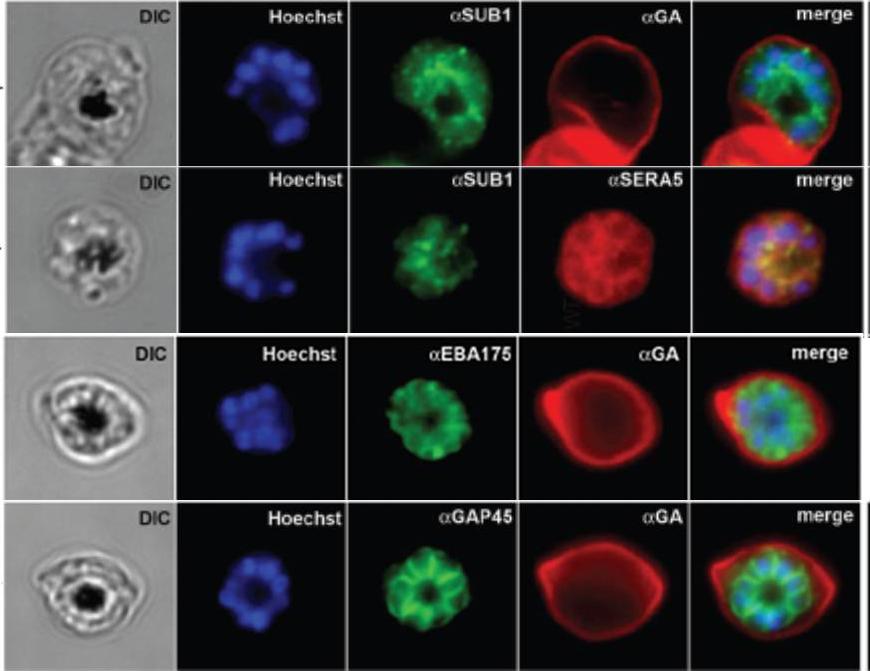
IFA of WT schizonts with glycophorin A (GA) labeled erythrocytes, localized SUB1 in an apical punctate pattern similar to SERA5. EBA-175 was detected in the apical ends of merozoites. GAP45 appeared submembrane. DIC, differential interference contrast; Hoechst = nucleic acid stain.Balu B, Maher SP, Pance A, Chauhan C, Naumov AV, Andrews RM, Ellis PD, Khan SM, Lin JW, Janse CJ, Rayner JC, Adams JH. CCR4-associated factor-1 coordinates expression of Plasmodium falciparum egress and invasion proteins. Eukaryot Cell. 2011 10(9):1257-63
See original on MMP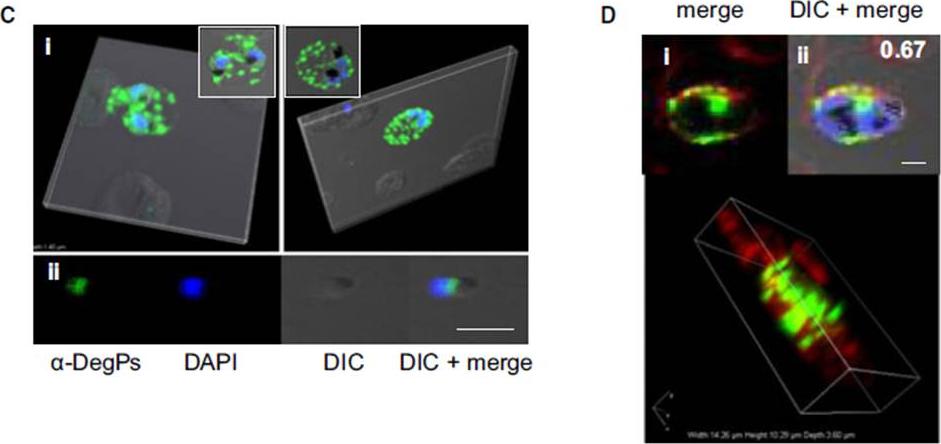
Indirect immunofluorescence assay and confocal microscopy of late stages of P. falciparum-infected erythrocytes and free merozoites. P. falciparum parasites were probed using rat anti-DegP antibody followed by fluorescein isothiocyanate-conjugated anti-rat IgG (green). Other marker proteins were stained with the rabbit antiserum/ antibody followed by Cy3-conjugated anti-rabbit IgG (red). The parasite nuclei were counterstained with 4′,6-diamidino-2-phenylindole (DAPI) (blue), and slides were visualized using a confocal laser scanning microscope. z-section of late-stage parasites stained for DegP showed green fluorescence signals in the cytosol of the parasite and the cytosol and membrane of infected RBCs (Ci). DegP signals were observed on free merozoites (Cii). The surface localization of PfDegP in infected RBCs was confirmed by co-localization with an erythrocyte membrane marker (red) (Di). Yellow spots indicate merged fluorescent signals from DegP and band 3 antibodies (Dii). SERA5 and MIF were used as markers for parasitophorous vacuole and secretory protein in the culture supernatant, respectively. Sharma S, Jadli M, Singh A, Arora K, Malhotra P. A secretory multifunctional serine protease, DegP of Plasmodium falciparum, plays an important role in thermo-oxidative stress, parasite growth and development. FEBS J. 2014 Mar;281(6):1679-99.
See original on MMP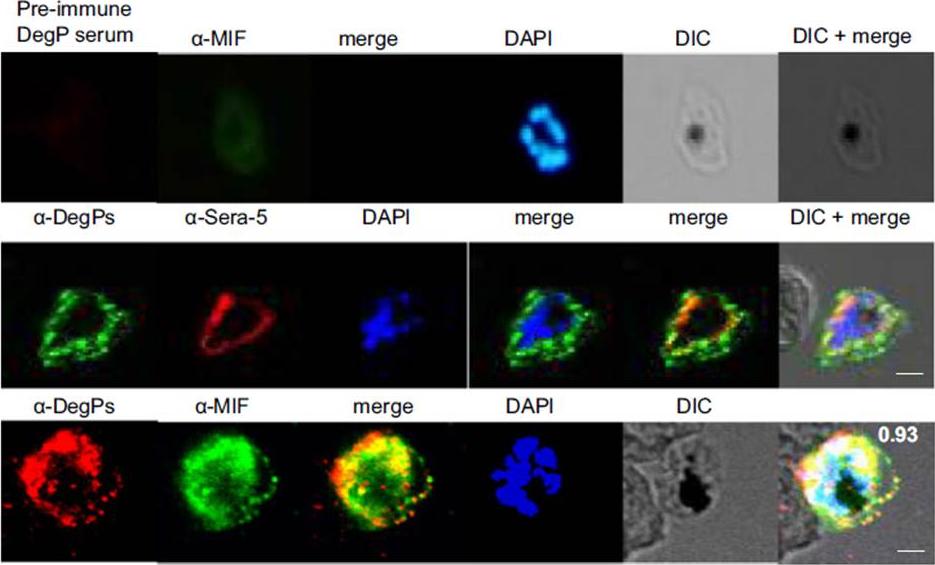
Indirect immunofluorescence assay and confocal microscopy of late stages of P. falciparum-infected erythrocytes and free merozoites. P. falciparum parasites were probed using rat anti-DegP antibody followed by fluorescein isothiocyanate-conjugated anti-rat IgG (green). Other marker proteins were stained with the rabbit antiserum/antibody followed by Cy3-conjugated anti-rabbit IgG (red). PfSera5 and PfMIF were used as markers for parasitophorous vacuole and secretory protein in the culture supernatant, respectively. Significant merging of the fluorescent signal for DegP was observed with SERA5 and MIF signals, indicating the presence of DegP in the cytosol of infected RBCs.Sharma S, Jadli M, Singh A, Arora K, Malhotra P. A secretory multifunctional serine protease, DegP of Plasmodium falciparum, plays an important role in thermo-oxidative stress, parasite growth and development. FEBS J. 2014 281(6):1679-99.
See original on MMP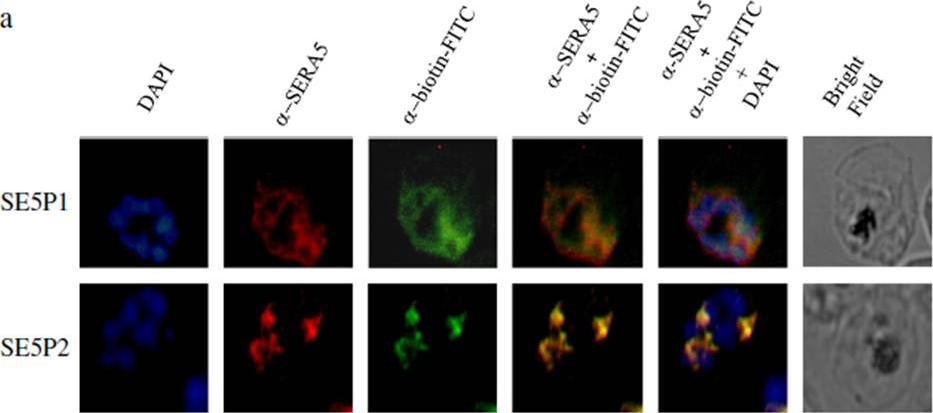
Localization of SE5 P1 and SE5 P2 peptides with PfSERA5. a) The biotinylated peptides SE5 P1 and SE5 P2 were allowed to incubate with the mature blood stage parasites. The parasite was then probed for biotinylated peptides (green) and PfSERA5 (red). Parasite nuclei were stained with DAPI, a nuclear dye (blue). On superimposing, the areas of colocalization were seen as yellow. SE5 P1 and SE5 P2 were seen to be colocalized with the PfSERA5 within the parasitophorous vacuole.Kanodia S, Kumar G, Rizzi L, Pedretti A, Hodder AN, Romeo S, Malhotra P. Synthetic peptides derived from the C-terminal 6kDa region of Plasmodium falciparum SERA5 inhibit the enzyme activity and malaria parasite development. Biochim Biophys Acta. 2014 25;1840(9):2765-2775. PMID:
See original on MMP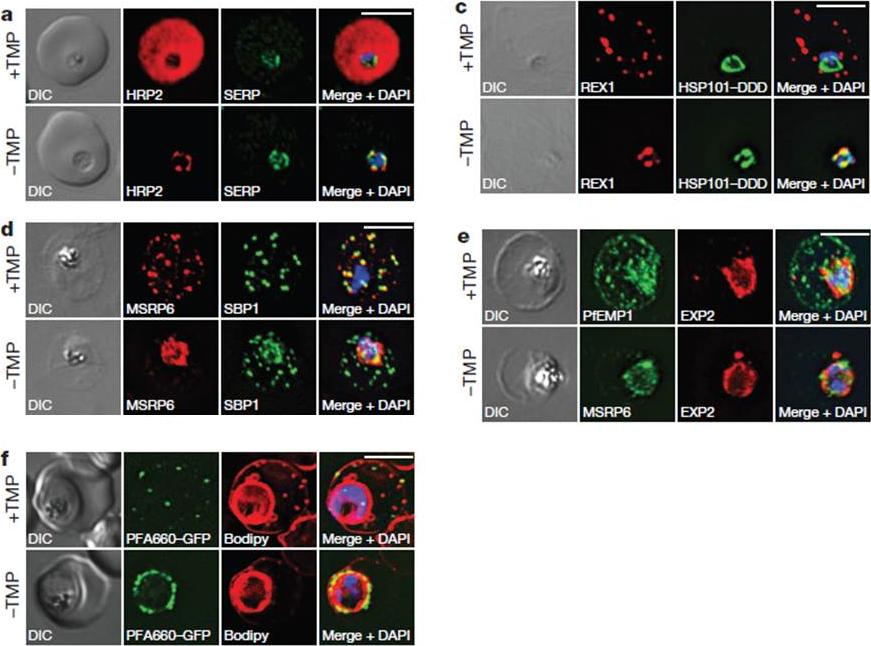
HSP101 is required for export of PEXEL and PNEP proteins. a, c, Immunofluorescence assay (IFA) of ring-stage 13F10 parasites with orwithout TMP (DDD-stabilizing small molecule trimethoprim). TMP was removed in late schizont stage and parasites were allowed to reinvade and grow 18–24 h before fixation with paraformaldehyde (a) or acetone (c). a, IFA of the exported PEXEL-containing protein HRP2. c, IFAs of the PNEP REX1, which colocalizes with HSP101DDD at the PVM in the absence of TMP. d, e, IFA of trophozoite-stage 13F10 parasites with or withoutTMP. TMP was removed in late ring stage and parasites were allowed to develop 12–24 h before fixation with acetone. f, Live fluorescence imaging of 13F10 parasites expressing a PFA660–GFP fusion and labelled with Bodipy TR Ceramide to demarcate the PVM (other membranes are also labelled). TMP treatment as in d, e. All scale bars, 5 mm. Images in c–f are representative of two independent experiments.Beck JR, Muralidharan V, Oksman A, Goldberg DE. PTEX component HSP101 mediates export of diverse malaria effectors into host erythrocytes. Nature. 2014 Jul 16. [Epub ahead of print]
See original on MMP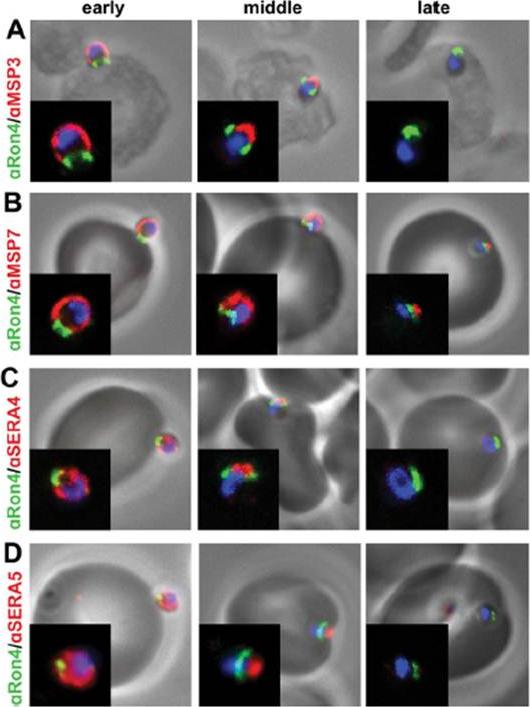
Peripherally associated proteins MSP3, MSP7, SERA4, and SERA5 are shed during invasion. Invading merozoites were labeled with PfRON4 as a marker of the tight junction (green) and colabeled with antibodies directed against MSP3 (A), MSP7 (B), SERA4 (C), or SERA5 (D). All proteins were shed at the point of tight junction, with antibody reactivity to the merozoite surface being observed only on the proportion of the merozoite external to the RBC. All primary antibodies were used at a 1:100 serum dilution, and secondary antibodies were used at a 1:500 dilution.Boyle MJ, Langer C, Chan JA, Hodder AN, Coppel RL, Anders RF, Beeson JG. Sequential processing of merozoite surface proteins during and after erythrocyte invasion by Plasmodium falciparum. Infect Immun. 2014 82(3):924-36.
See original on MMP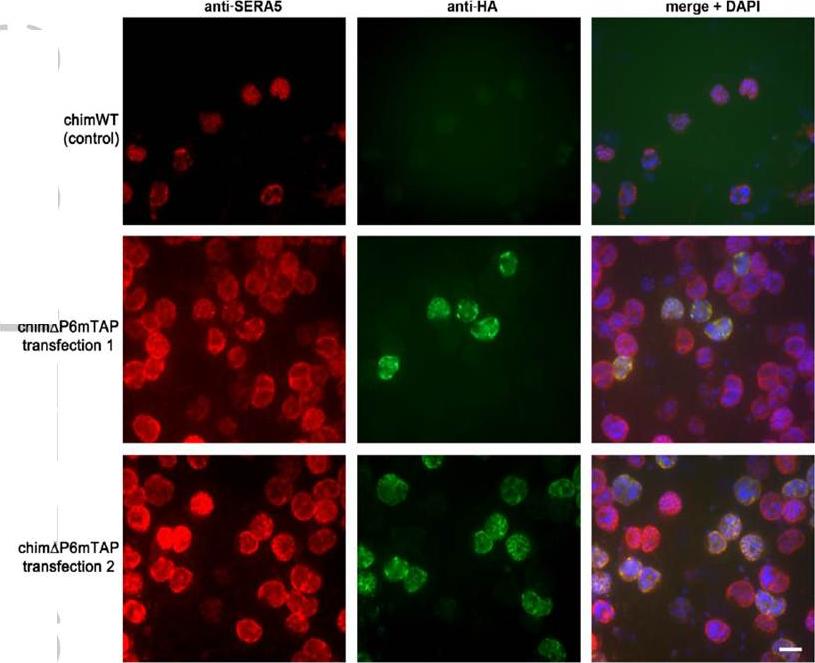
The P6 segment of SERA5 can be readily replaced with a tandem affinity-purification tag. IFA of uncloned drug-resistant parasite lines examined just one cycle of drug selection following transfection with control construct pHH1SERA5chimWT (top row), designed to simply reconstitute the wild-type SERA5 coding sequence, or with pHH1SERA5chimDP6mTAP (middle and bottom row), designed to introduce a mini TAP tag into the SERA5 coding sequence in place of the region encompassed by the P6 region (see Supplementary Fig. S8). Fixed parasites were probed with a polyclonal rabbit anti-SERA5 antibody (red) or with the anti-HA3 mAb 12CA5 (green). A significant proportion of the pHH1SERA5chimDP6mTAP-transfected parasites exhibit an HA3-specific signal, indicating rapid integration of the transfection construct. Scale bar, 5 mm. Examination of uncloned drug-resistant cultures by IFA using anti-HA3 antibodies showed a strong positive signal in up to ~20% of schizonts, which co-localised with the anti-SERA5 signal, indicating efficient integration of the input construct in the predicted manner.Stallmach R, Kavishwar M, Withers-Martinez C, Hackett F, Collins CR, Howell SA, Yeoh S, Knuepfer E, Atid AJ, Holder AA, Blackman MJ. Plasmodium falciparum SERA5 plays a non-enzymatic role in the malarial asexual blood-stage lifecycle. Mol Microbiol. 2015 Jan 20. [Epub ahead of print]
See original on MMP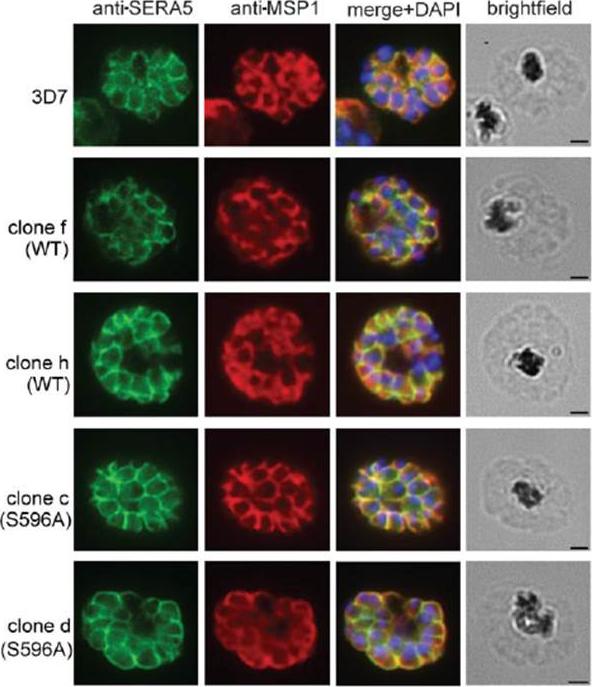
IFA images of schizonts of the indicated transgenic and parental 3D7 clones, probed with a polyclonal rabbit antiserum raised against full-length rSERA5wt (green) or the anti-MSP1 monoclonal antibody (mAb) 89.1 (red). The merged images include the fluorescence signal of the nuclear stain DAPI. No morphological differences were evident between the parental and transgenic parasites. Scale bar, 1 mm. IFA analysis showed normal expression and distribution of SERA5 in all clones.Stallmach R, Kavishwar M, Withers-Martinez C, Hackett F, Collins CR, Howell SA, Yeoh S, Knuepfer E, Atid AJ, Holder AA, Blackman MJ. Plasmodium falciparum SERA5 plays a non-enzymatic role in the malarial asexual blood-stage lifecycle. Mol Microbiol. 2015 Jan 20. [Epub ahead of print]
See original on MMP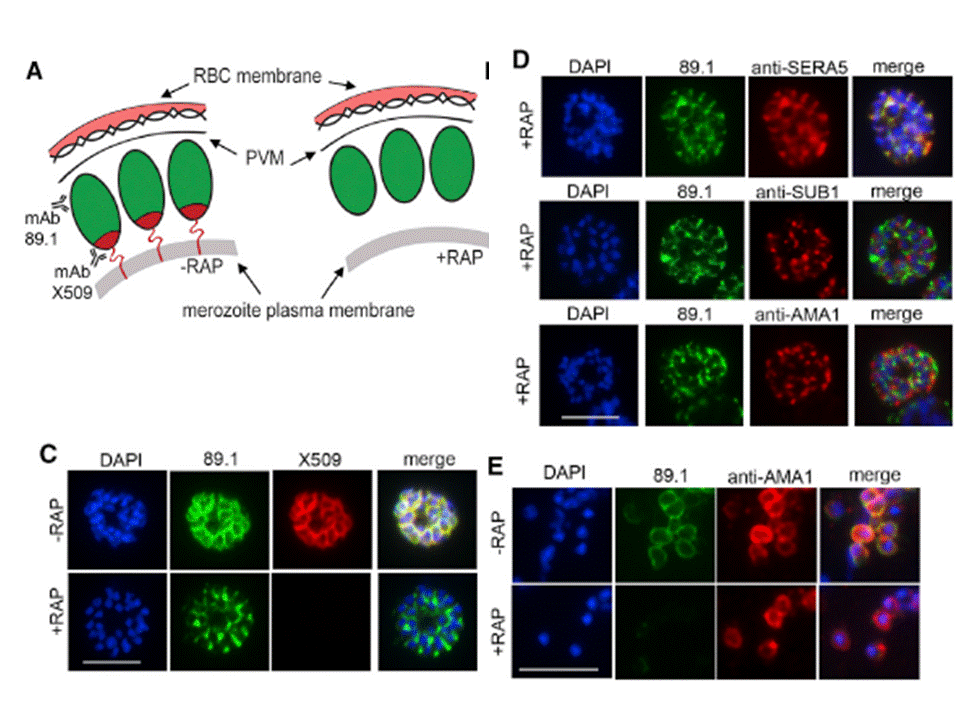
Truncation of MSP1 Produces anEgress Defect (A) Predicted RAP-induced MSP1 truncation in the 3D7MSP1flox42C clones, showing loss of the GPI anchor and C-terminal domain containing the mAb X509 epitope. (C) RAP treatment produces a loss of mAb X509 reactivity and a shift in the IFA pattern of MSP1 to one typical of PV proteins, consistent with the predicted truncation. Numbers of DAPI-stained nuclei did not differ between control and RAPtreated. (D) IFA showing co-localization of truncated MSP1 with SERA5 indicating a PV location. The punctate localization of SUB1 and the microneme protein AMA1 indicates normal organelle biogenesis. (E) IFA showing lack of surface-bound MSP1 on merozoites of RAP-treated 3D7MSP1flox42C1 clone E3. Antibodies to AMA1 (which is expressed on free merozoites) were used as a control.Das S, Hertrich N, Perrin AJ, Withers-Martinez C, Collins CR, Jones ML, Watermeyer JM, Fobes ET, Martin SR, Saibil HR, Wright GJ, Treeck M, Epp C, Blackman MJ. Processing of Plasmodium falciparum Merozoite Surface Protein MSP1 Activates a Spectrin-Binding Function Enabling Parasite Egress from RBCs. Cell Host Microbe. 2015 18(4):433-44.
See original on MMP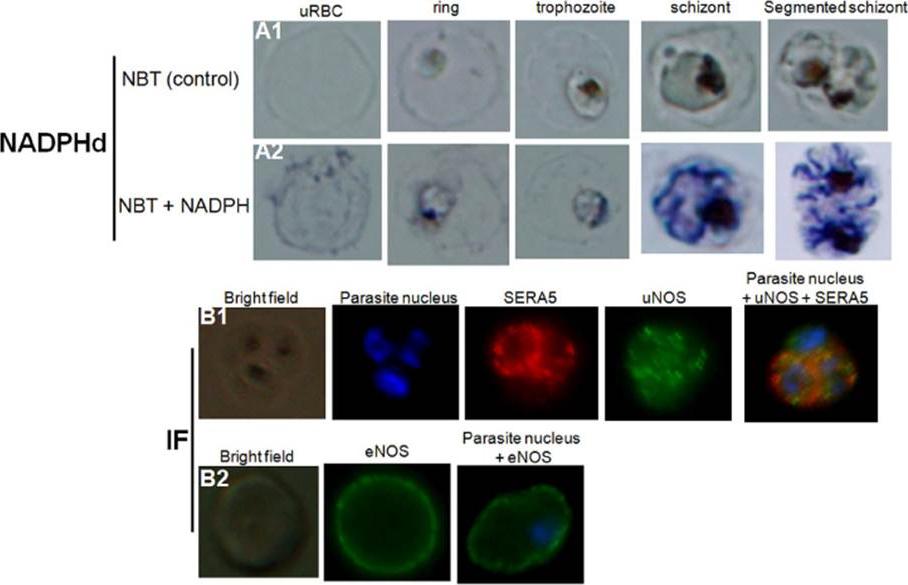
Detection of nitric oxide synthase in both healthy and P. falciparum-harboring red blood cells. NADPH-diaphorase activity within healthy RBCs or PfRBCs at different stages of parasite development (A). The panel (A1) represents the negative control (uRBCs or PfRBCs incubated with NBT without NADPH). NADPHd staining (blue color) was observed in both uRBC and parasite (A2). Identification by immunofluorescence assay (IF) of NOS within uRBCs or PfRBCs using rabbit polyclonal antibody against uNOS (B1, green) or mouse monoclonal antibody against eNOS (B2, green). To differentiate infected from uninfected RBCs, parasite nucleus was labeled in blue with Hoechst 33342. A monoclonal antibody against parasite SERA5 was used to localize the parasitophorous vacuole (red).Rey J, Buffet PA, Ciceron L, Milon G, Mercereau-Puijalon O, Safeukui I. Reduced erythrocyte deformability associated with hypoargininemia during Plasmodium falciparum malaria. Sci Rep. 2014 Jan 20;4:3767
See original on MMP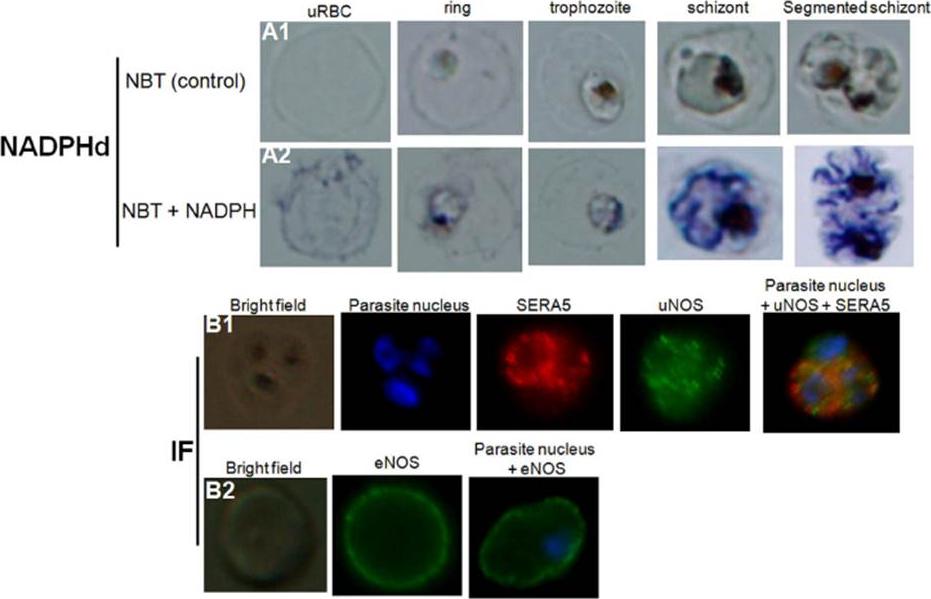
Detection of nitric oxide synthase in both healthy and P. falciparum-harboring red blood cells. NADPH-diaphorase activity within healthy RBCs or PfRBCs at different stages of parasite development (A). The panel (A1) represents the negative control (uRBCs or PfRBCs incubated with NBT without NADPH). NADPHd staining (blue color) was observed in both uRBC and parasite (A2). Identification by immunofluorescence assay (IF) of NOS within uRBCs or PfRBCs using rabbit polyclonal antibody against uNOS (B1, green) or mouse monoclonal antibody against eNOS (B2, green). To differentiate infected from uninfected RBCs, parasite nucleus was labeled in blue with Hoechst 33342. A monoclonal antibody against parasiteSERA5 was used to localize the parasitophorous vacuole (red).Rey J, Buffet PA, Ciceron L, Milon G, Mercereau-Puijalon O, Safeukui I. Reduced erythrocyte deformability associated with hypoargininemia during Plasmodium falciparum malaria. Sci Rep. 2014 Jan 20;4:3767.
See original on MMP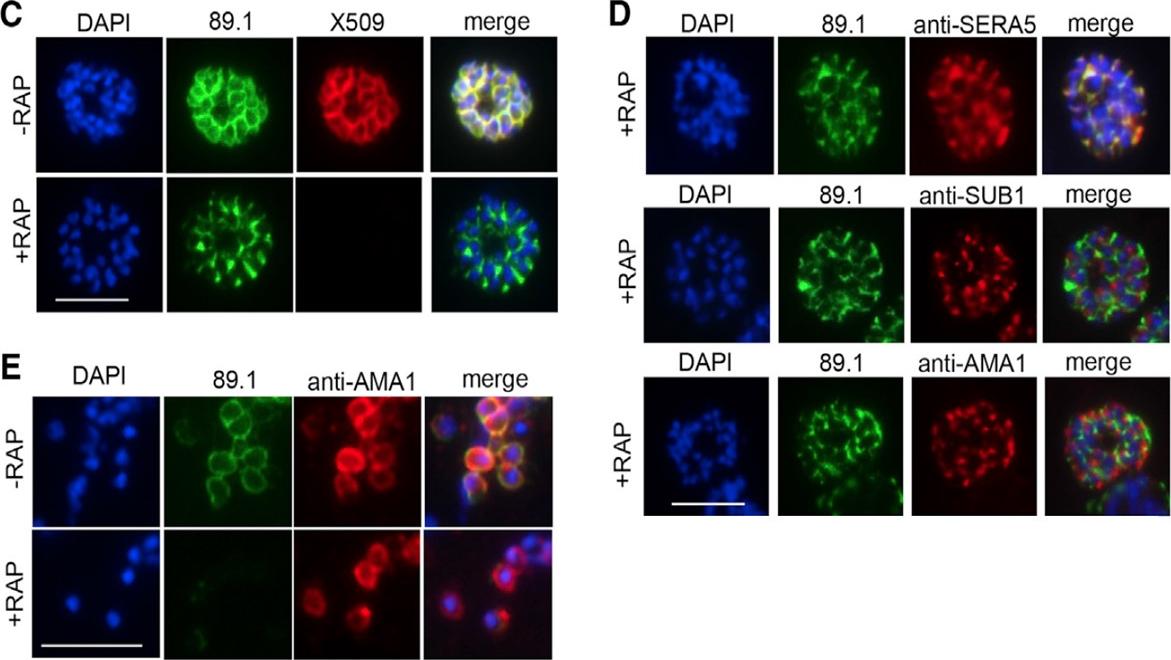
Truncation of MSP1 Produces an Egress Defect. (C) RAP treatment produces a loss of mAb X509 reactivity and a shift in the IFA pattern of MSP1 to one typical of PV proteins, consistent with the predicted truncation. Numbers of DAPI-stained nuclei did not differ between control and RAP treated schizonts (mean values: 21.2 ± 3.4 and 20.6 ± 4.0 nuclei per schizont, respectively, n = 24). (D) IFA showing co-localization of truncated MSP1 with SERA5 indicating a PV location. The punctate localization of SUB1 and the microneme protein AMA1 indicates normal organelle biogenesis. (E) IFA showing lack of surface-bound MSP1 on merozoites of RAP-treated 3D7MSP1flox42C1 clone E3. Antibodies to AMA1 (which is expressed on free merozoites) were used as a control. Analysis of RAP-treated 3D7MSP1flox42C parasites showed highly efficient excision, resulting in exclusive expression of truncated MSP1 in mature schizonts at the end of the same erythrocytic cycle (C). No effects on merozoite development were discernible. The modified MSP1 was trafficked to the PV as expected for a non-membrane bound merozoite surface protein (C–D) but was not present on the surface of free merozoites (E)Das S, Hertrich N, Perrin AJ, Withers-Martinez C, Collins CR, Jones ML, Watermeyer JM, Fobes ET, Martin SR, Saibil HR, Wright GJ, Treeck M, Epp C, Blackman MJ. Processing of Plasmodium falciparum Merozoite Surface Protein MSP1 Activates a Spectrin-Binding Function Enabling Parasite Egress from RBCs. Cell Host Microbe. 2015 18(4):433-44. PMID: 26468747
See original on MMP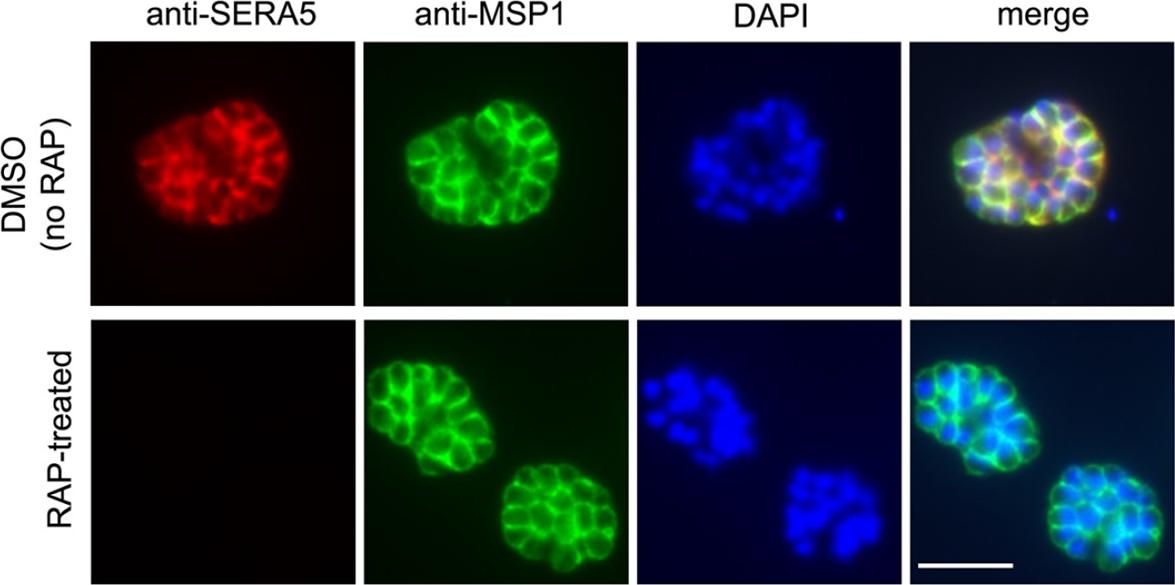
To establish the effects of SERA5 loss on parasite growth, we compared the replication rates of mock-treated and RAP-treated floxSERA5-1B6 and floxSERA5-3B6 clones, using the parental 1G5DC clone as a controlIFA of mature schizonts of control (DMSO-treated) and RAP-treated integrant clone floxSERA5-1B6 ~44 h following treatment. No SERA5-specific signal was detectable in the majority (~98%) of the rapamycin-treated population. Scale bar, 5 μmCollins CR, Hackett F, Atid J, Tan MSY, Blackman MJ. The Plasmodium falciparum pseudoprotease SERA5 regulates the kinetics and efficiency of malaria parasite egress from host erythrocytes. PLoS Pathog. 2017 13(7):e1006453.
See original on MMP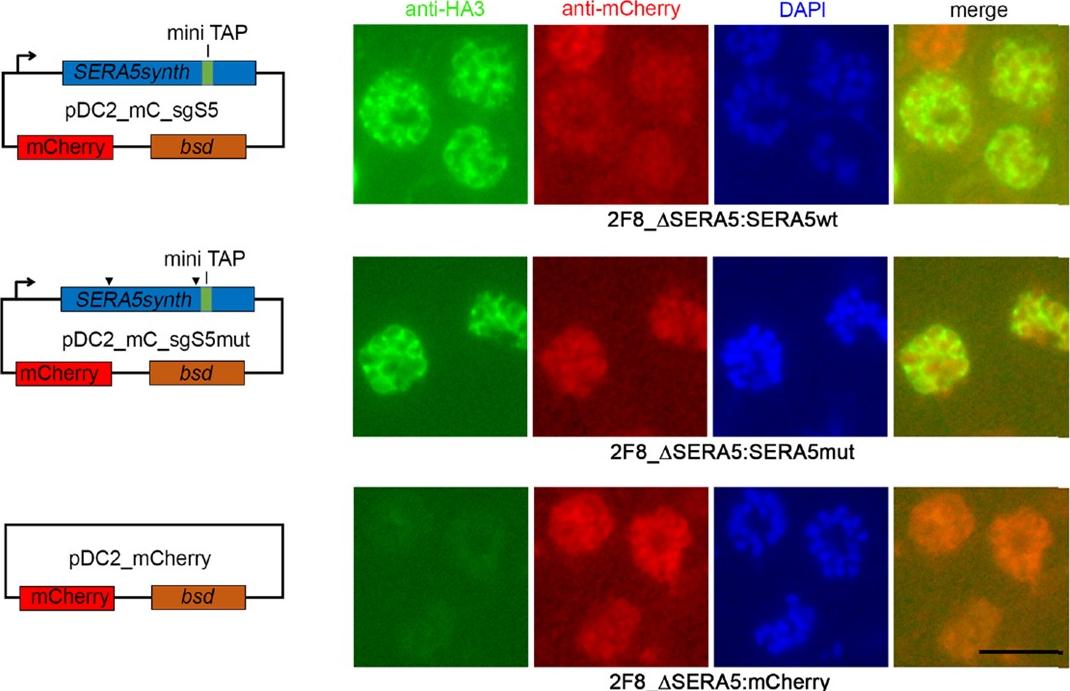
DiCre-mediated conditional disruption of P. falciparum SERA5 expression. IFA of mature schizonts of control (DMSO-treated) and RAP-treated integrant clone floxSERA5-1B6 ~44 h following treatment. No SERA5-specific signal was detectable in the majority (~98%) of the RAP-treated population. Scale bar, 5 μm. To assess the effects of SERA5 gene deletion on SERA5 protein expression, mature flox-SERA5-1B6 and floxSERA5-3B6 schizonts were examined by indirect immunofluorescence (IFA) at the end of the first erythrocytic cycle (~44 h) following RAP-treatment. No SERA5-specific signal was detectable by IFA in most of the RAP-treated floxSERA5-1B6 and flox-SERA5-3B6 schizontsCollins CR, Hackett F, Atid J, Tan MSY, Blackman MJ. The Plasmodium falciparum pseudoprotease SERA5 regulates the kinetics and efficiency of malaria parasite egress from host erythrocytes. PLoS Pathog. 2017 Jul 6;13(7):e1006453. PMID: 28683142
See original on MMP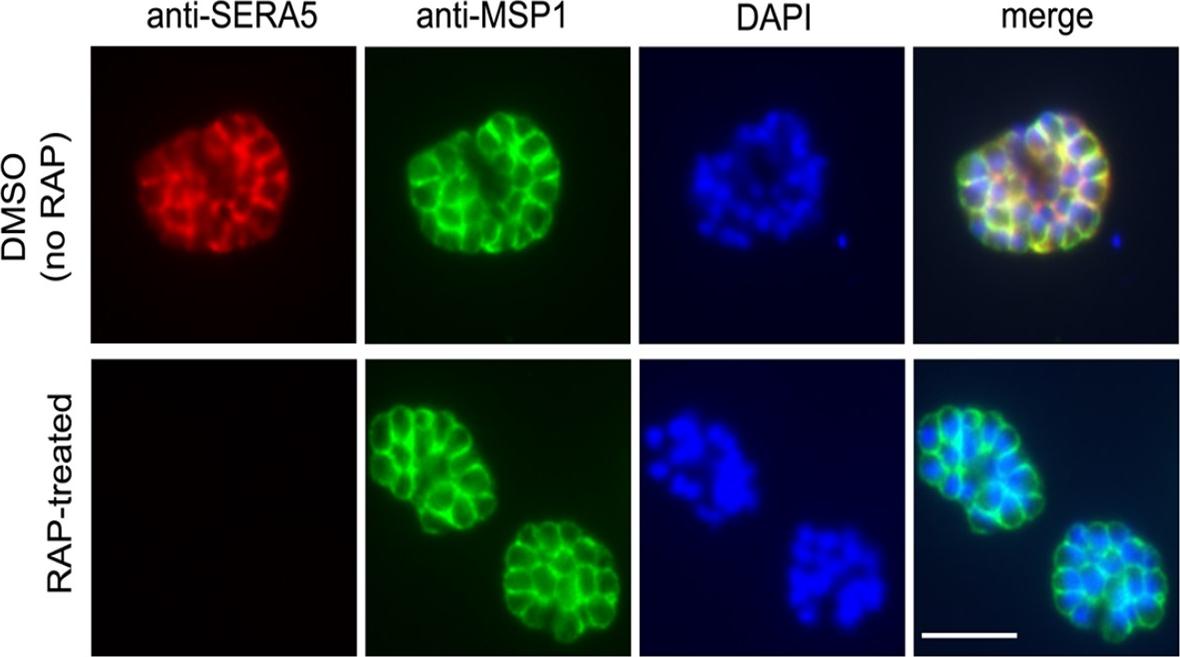
DiCre-mediated conditional disruption of P. falciparum SERA5 expression. IFA of mature schizonts of control (DMSO-treated) and RAP-treated integrant clone floxSERA5-1B6 ~44 h following treatment. No SERA5-specific signal was detectable in the majority (~98%) of the RAP-treated population. Scale bar, 5 μm. To assess the effects of SERA5 gene deletion on SERA5 protein expression, mature flox-SERA5-1B6 and floxSERA5-3B6 schizonts were examined by indirect immunofluorescence (IFA) at the end of the first erythrocytic cycle (~44 h) following RAP-treatment. No SERA5-specific signal was detectable by IFA in most of the RAP-treated floxSERA5-1B6 and flox-SERA5-3B6 schizontsCollins CR, Hackett F, Atid J, Tan MSY, Blackman MJ. The Plasmodium falciparum pseudoprotease SERA5 regulates the kinetics and efficiency of malaria parasite egress from host erythrocytes. PLoS Pathog. 2017 Jul 6;13(7):e1006453.
See original on MMP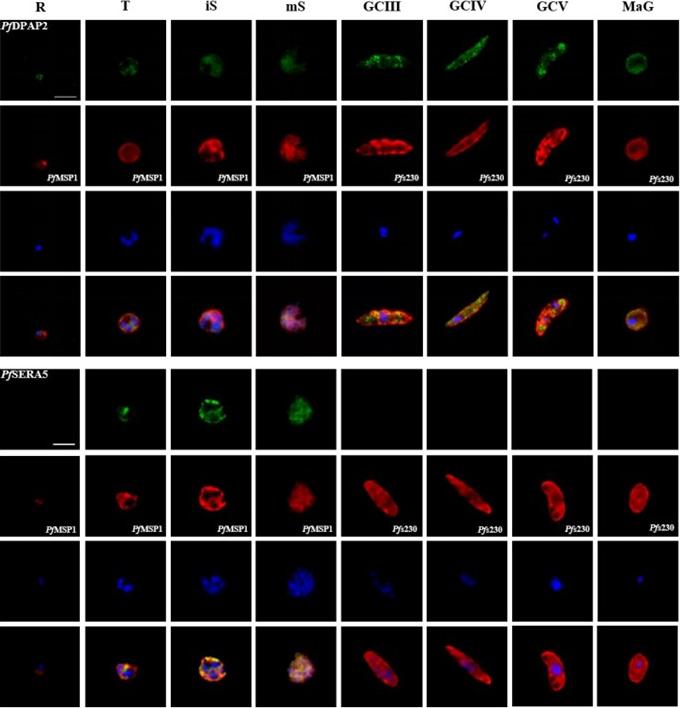
Protein expression profile of selected P. falciparum proteases. IFAs using polyclonal mouse antisera directed against 12 of the proteases was used to demonstrate protein expression in the asexual blood stages (R, ring; T, trophozoite; iS, immature schizont; mS, mature schizont) and gametocytes (GC stages III, IV and V) as well as in macrogametes (MaG) at 20 min post-activation (in green). The asexual and the sexual blood sstages were visualized via immunolabelling with polyclonal rabbit antisera against PfMSP1 and Pfs230, respectively (in red). Hoechst 33342 staining was used to highlight the parasite nuclei (in blue). Bar, 5 μm. Results are representative of three to four independent experiments.Weißbach T, Golzmann A, Bennink S, Pradel G, Julius Ngwa C. Transcript and protein expression analysis of proteases in the blood stages of Plasmodium falciparum. Exp Parasitol. 2017 Mar 25. [Epub ahead of print]
See original on MMP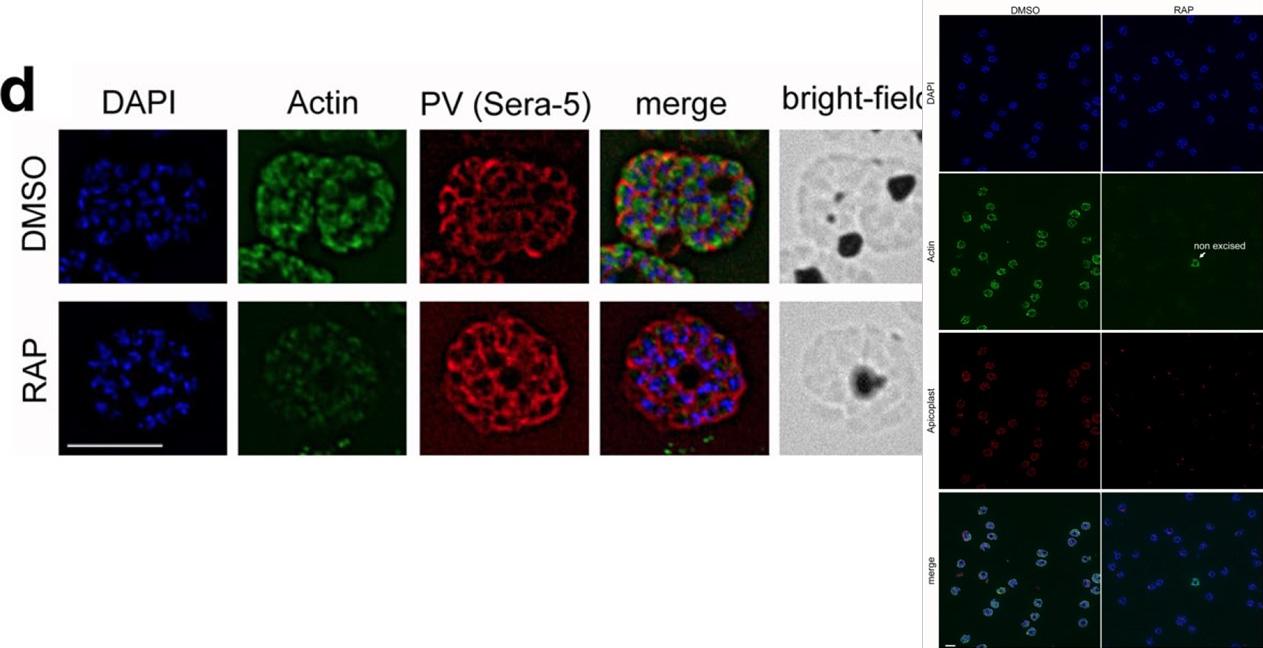
IFA shows a loss of reactivity of PfACT1 KOs to the anti-PfACT1 antibody. The PV is depicted in red (Sera-5). Scale bar 5 μ. PfACT1 disruption was apparent by immunofluorescence assay (IFA) in schizonts 44 h post-induction. Based on IFAs, it was estimated that ~98% of the population had undergone excision of pfact1, resulting in an almost pure population of pfact1 disrupted parasites (PfACT1 KO) for phenotypic analysis. Only a weak, potentially non-specific signal could be detected by IFA in PfACT1 KO schizonts.Das S, Lemgruber L, Tay CL, Baum J, Meissner M. Multiple essential functions of Plasmodium falciparum actin-1 during malaria blood-stage development. BMC Biol. 2017 Aug 15;15(1):70.
See original on MMPMore information
| PlasmoDB | PF3D7_0207600 |
| GeneDB | PF3D7_0207600 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | PF02_0072, PFB0340c |
| Orthologs | PBANKA_0304800 , PBANKA_0304900 , PBANKA_0305000 , PBANKA_0305100 , PCHAS_0307000 , PCHAS_0307100 , PCHAS_0307200 , PCHAS_0307300 , PKNH_0413100 , PKNH_0413200 , PKNH_0413400 , PKNH_0413600 , PKNH_0413700 , PVP01_0416800 , PVP01_0416900 , PVP01_0417000 , PVP01_0417100 , PVP01_0417200 , PVP01_0417300 , PVP01_0417400 , PVP01_0417600 , PVP01_0417700 , PVP01_0417800 , PVP01_0417900 , PVX_003795 , PVX_003800 , PVX_003805 , PVX_003810 , PVX_003820 , PVX_003825 , PVX_003830 , PVX_003835 , PVX_003840 , PVX_003845 , PVX_003850 , PY17X_0305600 , PY17X_0305700 |
| Google Scholar | Search for all mentions of this gene |