PCHAS_1030900 actin II, putative
Disruptability [+]
| Species | Disruptability | Reference | Submitter | |
|---|---|---|---|---|
| P. berghei ANKA |
Possible |
RMgm-632 | Imported from RMgmDB | |
| P. berghei ANKA |
Possible |
PlasmoGEM (Barseq) | PlasmoGEM | |
| P. berghei ANKA |
Possible |
RMgm-5277 | Imported from RMgmDB | |
| P. falciparum 3D7 |
Possible |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen | |
Mutant phenotypes [+]
| Species | Stage | Phenotype | Reference | Submitter |
|---|---|---|---|---|
| P. berghei ANKA | Asexual |
No difference |
RMgm-632 | Imported from RMgmDB |
| P. berghei ANKA | Asexual |
No difference |
PlasmoGEM (Barseq) | PlasmoGEM |
| P. berghei ANKA | Asexual |
No difference |
RMgm-5277 | Imported from RMgmDB |
| P. berghei ANKA | Gametocyte |
Difference from wild-type |
RMgm-632
Production of normal numbers of male and female gametocytes. Male gametocytes show a strong reduction in gamete formation (exflagellation) |
Imported from RMgmDB |
| P. berghei ANKA | Gametocyte |
Difference from wild-type |
RMgm-5277
Gametocyte conversion rates, male:female ratios, and exflagellation rates of act2::mCherry were comparable to those of wild type parasites. In contrast, male exflagellation of act2::mCherry was completely abrogated, reproducing the act2phenotype.Ring and trophozoite stage parasites showed virtually undetectable levels of mCherry expression. mCherry expression is considerably higher in male gametocytes compared to female gametocytes. |
Imported from RMgmDB |
| P. berghei ANKA | Ookinete |
Difference from wild-type |
RMgm-632
Production of normal numbers of male and female gametocytes. Male gametocytes show a strong reduction in gamete formation (exflagellation) and ookinete formation was reduced ~50 fold compared to wild type. |
Imported from RMgmDB |
| P. berghei ANKA | Oocyst |
Difference from wild-type |
RMgm-632
Male gametocytes show a strong reduction in gamete formation (exflagellation) and ookinete formation is reduced ~50 fold compared to wild type. No oocysts are formed in Anopheles gambiae mosquitoes. |
Imported from RMgmDB |
| P. berghei ANKA | Sporozoite |
Difference from wild-type |
RMgm-632
Ookinete formation is reduced ~50 fold compared to wild type and no oocysts are formed in Anopheles gambiae mosquitoes. |
Imported from RMgmDB |
Imaging data (from Malaria Metabolic Pathways)
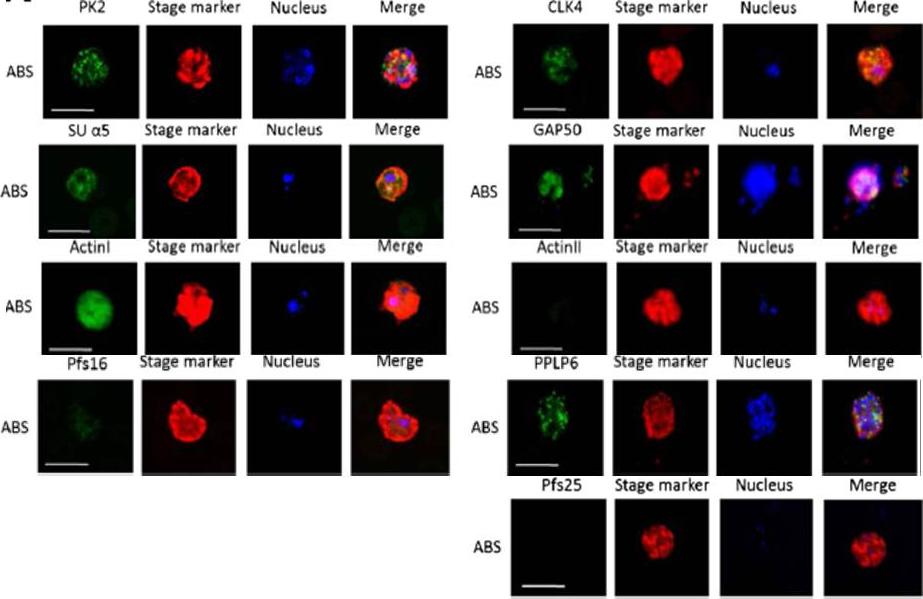
Immunofluorescence assays, using specific antibodies, detected the proteins of interest (in green) in asexual blood stage (ABS) parasites, using MSP1 (in red). Nuclei were highlighted by Hoechst nuclear stain (in blue). Bar, 5 μm. Ngwa CJ, Scheuermayer M, Mair GR, Kern S, Brügl T, Wirth CC, Aminake MN, Wiesner J, Fischer R, Vilcinskas A, Pradel G. Changes in the transcriptome of the malaria parasite Plasmodium falciparum during the initial phase of transmission from the human to the mosquito. BMC Genomics. 2013 Apr 15;14:256
See original on MMP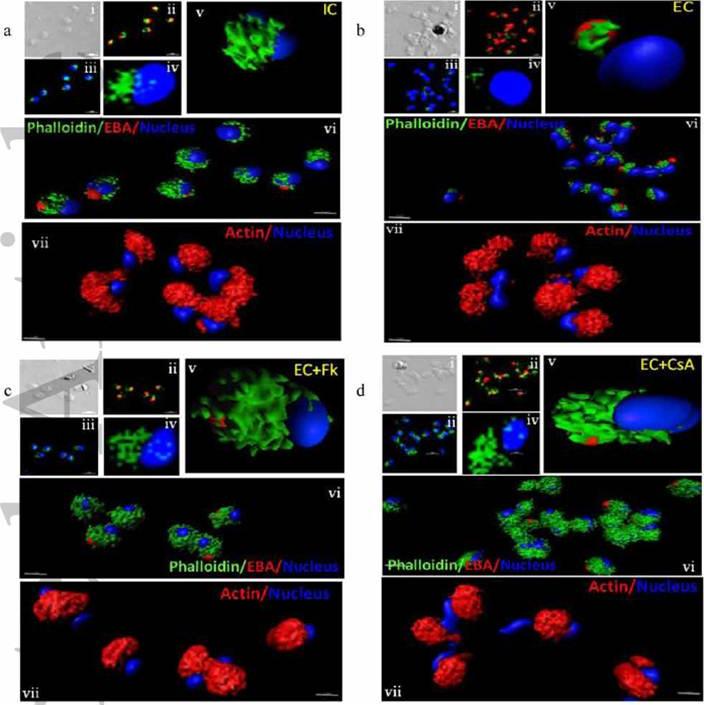
Actin polymerization status in P. falciparum merozoites. Merozoites were stained with Alexa 488-labeled phalloidin to detect polymerized F-actin (green), mouse anti EBA-175 mouse sera and Alexa Fluor 594-conjugated anti-mouse IgG goat antibodies to define location of micronemes (red) and DAPI to define location of nuclei (blue). Slides were observed under a confocal laser microscope (Nikon A1R). Bright field (i) and single slice confocal fluorescence images of merozoites (ii – iv) are shown. Three-dimensional reconstruction of confocal z stack fluorescence images of merozoites was performed using Imaris software (v –vii). Total actin (polymerized F-actin and monomeric G-actin) was detected with anti-actin rabbit sera followed by Alexa Fluor 594-conjugated anti-rabbit IgG goat antibodies (red in vii). Levels of total actin are similar in all conditions tested. Transfer of merozoites from IC to EC buffer leads to disassembly of polymerized F-actin at the apical tip of merozoites. Treatment of merozoites with FK506 and CsA prior to transfer to EC buffer leads to accumulation of polymerized actin at the apical end. Actin depolymerizing agent CytD and actin stabilizing agent JAS were used as control and result in reduced F-actin and increased F-actin at the apical tip of the merozoites respectively. Scale bar represents 1 μm. Treatment of merozoites with actin depolymerizing agents, CytD, ML-B and LA-B, resulted in increased secretion of microneme protein PfAMA-1 (a-c) whereas treatment with polymerized actin stabilizer JAS resulted in reduced secretion of PfAMA-1 (d).Singh S, More KR, Chitnis CE. Role of Calcineurin and Actin Dynamics in Regulated Secretion of Microneme Proteins in Plasmodium falciparum Merozoites during Erythrocyte Invasion. Cell Microbiol. 2013 Jun;21(2):125-7
See original on MMP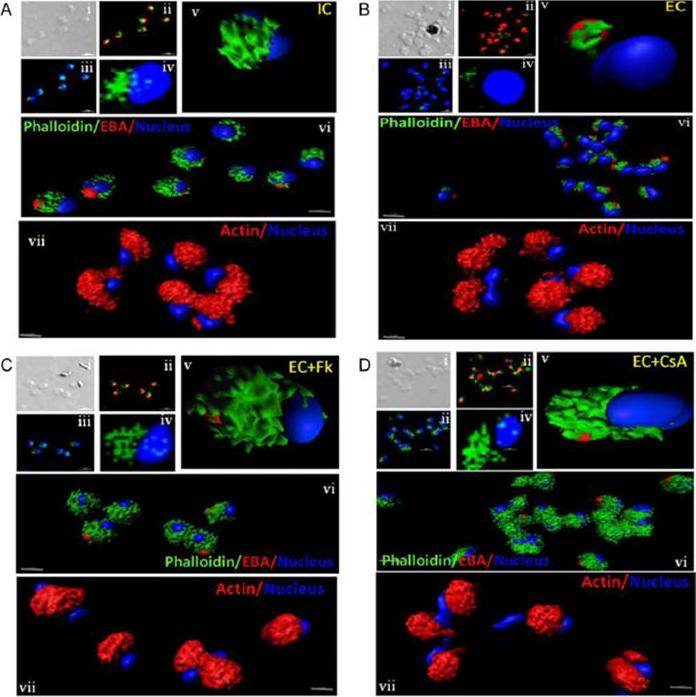
Actin polymerization status in P. falciparum merozoites. Merozoites were stained with Alexa 488-labeled phalloidin to detect polymerized F-actin (green), mouse anti EBA-175 mouse sera and Alexa Fluor 594-conjugated anti-mouse IgG goat antibodies to define location of micronemes (red) and DAPI to define location of nuclei (blue). Slides were observed under a confocal laser microscope (Nikon A1R). Bright field (i) and single slice confocal fluorescence images of merozoites (ii – iv) are shown. Three-dimensional reconstruction of confocal z stack fluorescence images of merozoites was performed using Imaris software (v –vii). Total actin (polymerized F-actin and monomeric G-actin) was detected with anti-actin rabbit sera followed by Alexa Fluor 594-conjugated anti-rabbit IgG goat antibodies (red in vii). Levels of total actin are similar in all conditions tested. Transfer of merozoites from IC to EC buffer leads to disassembly of polymerized F-actin at the apical tip of merozoites. Treatment of merozoites with FK506 and CsA prior to transfer to EC buffer leads to accumulation of polymerized actin at the apical end. Actin depolymerizing agent CytD and actin stabilizing agent JAS were used as control and result in reduced F-actin and increased F-actin at the apical tip of the merozoites respectively. Scale bar represents 1 μm. Treatment of merozoites with actin depolymerizing agents, CytD, ML-B and LA-B, resulted in increased secretion of microneme protein PfAMA-1 (a-c) whereas treatment with polymerized actin stabilizer JAS resulted in reduced secretion of PfAMA-1 (d).Singh S, More KR, Chitnis CE. Role of Calcineurin and Actin Dynamics in Regulated Secretion of Microneme Proteins in Plasmodium falciparum Merozoites during Erythrocyte Invasion. Cell Microbiol. 2013 16(1), 50–63
See original on MMP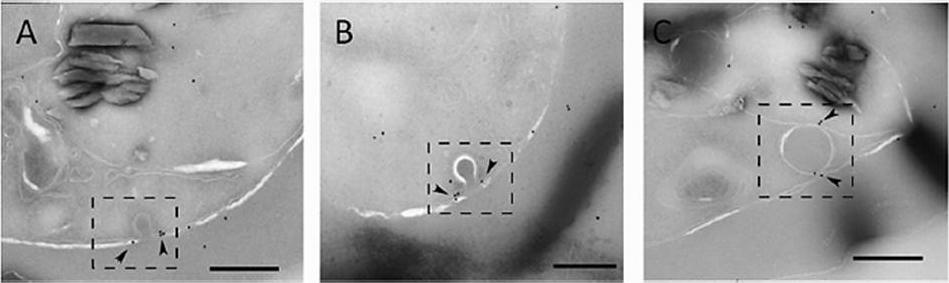
Actin localizes to the cytostome neck in IRBC. Representative immunoelectron micrographs of P. falciparum trophozoites probed with anti-T. gondii actin antibody. (A and B) Micrographs showing actin labeled with 12-nm gold particles (arrowheads) localized to electron-dense collars at the cytostome neck (dashed box). (C) Actin (arrowheads) associated with a hemoglobin-containing compartment in the parasite cytosol (dashed box). Scale bars, 250 nm.Milani KJ, Schneider TG, Taraschi TF. Defining the Morphology and Mechanism of the Hemoglobin Transport Pathway in Plasmodium falciparum-Infected Erythrocytes. Eukaryot Cell. 2015 14(4):415-26. PMID:
See original on MMP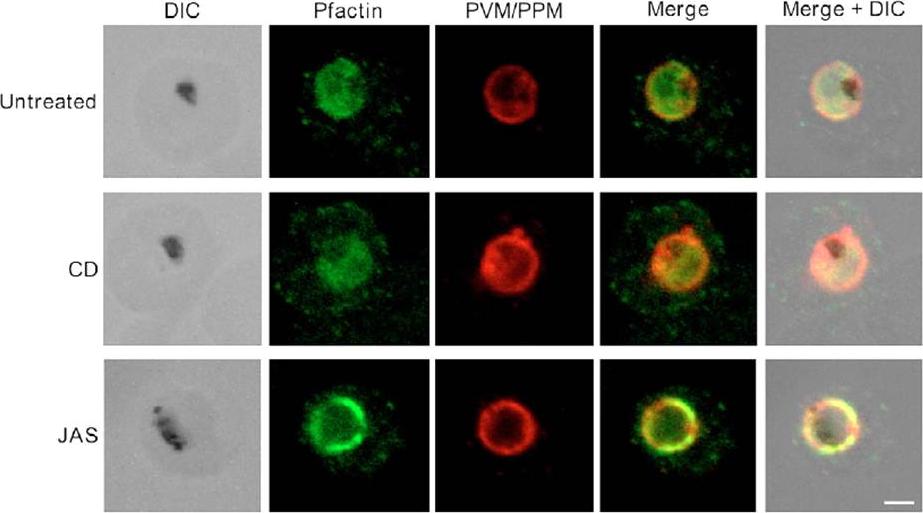
Pfactin distribution and localization in untreated, JAS- or CD-treated trophozoite stage PE. Representative confocal microscopy images showing Pfactin localization (green) in relation to the PVM/PPM (red) in untreated, JAS- and CD-treated trophozoite PE. Merge is a composite of the green and red images; merged+DIC is a composite of the DIC, green and red images. The electron-dense inclusion (hemozoin) in the DIC images indicates the location of the parasite FV. Bar, 2.0 μm.Lazarus MD, Schneider TG, Taraschi TF. A new model for hemoglobin ingestion and transport by the human malaria parasite Plasmodium falciparum. J Cell Sci. 2008 Jun 1;121(Pt 11):1937-49. PMID:
See original on MMPMore information
| PlasmoDB | PCHAS_1030900 |
| GeneDB | PCHAS_1030900 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | PC301589.00.0, PC301600.00.0, PCAS_102990, PCHAS_103090 |
| Orthologs | PBANKA_1030100 , PF3D7_1412500 , PKNH_1345800 , PVP01_1336400 , PVX_085830 , PY17X_1032500 |
| Google Scholar | Search for all mentions of this gene |