PY17X_1446500 macrophage migration inhibitory factor (MIF)
Disruptability [+]
| Species | Disruptability | Reference | Submitter | |
|---|---|---|---|---|
| P. yoelii yoelii 17X |
Possible |
RMgm-1433 | Imported from RMgmDB | |
| P. yoelii yoelii 17X |
Possible |
RMgm-665 | Imported from RMgmDB | |
| P. berghei ANKA |
Possible |
RMgm-26 | Imported from RMgmDB | |
| P. berghei ANKA |
Possible |
PlasmoGEM (Barseq) | PlasmoGEM | |
| P. falciparum 3D7 |
Possible |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen | |
Mutant phenotypes [+]
| Species | Stage | Phenotype | Reference | Submitter |
|---|---|---|---|---|
| P. yoelii yoelii 17X | Asexual |
No difference |
RMgm-1433 | Imported from RMgmDB |
| P. yoelii yoelii 17X | Asexual |
No difference |
RMgm-665 | Imported from RMgmDB |
| P. yoelii yoelii 17X | Gametocyte |
No difference |
RMgm-665 | Imported from RMgmDB |
| P. yoelii yoelii 17X | Ookinete |
No difference |
RMgm-665 | Imported from RMgmDB |
| P. yoelii yoelii 17X | Oocyst |
No difference |
RMgm-665 | Imported from RMgmDB |
| P. yoelii yoelii 17X | Sporozoite |
No difference |
RMgm-665 | Imported from RMgmDB |
| P. yoelii yoelii 17X | Liver |
Difference from wild-type |
RMgm-665
Less than 50% of mice infected with 1x10(3) P.yoeliimif sporozoites became patent. For mice that became patent, the onset of blood-stage parasitemia was delayed 2 days compared to that with the control. Patency was more frequent in mice after injecting with 1x10(3) P.yoeliimif sporozoites, and the onset of patency was delayed 1 day in mice that did develop blood-stage infection.Fewer P.yoeliimif liverstage parasites than control parasites were observed in mouse livers; however, these differences were not statistically significant Thus, the initial steps of hepatocyte invasion and dedifferentiation into a trophozoite appeared not to be affected significantly by the deletion of the mif gene. Microscopic valuation of infected livers revealed that the P.yoeliimif parasites were significantly smaller than control P. yoelii parasites at 44 hpi, with the average diameter of a control parasite being 40.1 10.3 m and the diameter of a mif-deficient parasite being 24.9 7.6 m |
Imported from RMgmDB |
| P. berghei ANKA | Asexual |
Difference from wild-type |
RMgm-26
Normal growth and multiplication rate. Infections in C57BL/6 and BALB/c mice result in increased numbers of reticulocytes |
Imported from RMgmDB |
| P. berghei ANKA | Asexual |
No difference |
PlasmoGEM (Barseq) | PlasmoGEM |
| P. berghei ANKA | Gametocyte |
No difference |
RMgm-26 | Imported from RMgmDB |
| P. berghei ANKA | Ookinete |
No difference |
RMgm-26 | Imported from RMgmDB |
| P. berghei ANKA | Oocyst |
No difference |
RMgm-26 | Imported from RMgmDB |
| P. berghei ANKA | Sporozoite |
No difference |
RMgm-26 | Imported from RMgmDB |
| P. berghei ANKA | Liver |
No difference |
RMgm-26 | Imported from RMgmDB |
Imaging data (from Malaria Metabolic Pathways)
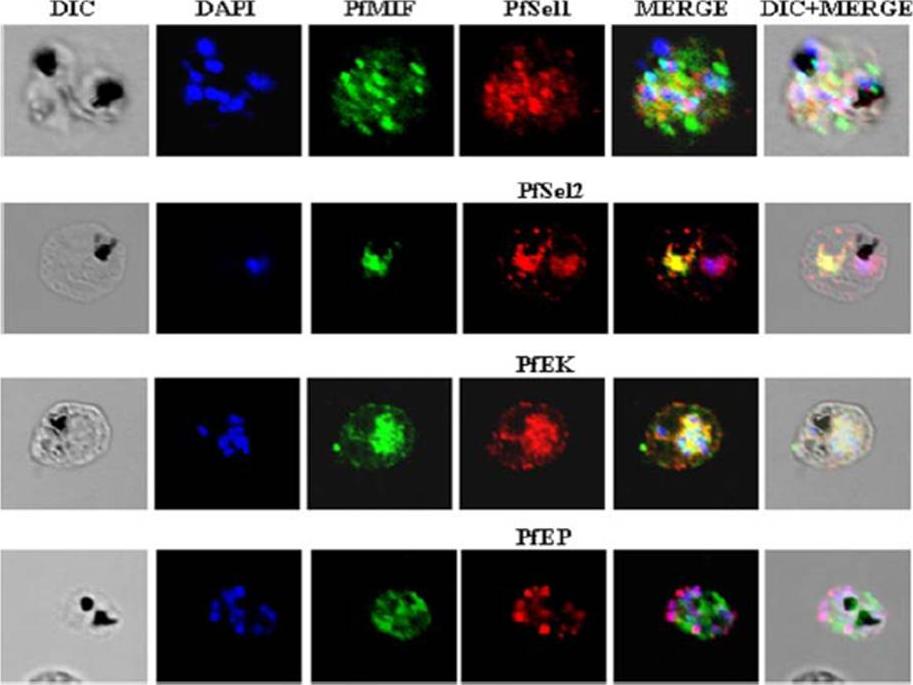
Indirect immunofluorescence assay and confocal microscopy to localize the selected excretory-secretory antigens by coimmunostaining of P. falciparum-infected erythrocytes. Air-dried infected erythrocytes were incubated with mouse antibodies specific to the selected proteins at 1:100 dilution (column 4; red). Rabbit aPfMIF antibody was used as a co-localization marker (column 3; green). The parasite nuclei were stained with 4’,6-diamidino-2-phenylindole (DAPI) (column 2; blue). Columns 1, 5, and 6 show a differential interference contrast (DIC) image, merged image, and differential interference contrast with merged image, respectively. Images showed punctate vesicle-like staining of the infected RBCs with antibodies to PfSEL1 (PFB0190c), PfSEL2 (Hrd3), PfEKand PfEP. PfEK also showing specific rimlike staining of the plasma membrane of the infected RBC. PfMIF is exported via the Maurer clefts to the extracellular medium after schizont rupture. A partial co-localization of PfMIF was seen with each of the four proteins, suggesting a distinct pathway for the release of these ESAs.Singh M, Mukherjee P, Narayanasamy K, Arora R, Sen SD, Gupta S, Natarajan K, Malhotra P. Proteome analysis of Plasmodium falciparum extracellular secretory antigens at asexual blood stages reveals a cohort of proteins with possible roles in immune modulation and signaling. Mol Cell Proteomics. 2009 8(9):2102-18.
See original on MMP
Localization of Plasmodium falciparum macrophage-migration inhibitory factor (PfMIF) in blood-stage parasites. Tightly synchronized parasites at different stages were stained with 4-6-diamidino-2-phenylindole (DAPI) to identify infected red blood cells (iRBCs), with P. falciparum skeleton bindingprotein 1 (PfSBP1) antiserum to indicate the iRBC cytosol, and with anti–PfMIF peptide IgG to determine their localization within iRBCs. The arrows in the bright-field micrographs indicate iRBCs. In ring-stage parasites, PfMIF was almost exclusively located within the parasite. However, as the parasites developed into trophozoites, PfMIF was clearly detected in both the parasite and the iRBC cytosol. Copyright Oxford University Press 2011.Cordery DV, Kishore U, Kyes S, Shafi MJ, Watkins KR, Williams TN, Marsh K, Urban BC. Characterization of a Plasmodium falciparum macrophage-migration inhibitory factor homologue. J Infect Dis. 2007 195:905-12.
See original on MMP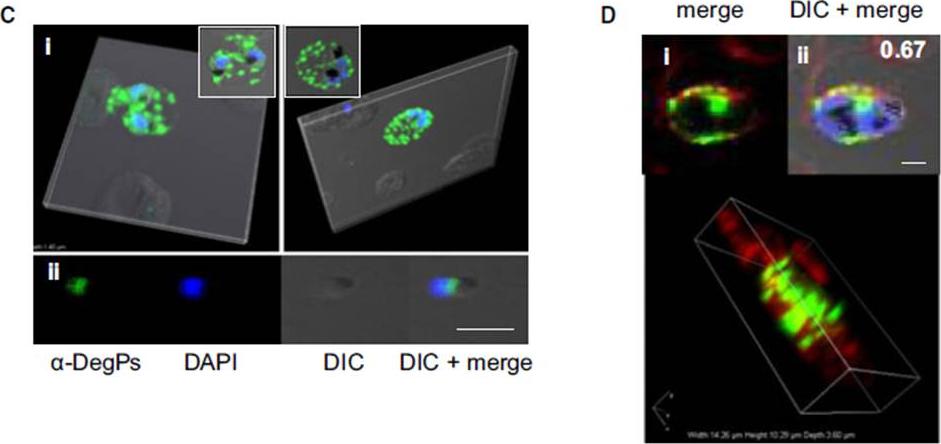
Indirect immunofluorescence assay and confocal microscopy of late stages of P. falciparum-infected erythrocytes and free merozoites. P. falciparum parasites were probed using rat anti-DegP antibody followed by fluorescein isothiocyanate-conjugated anti-rat IgG (green). Other marker proteins were stained with the rabbit antiserum/ antibody followed by Cy3-conjugated anti-rabbit IgG (red). The parasite nuclei were counterstained with 4′,6-diamidino-2-phenylindole (DAPI) (blue), and slides were visualized using a confocal laser scanning microscope. z-section of late-stage parasites stained for DegP showed green fluorescence signals in the cytosol of the parasite and the cytosol and membrane of infected RBCs (Ci). DegP signals were observed on free merozoites (Cii). The surface localization of PfDegP in infected RBCs was confirmed by co-localization with an erythrocyte membrane marker (red) (Di). Yellow spots indicate merged fluorescent signals from DegP and band 3 antibodies (Dii). SERA5 and MIF were used as markers for parasitophorous vacuole and secretory protein in the culture supernatant, respectively. Sharma S, Jadli M, Singh A, Arora K, Malhotra P. A secretory multifunctional serine protease, DegP of Plasmodium falciparum, plays an important role in thermo-oxidative stress, parasite growth and development. FEBS J. 2014 Mar;281(6):1679-99.
See original on MMP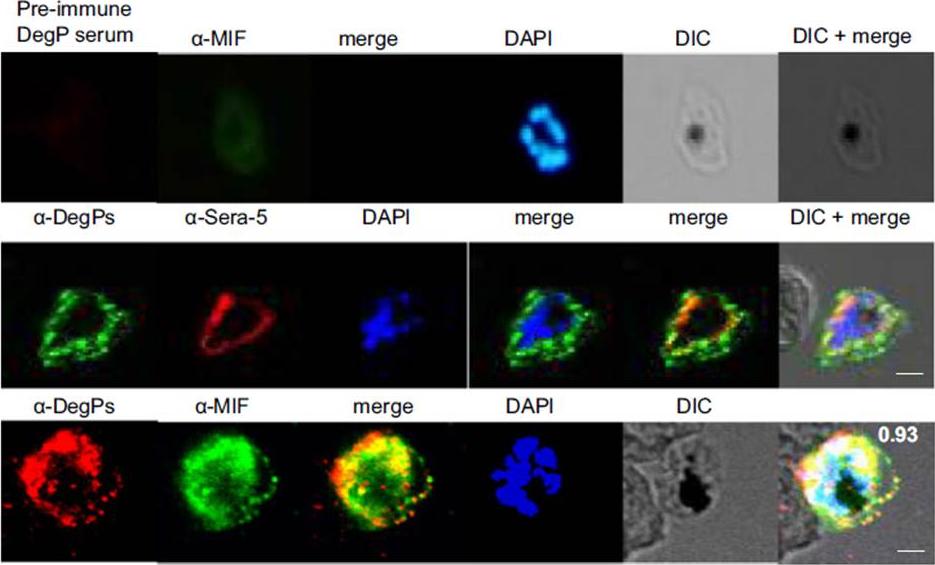
Indirect immunofluorescence assay and confocal microscopy of late stages of P. falciparum-infected erythrocytes and free merozoites. P. falciparum parasites were probed using rat anti-DegP antibody followed by fluorescein isothiocyanate-conjugated anti-rat IgG (green). Other marker proteins were stained with the rabbit antiserum/antibody followed by Cy3-conjugated anti-rabbit IgG (red). PfSera5 and PfMIF were used as markers for parasitophorous vacuole and secretory protein in the culture supernatant, respectively. Significant merging of the fluorescent signal for DegP was observed with SERA5 and MIF signals, indicating the presence of DegP in the cytosol of infected RBCs.Sharma S, Jadli M, Singh A, Arora K, Malhotra P. A secretory multifunctional serine protease, DegP of Plasmodium falciparum, plays an important role in thermo-oxidative stress, parasite growth and development. FEBS J. 2014 281(6):1679-99.
See original on MMPMore information
| PlasmoDB | PY17X_1446500 |
| GeneDB | PY17X_1446500 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | null |
| Orthologs | PBANKA_1444000 , PCHAS_1446200 , PF3D7_1229400 , PKNH_1448700 , PVP01_1447500 , PVX_124095 |
| Google Scholar | Search for all mentions of this gene |