PF3D7_1116800 heat shock protein 101 (HSP101)
Disruptability [+]
| Species | Disruptability | Reference | Submitter | |
|---|---|---|---|---|
| P. falciparum 3D7 |
Refractory |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen | |
| P. berghei ANKA |
Refractory |
RMgm-945 | Imported from RMgmDB | |
| P. berghei ANKA |
Refractory |
RMgm-914 | Imported from RMgmDB | |
| P. berghei ANKA |
Refractory |
PlasmoGEM (Barseq) | PlasmoGEM | |
Mutant phenotypes [+]
| Species | Stage | Phenotype | Reference | Submitter |
|---|---|---|---|---|
| P. falciparum 3D7 | Asexual |
Cell cycle arrest |
25043010 (Conditional)
Ring stage arrest, with a near complete block in export. Substrates accumulate in the vacuole. |
Theo Sanderson, Wellcome Trust Sanger Institute |
| P. falciparum 3D7 | Gametocyte |
Attenuated |
25043010 (Conditional)
Strongly reduced gametocyte production. |
Theo Sanderson, Wellcome Trust Sanger Institute |
Imaging data (from Malaria Metabolic Pathways)
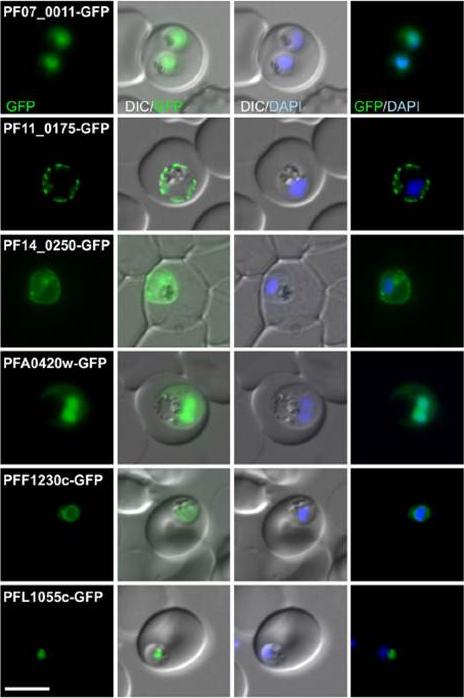
Fluorescence pattern of non-exported (B) or exported (C) GFP fusion proteins. PF07_0011 . unknown function; PF11_0175 heat shock protein 101; PF14_0250 lipase; PFA0429w unknown function; PFF1230c unknown function; PFL1055c unknown function. PF07_0011 and PFA0420w were in the nucleus with a cytoplasmic pool, PF11_0175 and PF14_0250 were at the parasite periphery (consistent with a PPM, PV or PVM location, PFF1230c was perinuclear, suggesting an ER location, and PFL1055c was found in a nucleus proximal focus that co-localized with the GolgiHeiber A, Kruse F, Pick C, Grüring C, Flemming S, Oberli A, Schoeler H, Retzlaff S, Mesén-Ramírez P, Hiss JA, Kadekoppala M, Hecht L, Holder AA, Gilberger TW, Spielmann T. Identification of New PNEPs Indicates a Substantial Non-PEXEL Exportome and Underpins Common Features in Plasmodium falciparum Protein Export. PLoS Pathog. 2013 9(8):e1003546.
See original on MMP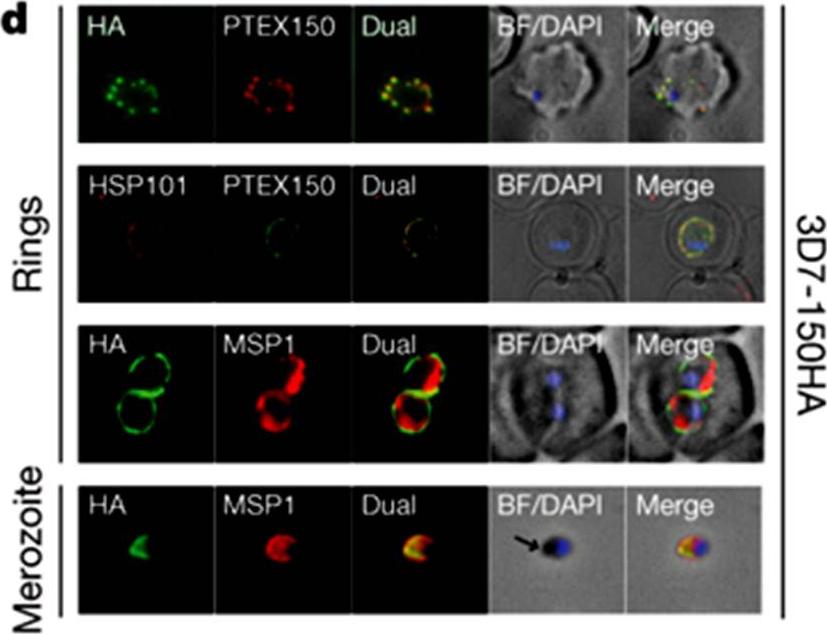
HSP101 and PTEX150 co-localize and have dual apical merozoite and PVM localization. Double labelling IFA on fixed 3D7-150HA ring- and merozoite-stage parasites using the antibodies as indicated. 3D7-150HA is a transgenic P. falciparum (3D7) parasite line where the endogenous gene was modified at its carboxy terminus to include a triple haemagglutinin (HA) epitope tag. The arrows indicate the apical end of the merozoite. BF, bright-field; DAPI, nucleic acid stain 49,6-diamidino-2-phenylindole. PTEX150 and HSP101 co-localize and are found in discrete foci in the membranes surrounding the ring-stage parasite. This membrane is the PVM, as fluorescence surrounds the parasite membrane marker MSP1 (PFI1475w).de Koning-Ward TF, Gilson PR, Boddey JA, Rug M, Smith BJ, Papenfuss AT, Sanders PR, Lundie RJ, Maier AG, Cowman AF, Crabb BS. A newly discovered protein export machine in malaria parasites. Nature. 2009 459(7249):945-9.
See original on MMP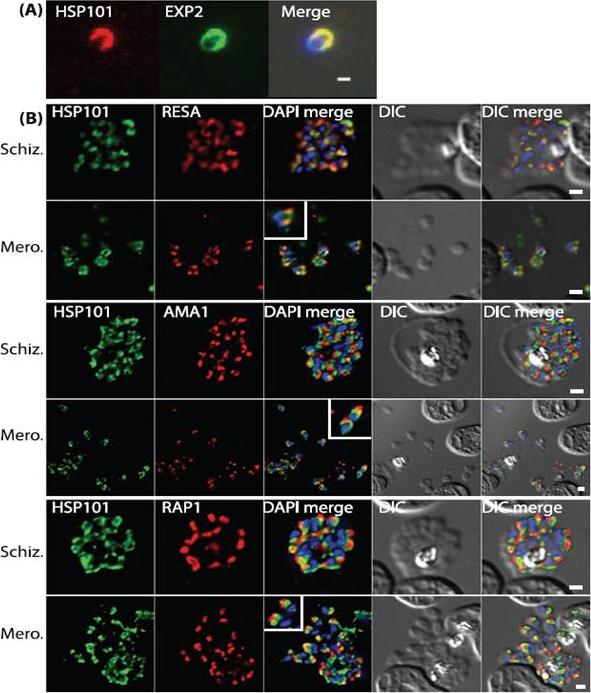
PTEX components are apically localised in merozoite dense granules. (A and B) Immunofluorescence assays were completed during schizogony and were probed for either PTEX components or markers of distinct organelles as indicated in each of the panels. (A) Merozoites probed with antibodies specific to both HSP101 and EXP2 demonstrate apical localization for both proteins. (B) Schizonts and merozoites were labeled with antibodies specific to HSP101 and either RESA (dense granules), AMA1 (micronemes) or RAP1 (rhoptries). Co-localisation quantitation reveals that HSP101 localises most closely with dense granule associated RESA (top two panels). Scale bar: 1μm. Bullen HE, Charnaud SC, Kalanon M, Riglar DT, Dekiwadia C, Kangwanrangsan N, Torii M, Tsuboi T, Baum J, Ralph SA, Cowman AF, de Koning-Ward TF, Crabb BS, Gilson PR. Biosynthesis, localisation and macromolecular arrangement of the Plasmodium falciparum translocon of exported proteins; PTEX. J Biol Chem. 2012 287(11):7871-84.
See original on MMP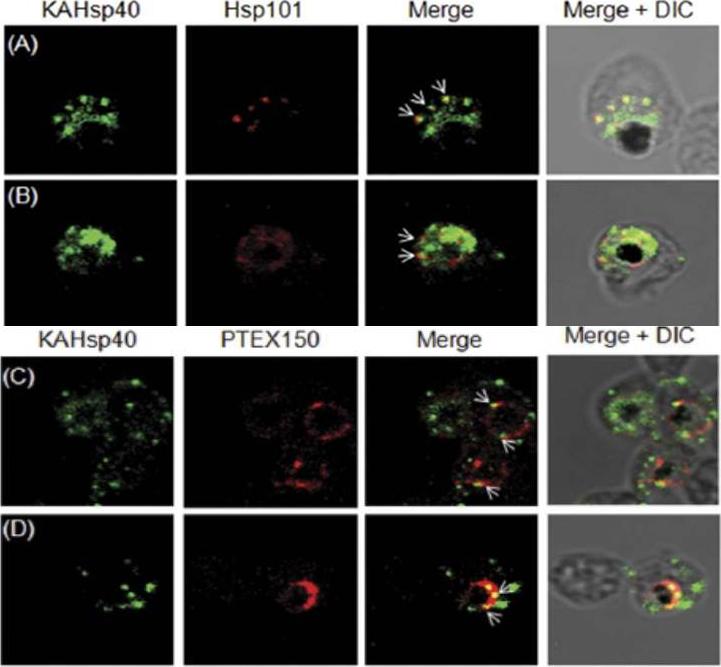
KAHsp40 associates with the PEXEL translocon on the PVM. (A, B) IFA analysis reveals that KAHsp40 and Hsp101 co-localize with each other on the PVM. (C, D) IFA analysis reveals that KAHsp40 and PTEX150 co-localize with each other on the PVM. White arrows indicate the discrete foci in which they co-localize.Acharya P, Chaubey S, Grover M, Tatu U. An Exported Heat Shock Protein 40 Associates with Pathogenesis-Related Knobs in Plasmodium falciparum Infected Erythrocytes. PLoS One. 2012;7(9):e44605.
See original on MMP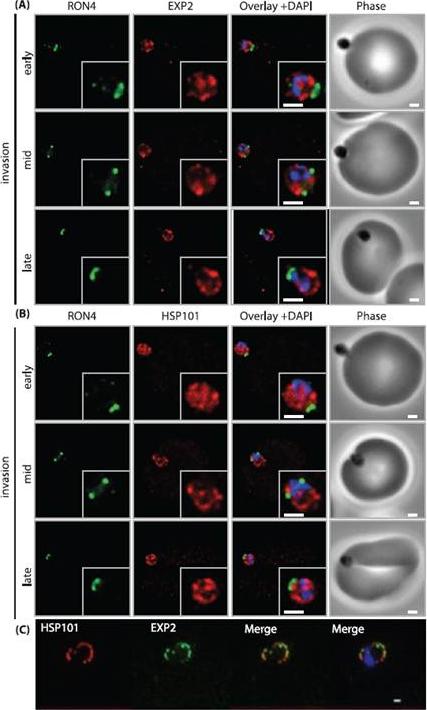
PTEX components are carried into the newly infected erythrocyte and are present at the PVM throughout intraerythrocytic development. (A and B) Time-course of merozoite invasion by widefield immunofluorescence microscopy with deconvolution (single slice shown) labelled for (A) tight junction component RON4 and PTEX component EXP2 or (B) RON4 and PTEX component HSP101. Both PTEX components are carried by the invading merozoite into the erythrocyte. Scale bar: 1μm. (C) HSP101 and EXP2 co-localise to distinct puncta at the PVM during later ring stage development. Scale bar: 1μm. Bullen HE, Charnaud SC, Kalanon M, Riglar DT, Dekiwadia C, Kangwanrangsan N, Torii M, Tsuboi T, Baum J, Ralph SA, Cowman AF, de Koning-Ward TF, Crabb BS, Gilson PR. Biosynthesis, localisation and macromolecular arrangement of the Plasmodium falciparum translocon of exported proteins; PTEX. J Biol Chem. 2012 287(11):7871-84
See original on MMP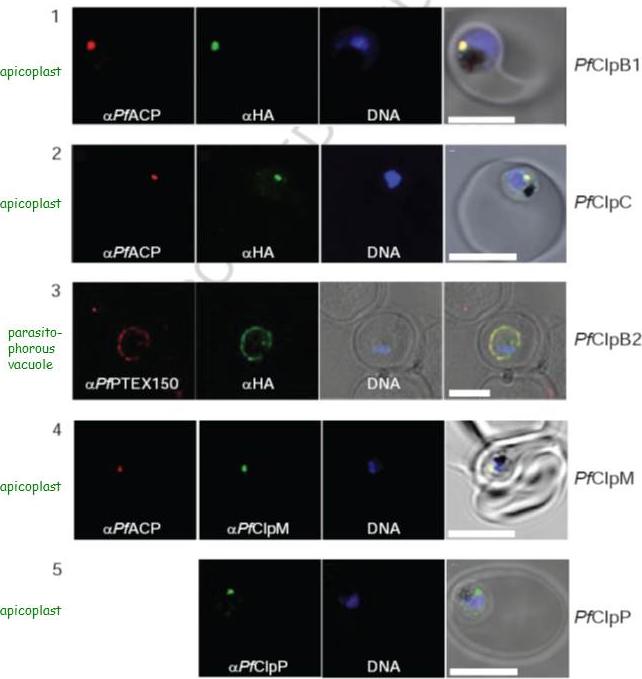
Localization of PfClpB1, PfClpC, PfClpB2, PfClpM, and PfClpP of P. falciparum was tested by immunofluorescent colocalization. In panels 1 and 2, PfClpB1-Strp-3×HA or PfClpC-Strp-3×HA parasites were probed with the apicoplast marker anti-ACP PFB0385w antisera (red), with anti-HA antisera (green), and with Hoescht 33342 to stain for DNA (blue). In panel 3, PfClpB2-Strp-3×HA parasites were probed with the parasitophorous vacuole marker anti-PTEX150 antisera (red), with anti-HA antisera (green), and with Hoescht 33342 (blue). In panels 4 and 5, wild type 3D7 strain was probed with anti-ACP antisera (red), with anti-PfClpM antisera or anti-ClpP antisera (green), and with Hoescht 33342 (blue). For all panels, the right most image represents the merged fluorescence images with the DIC or transmission image. The scale bars correspond to 5 mm.El Bakkouri M, Pow A, Mulichak A, Cheung KL, Artz JD, Amani M, Fell S, de Koning T, Goodman CD, McFadden GI, Ortega J, Hui R, Houry WA. The Clp chaperones and proteases of the human malaria parasite Plasmodium falciparum. J Mol Biol. 2010 404(3):456-77
See original on MMP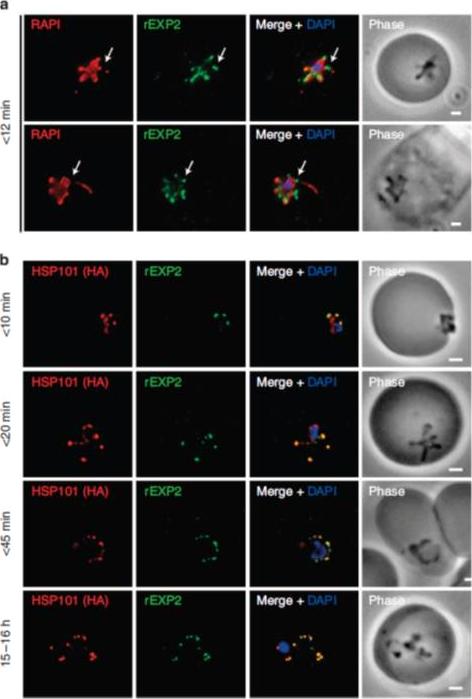
PTEX components localize to the parasite periphery immediately following invasion. (a) Widefield deconvolution imaging of parasites fixed o12 min following erythrocyte invasion and labelled by IFA for EXP2 (green), the PV (RAP1, red) and the nucleus (40,6–diamidino-2-phenylindole (DAPI), blue) showed EXP2 localized in puncta at the parasite periphery at this time point (scale bar, 1 mm). HSP101HA parasites were fixed o10 min, <20 min, <45 min and 15–16 h following erythrocyte invasion and labelled by IFA for EXP2 (green), HSP101HA (HA, red) and the nucleus (DAPI, blue). (b)Widefield deconvolution microscopy (scale bar, 1 mm).Riglar DT, Rogers KL, Hanssen E, Turnbull L, Bullen HE, Charnaud SC, Przyborski J, Gilson PR, Whitchurch CB, Crabb BS, Baum J, Cowman AF. Spatial association with PTEX complexes defines regions for effector export into Plasmodium falciparum-infected erythrocytes. Nat Commun. 2013 Jan 29;4:1415.
See original on MMP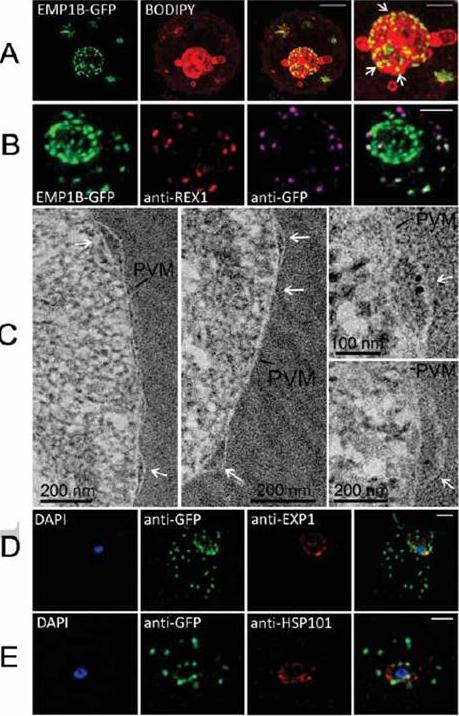
Characterization of an intermediate compartment in PfEMP1 export. (A) Transfectants (ITK strain) expressing the PfEMP1B-GFP chimera were co-labeled with BODIPY-ceramide and imaged using 3D-SIM. See higher magnification image at right. (B) PfEMP1B-GFP transfectants (endogenous GFP fluorescence; green) were permeabilized with EqtII and antibody-accessible epitopes were labeled with anti-GFP (magenta) and anti-REX1 (red). (C) PfEMP1B-GFP transfectants were fixed and resin-embedded and sections were labeled with anti-GFP and protein-A-gold (6 nm left panels, 10 nm right panels). Labeling is observed at bulges in the PV (arrows). (D) Smears of PfEMP1B-GFP transfectant-infected RBCs were fixed with acetone: methanol and stained with antibodies recognizing GFP and Exp1 (D) or Hsp101 (E). Scale bars = 2 μm (A, first three columns), 1 μm (A, last column), 2 μm (B), as marked (C) and 2 μm (D, E).McMillan PJ, Millet C, Batinovic S, Maiorca M, Hanssen E, Kenny S, Muhle RA, Melcher M, Fidock DA, Smith JD, Dixon MW, Tilley L. Spatial and temporal mapping of the PfEMP1 export pathway in Plasmodium falciparum. Cell Microbiol. 15(8):1401-18
See original on MMP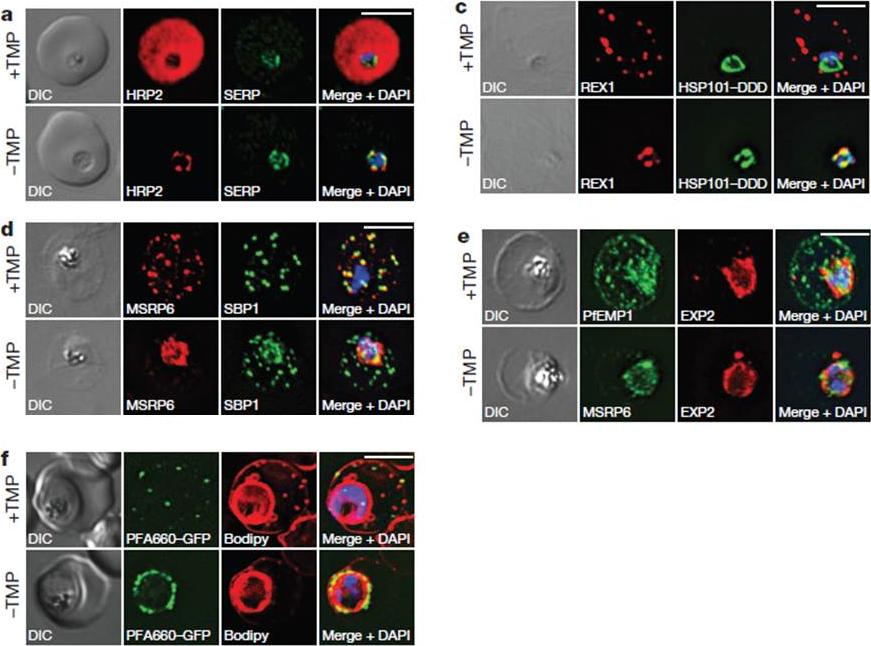
HSP101 is required for export of PEXEL and PNEP proteins. a, c, Immunofluorescence assay (IFA) of ring-stage 13F10 parasites with orwithout TMP (DDD-stabilizing small molecule trimethoprim). TMP was removed in late schizont stage and parasites were allowed to reinvade and grow 18–24 h before fixation with paraformaldehyde (a) or acetone (c). a, IFA of the exported PEXEL-containing protein HRP2. c, IFAs of the PNEP REX1, which colocalizes with HSP101DDD at the PVM in the absence of TMP. d, e, IFA of trophozoite-stage 13F10 parasites with or withoutTMP. TMP was removed in late ring stage and parasites were allowed to develop 12–24 h before fixation with acetone. f, Live fluorescence imaging of 13F10 parasites expressing a PFA660–GFP fusion and labelled with Bodipy TR Ceramide to demarcate the PVM (other membranes are also labelled). TMP treatment as in d, e. All scale bars, 5 mm. Images in c–f are representative of two independent experiments.Beck JR, Muralidharan V, Oksman A, Goldberg DE. PTEX component HSP101 mediates export of diverse malaria effectors into host erythrocytes. Nature. 2014 Jul 16. [Epub ahead of print]
See original on MMP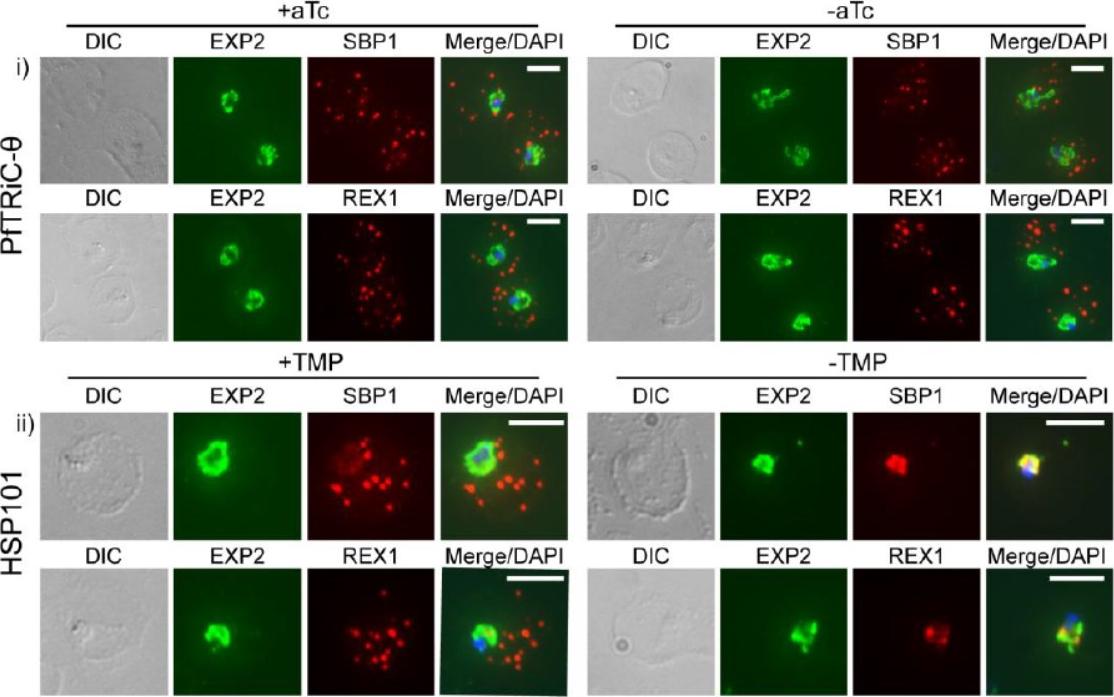
Loss of PfTRiC-θ does not alter export of PfSBP1 or PfREX1. IFA of acetone-fixed ring-stage infected RBC. i) PfTRiC-θ ± 0.5 μM aTc (anhydrotetracycline), ii) HSP101 ± 10 μM trimethoprim (TMP). DAPI stains the parasite nuclei and PfEXP2 delineates the parasite parasitophorous vacuole compartment. Scale bars = 5 μm. Data are representative of two independent assays. DIC = differential interference contrast. Loss of the PfTRiC complex does not alter export of PfSBP1 or PfREX1 (i). This was in contrast to the block in export of PfSBP1 and PfREX1 observed upon TMP removal in the previously characterized PfHSP101-DDD line (ii).Spillman NJ, Beck JR, Ganesan SM, Niles JC, Goldberg DE. The chaperonin TRiC forms an oligomeric complex in the malaria parasite cytosol. Cell Microbiol. 2017 Jan 9.
See original on MMPMore information
| PlasmoDB | PF3D7_1116800 |
| GeneDB | PF3D7_1116800 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | PF11_0175 |
| Orthologs | PBANKA_0931200 , PCHAS_0913100 , PKNH_0914500 , PVP01_0917400 , PVX_091470 , PY17X_0933200 |
| Google Scholar | Search for all mentions of this gene |