PF3D7_1016300 glycophorin binding protein (GBP)
Disruptability [+]
| Species | Disruptability | Reference | Submitter |
|---|---|---|---|
| P. falciparum 3D7 |
Possible |
18614010 | Theo Sanderson, Wellcome Trust Sanger Institute |
| P. falciparum 3D7 |
Possible |
18614010 Increase in membrane rigidity compared to wild type CS2 strain |
M. Pratap, Australian National University |
| P. falciparum 3D7 |
Possible |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen |
Mutant phenotypes [+]
| Species | Stage | Phenotype | Reference | Submitter |
|---|---|---|---|---|
| P. falciparum 3D7 | Asexual |
Altered cytoadherence |
18614010 Increase in membrane rigidity compared to wild type CS2 strain |
M. Pratap, Australian National University |
Imaging data (from Malaria Metabolic Pathways)
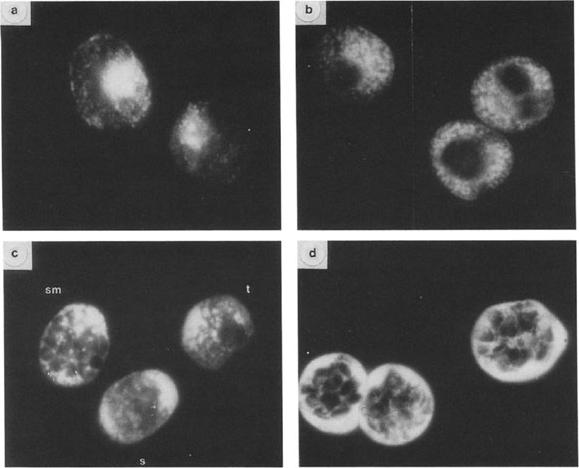
Immunofluorescent localization of GBP-130 at different stages. Thin smears of cultures overlaid with R311 serum and FITC-goat anti-rabbit serum. (a, c) cells fixed with acetone; (b, d) cells fixed with 1% paraformaldehyde; (a, b) trophozoite stage parasites; (d) segmenter stage parasites; (c) three different stages: schizont (s), segmenter (sm), trophozoite (t). Magnification: X 950. Differences in antigen localization are apparent depending on the methods used to fix to the parasites. In parasites fixed in acetone (Figs. a, c) the distribution of the antigen as detected by fluorescence is more diffuse. Immunofluorescence patterns shown in Figs. b and d are of cells fixed with paraformaldehyde. More internal structure is apparent in parasites fixed in this manner. At the trophozoite stage fixed in acetone (Fig. a) the antigen appears to be localized on the parasite and in small inclusions in the erythrocyte cytoplasm. In late trophozoites fixed in paraformaldehyde (Fig. b) the fluorescent staining is more intense and appears predominantly in vesicles or inclusions in the erythrocyte cytoplasm and perhaps the parasitophorous vacuole. The antigen is not present in uninfected erythrocytes. In mature schizonts (segmenters) fixed in paraformaldehyde (Fig. d) the antigen is localized principally in the space between the schizont and the erythrocyte membrane but also appears around the periphery of the merozoite.Perkins M. Stage-dependent processing and localization of a Plasmodium falciparum protein of 130,000 molecular weight. Exp Parasitol. 1988 65:61-8. Copyright Elsevier 2010.
See original on MMP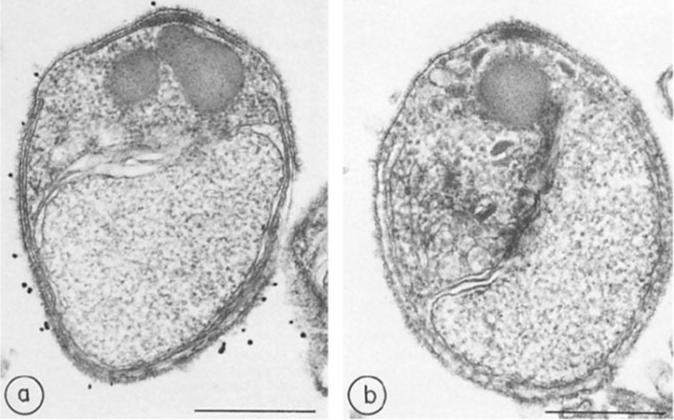
Immunocytochemistry of glycophorin-binding proteins, a, Merozoites treated with immune rabbit serum and protein A-gold; b, merozoites treated with preimmune rabbit serum and protein A-gold. x 45,000. Bar, 500 nm.Perkins ME. Surface proteins of Plasmodium falciparum merozoites binding to the erythrocyte receptor, glycophorin. J Exp Med. 1984 160:788-98. Copyright Elsevier 2010.
See original on MMP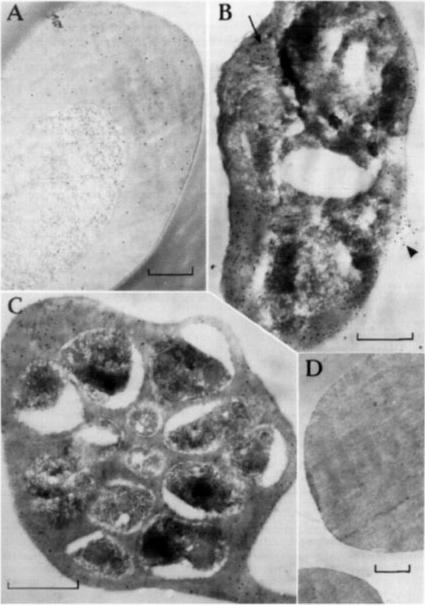
Post-embedding immunolabeling of FC27 with human antl-Ag 23 and protein A-gold (bars equal 0.5 mm) Samples were fixed for 10 rain with 0.25% glutaraldehyde and embedded m L.R. White resin, heat polymerized at 50°C. (A) Trophozoite-infected erythrocyte with labeling evenly dispersed over the cytoplasm. (B) Mature trophozoite-infected cell with more labeling over the erythrocyte cytoplasm and over a presumed food vacuole (arrow). Antigen is apparently leaking from an area of damaged erythrocyte plasma membrane (arrow head) (C) Schizont-infected erythrocyte containing developing merozoites. Label is again over the erythrocyte cytoplasm largely exterior to the presumed parasitophorous vacuole membrane. (D) Uninfected erythrocytes without significant labeling.Bianco AE, Culvenor JG, Coppel RL, Crewther PE, McIntyre P, Favaloro JM, Brown GV, Kemp DJ, Anders RF. Putative glycophorin-binding protein is secreted from schizonts of Plasmodium falciparum. Mol Biochem Parasitol. 1987 23:91-102. Copyright Elsevier 2010.
See original on MMP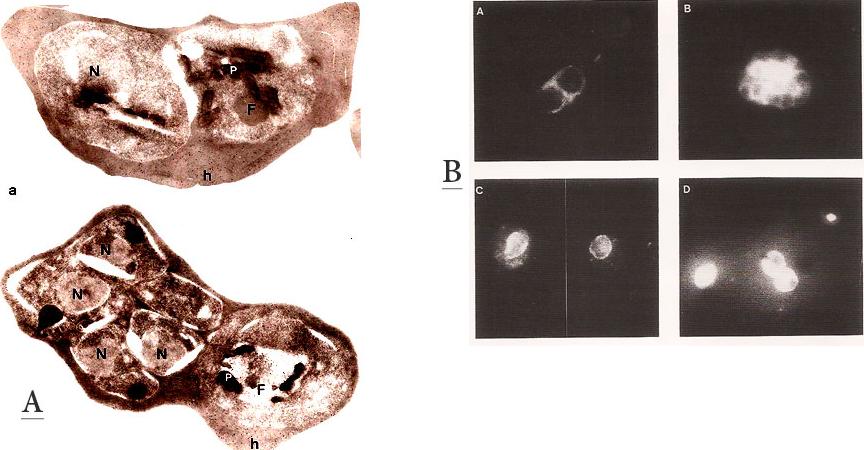
A. Immunofluorescence patterns of the 96.tR antigen. Young schizont (A) and segmenter stage parasites (B), fixed with 1 % formaldehyde in PBS. (C) trophozoite stage fixed with 100 % methanol at room temp. (D) Air-dried parasites showing trophozoite and schizont.B. Immunogold labeling of the 96.tR antigen on Lowicryl K4M-embedded parasites. Upper panel: Early schizont – the labeling is randomly scattered over the erythrocyte cytoplasm (h). No labeling is found in the parasite except in the food vacuole (F). Lower panel: Late schizont and trophozoite. Labeling is found only the host cell cytoplasm (h) and on the food vacuole of the trophozoite (F). x25,000. Bar = 1 mm.Bonnefoy S, Mattei D, Dubremetz JF, Guillotte M, Jouin H, Ozaki LS, Sibilli L, Mercereau-Puijalon O. Plasmodium falciparum: molecular analysis of a putative protective antigen, the thermostable 96-kDa protein. Exp Parasitol. 1988 65:69-83.
See original on MMP
GBP130 chimaeras localise to the endoplasmic reticulum in addition to the parasitophorous vacuole or erythrocyte cytosol. Images captured by immunofluorescence microscopy show substantial colocalisation of GBP130 chimaeras with the endoplasmic reticulum protein ERC PF11_0098 (upper panel). Intraparasitic fluorescence is also evident when chimaeras were colocalised with the parasitophorous vacuole membrane protein EXP-2 PF14_0678 (lower panel). Only the wild-type PEXEL chimaera is efficiently exported to the erythrocyte cytosol.Boddey JA, Moritz RL, Simpson RJ, Cowman AF. Role of the Plasmodium export element in trafficking parasite proteins to the infected erythrocyte. Traffic. 2009 10:285-99. Copyright John Wiley & Sons Ltd. 2010.
See original on MMP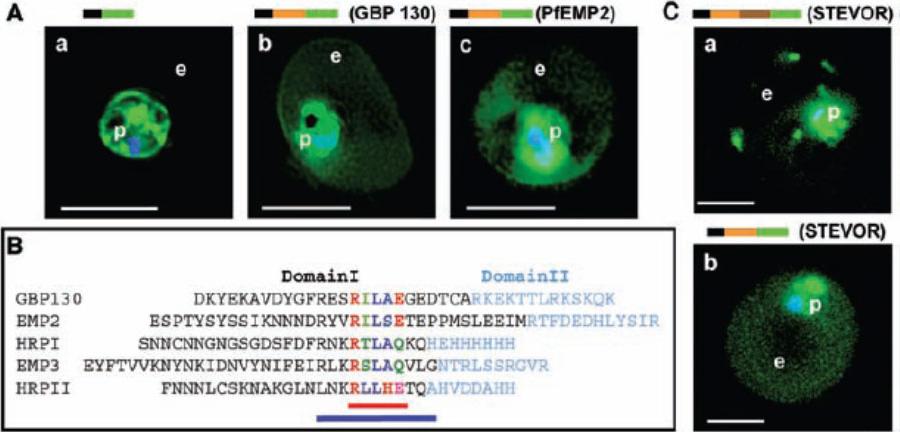
Identification of a conserved motif of 11 amino acids in VTSs of parasite proteins exported to the erythrocyte. (A) VTS of proteins PfGBP 130 and PfEMP2 target GFP to the erythrocyte. Projections (0-) of live cells expressing GFP chimeras of a SS alone (a), SSVTSGBP130 (b), and SSVTSEMP2 (c). (B) Alignment of MEME motifs in VTSs of indicated five exported soluble proteins. Red bar underlines 5–amino acid MEME motif 1. Blue bar underlines 11–amino acid MEME motif 2. (C) Projections (0-) of live cells expressing GFP chimeras of full-length STEVOR (a) and SSVTSSTEVOR (b).Hiller NL, Bhattacharjee S, van Ooij C, Liolios K, Harrison T, Lopez-Estraño C, Haldar K. A host-targeting signal in virulence proteins reveals a secretome in malarial infection. Science. 2004 306(5703):1934-7.
See original on MMP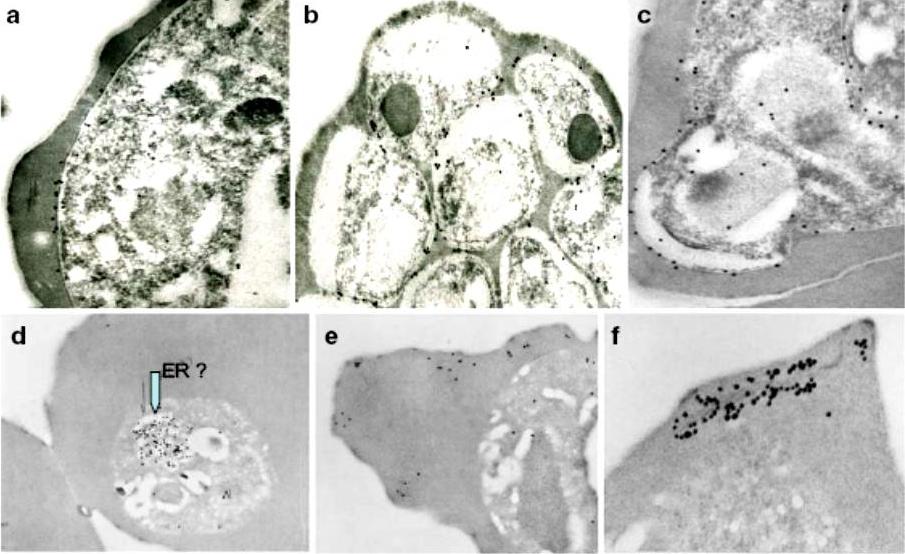
Immunoelectron micrographs of young trophozoite-, late trophozoite-, young schizont-, and late schizont-infected erythrocytes reactive with rabbit antisera against PfMC-2TM peptides, 703, 705, and 713 and Mab SP1A6 against the 130 kDa Maurer’s cleft protein. a–c Peptide-specific antisera are reactive with the PV/PVM and weakly around clefts. d–f Mab SP1A6 is reactive with structures within the parasite in young trophozoites, probably the endoplasmic reticulum (d), late trophozoites in erythrocyte cytoplasm under knobs (e), and in young schizonts around longitudinal clefts in the erythrocyte cytoplasm (f).Tsarukyanova I, Drazba JA, Fujioka H, Yadav SP, Sam-Yellowe TY. Proteins of the Plasmodium falciparum two transmembrane Maurer's cleft protein family, PfMC-2TM, and the 130 kDa Maurer's cleft protein define different domains of the infected erythrocyte intramembranous network. Parasitol Res. 2009 104(4):875-91.
See original on MMP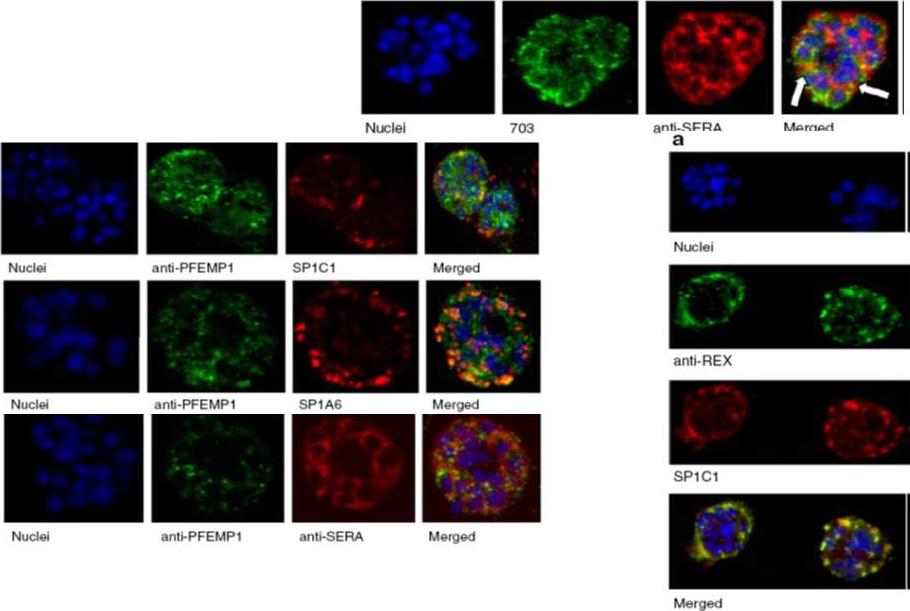
Upper row: Immunofluorescence assay of rabbit antisera against PfMC-2TM peptides colocalized with anti-SERA antibodies. Arrows indicate areas of colocalization between SERA and PfMC-2TM. Left Panel: Immunofluorescence assay of rabbit antisera against PfEMP1 with Mabs SP1C1 (a), Mab SP1A6 (b), and anti-SERA (c). PfEMP1 colocalized with PfMC-2TM, the 130 kDa Maurer’s cleft protein and SERA. Right panel: Immunofluorescence assay of rabbit antisera against REX PFI1735c colocalized with Mabs SP1C1. Tsarukyanova I, Drazba JA, Fujioka H, Yadav SP, Sam-Yellowe TY. Proteins of the Plasmodium falciparum two transmembrane Maurer's cleft protein family, PfMC-2TM, and the 130 kDa Maurer's cleft protein define different domains of the infected erythrocyte intramembranous network. Parasitol Res. 2009 104(4):875-91.
See original on MMP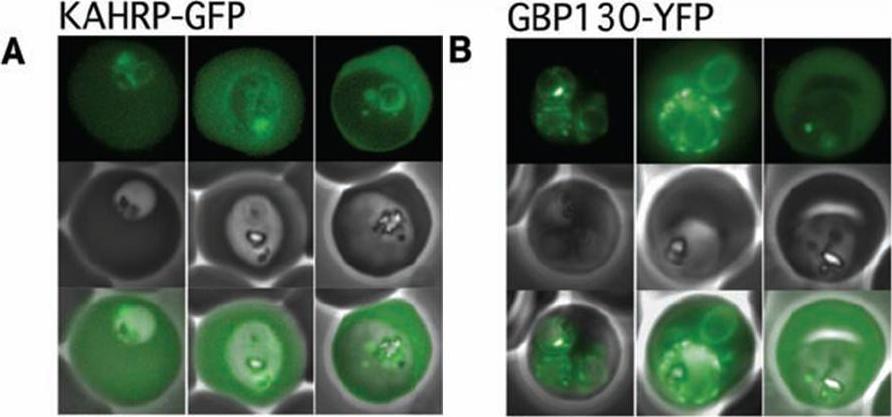
The Pexel motif is necessary for export of soluble proteins in P. falciparum. The reporter is exported only when fused to the N terminus of KAHRP (A) or GBP130 (B) including the complete Pexel motif. Representative cells expressing the exported reporter are shown in ring stage (left), early (center), and late trophozoites (right). Note that KAHRP-GFP is already exported into the host cell in early stages, whereas GBP130-YFP is retained in the parasitophorous vacuole and released after development into mature trophozoite stages, perhaps because of its unusually recessed or embedded signal sequence.Marti M, Good RT, Rug M, Knuepfer E, Cowman AF. Targeting malaria virulence and remodeling proteins to the host erythrocyte. Science. 2004 306(5703):1930-3. Copyright 2010.
See original on MMP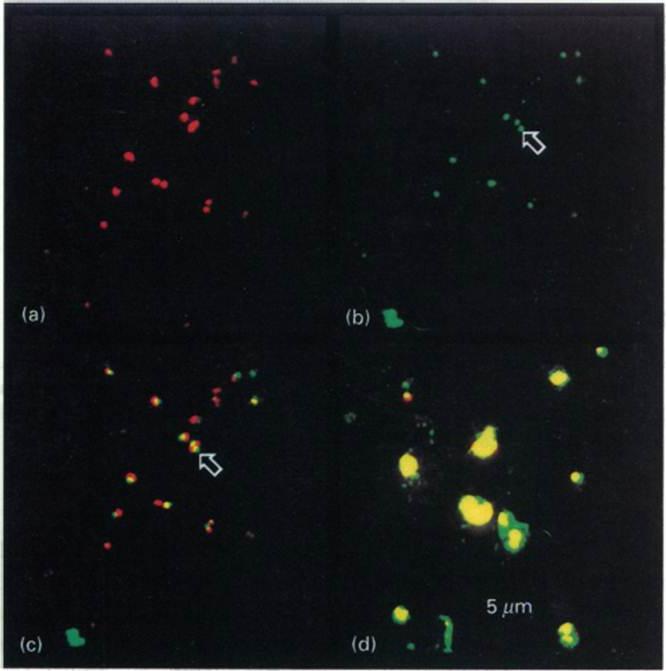
Intracellular distribution of GBP in the presence of BFA Ring-infected erythrocytes were cultured in the presence (a-c) or absence (d) of BFA, fixed and incubated with antibodies to GBP, followed by a mixture of fluorescein isothiocyanate-conjugated secondary antibody and propidium iodite. The same field was analysed for staining of the nuclei (a) or the presence of GBP (b). (e) Superimposition of (a) and (b). (d) Superimposition of nuclear and protein staining in control cells. The arrows highlight the location of GBP. In untreated cells GBP is found in the host cell cytoplasm. In the presence of BFA, GBP was not transported into the erythrocyte, but remained inside the parasite cell. Benting J, Mattei D, Lingelbach K. Brefeldin A inhibits transport of the glycophorin-binding protein from Plasmodium falciparum into the host erythrocyte. Biochem J. 1994 300:821-6.
See original on MMP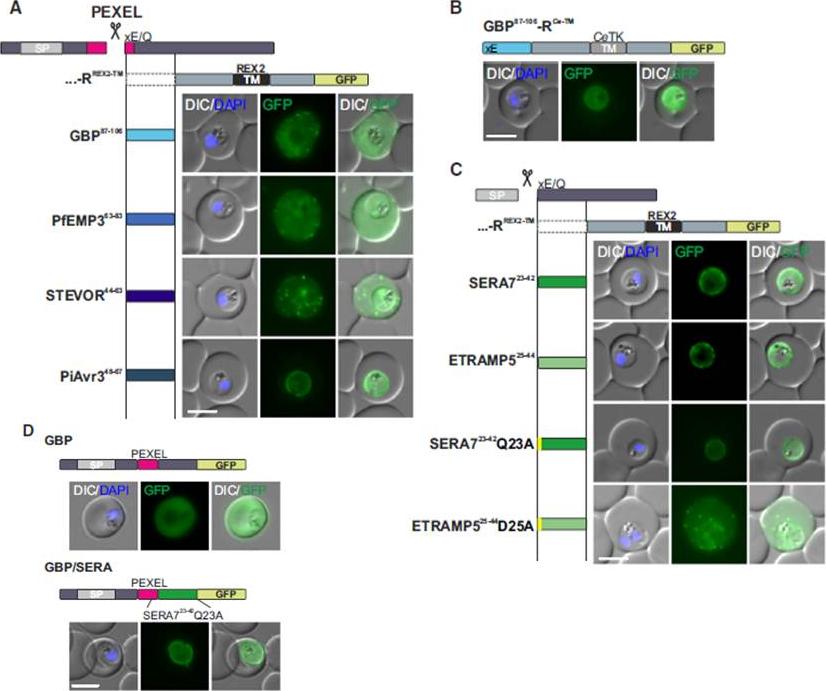
Mature PEXEL N Termini Promote Export of RREX2-TM (A–C) Images of live P. falciparum parasites expressing RREX2-TM fused with the mature N termini of PEXEL proteins (A), GBP87–106-RCe-TM (B), or RREX2-TM fused with the mature N termini of nonexported secretory proteins (C). The position of the appended region in the original protein is shown above each panel.(D) Images of live P. falciparum parasites expressing truncated GBP fused to GFP (GBP, top) or GBP-GFP with the mature N terminus of SERA7Q23A after the PEXEL (GBP/SERA, bottom). Although these N termini contain only the last of the conserved PEXEL residues (PEXEL position 5) and the nonconserved position 4, they promoted export of the reporter into the host cell (A). GFP fluorescence was detected in the erythrocyte cytosol and the Maurer’s clefts.Grüring C, Heiber A, Kruse F, Flemming S, Franci G, Colombo SF, Fasana E,Schoeler H, Borgese N, Stunnenberg HG, Przyborski JM, Gilberger TW, Spielmann T. Uncovering common principles in protein export of malaria parasites. Cell Host Microbe. 2012 12(5):717-29.
See original on MMP
Certain non-canonical PEXEL/HT variants are functional in a GBP reporter. A. Fluorescence microscopy. The structure of full-length GBP is indicated at the top; the schematic structure of the reporter proteins is depicted on the left of representative images of iRBCs. The ER-signal is shaded blue, the (non-)canonical PEXEL/HT is shaded black and GFP is shaded green. Putative cleavage sites are indicated by an arrow (blue: SP cleavage site; black: Plasmepsin V cleavage site. Fluorescence microscopy analyses indicated export of GBP-RILAE-GFP, GBP-RIIAE-GFP, GBPKILAE-GFP and GBP-HILAE-GFP, whereas GBP-KIIAEGFP, GBP-HIIAE-GFP and the control reporter (GBP- AILAE-GFP) were less efficiently exported.Schulze J, Kwiatkowski M, Borner J, Schlüter H, Bruchhaus I, Burmester T, Spielmann T, Pick C. The Plasmodium falciparum exportome contains non-canonical PEXEL/HT proteins. Mol Microbiol. 2015 Apr 8. [Epub ahead of print]
See original on MMP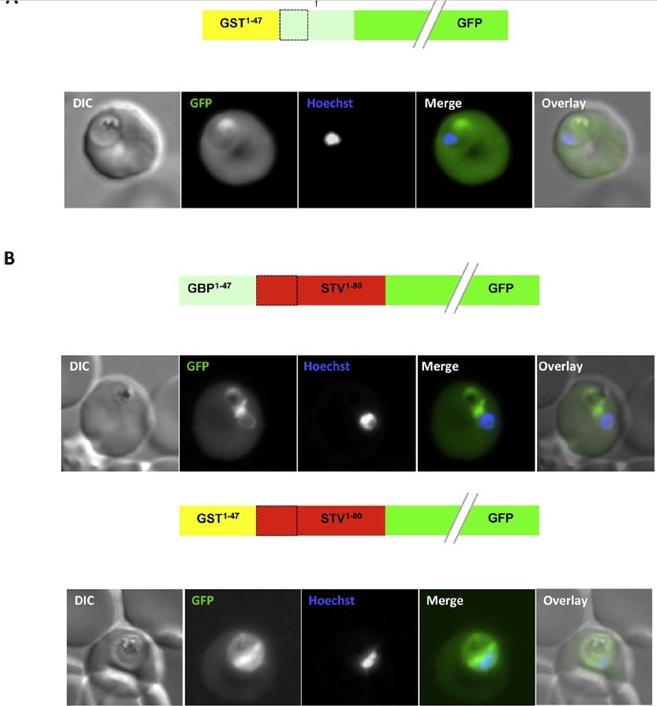
An artificial N-terminal extension has no effect on protein export efficiency. (B) Addition of the GBP130 N-terminal extension or an artificial extension to a canonical signal peptide has little effect on protein export efficiency. A schematic of each construct is shown above the respective images. All images are of live cells. DIC, differential interference contrast; GFP, green fluorescent protein; Hoechst, nuclear staining. In merge and overlay: green, GFP; blue, Hoechst.Meyer C, Barniol L, Hiss JA, Przyborski JM. The N-terminal extension of the P. falciparum GBP130 signal peptide is irrelevant for signal sequence function. Int J Med Microbiol. 2017 S1438-4221(17)30195-9.
See original on MMP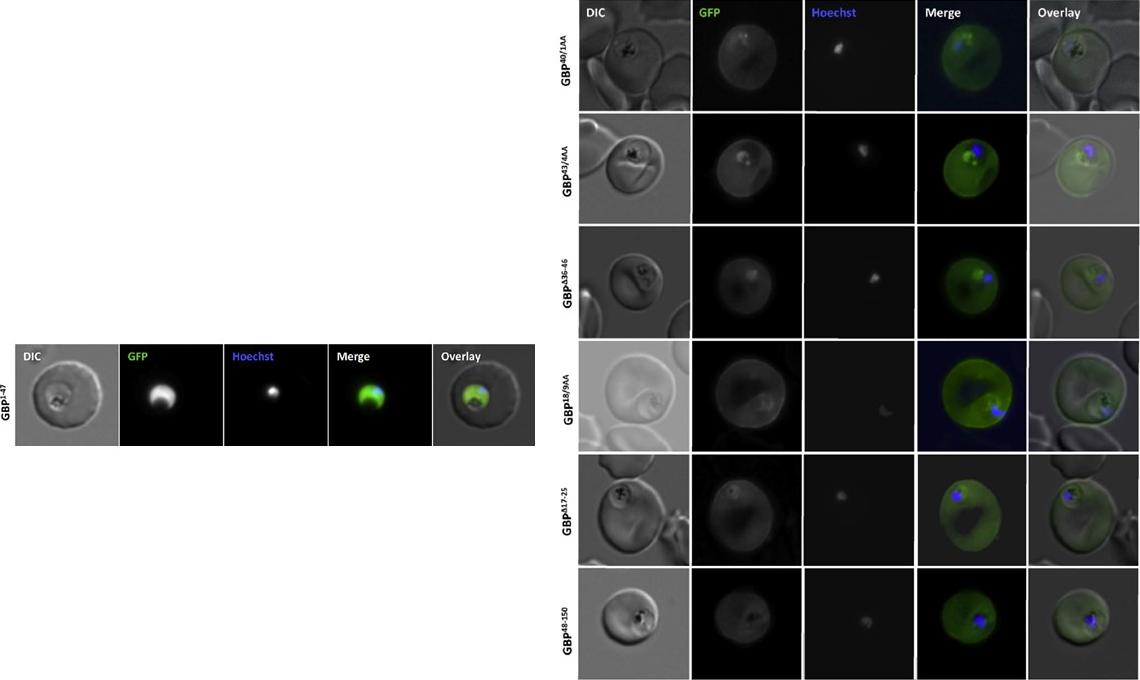
Analysis of the GBP130 N-terminal region. Live cell imaging (lower panel) of parasites expressing GBP1-47 fused to GFP reveals that this region contains no targeting information. Right panel: Mutations within, or deletion of the transition areas have no effect on protein entry to the secretory system. DIC, differential interference contrast; GFP, green fluorescent protein; Hoechst, nuclear staining. In merge and overlay: green, GFP; blue, HoechstMeyer C, Barniol L, Hiss JA, Przyborski JM. The N-terminal extension of the P. falciparum GBP130 signal peptide is irrelevant for signal sequence function. Int J Med Microbiol. 2017 S1438-4221(17)30195-9.
See original on MMP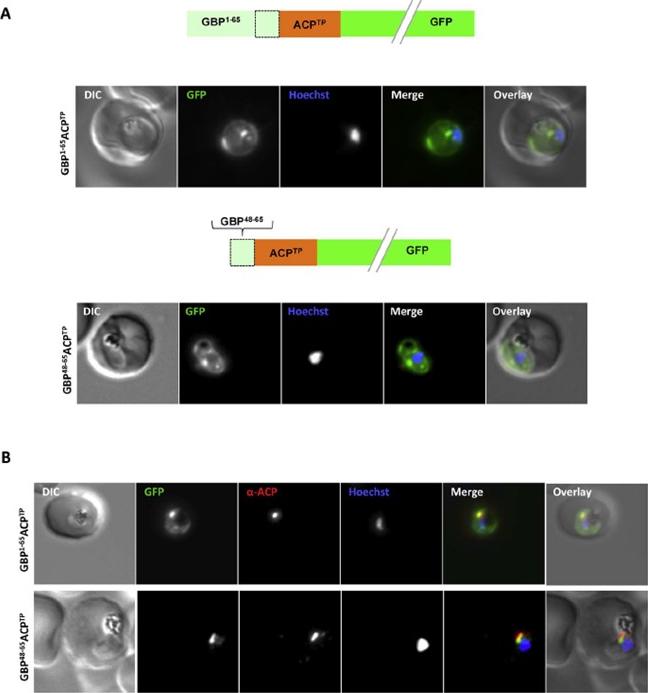
Both extended and shortened GBP130 signal peptides can replace the canonical signal peptide of an apicoplast protein. (A) Live cell imaging (B) Immunofluorescence. A schematic of each construct is shown above the respective images. DIC, differential interference contrast; GFP, green fluorescentprotein; Hoechst, nuclear staining. In merge and overlay: green, GFP; blue, Hoechst; red, anti-ACP. Traffic to the apicoplast is directed by an Nterminal canonical signal peptide which mediates entry into the secretory pathway, followed by a so-called transit peptide which is required for transport across the remaining apicoplast membranes. We wished to understand whether recessed signal peptides may target a sub-domain of the ER responsible for export to the host cell, or whether all proteins enter the secretory system at the same point. We thus generated parasites expressing the first 65aa of GBP130 fused to a transit peptide derived from acyl carrier protein (ACP). Additionally, we also fused the transit peptide to the C- domain of GBP130 (aa48-65). In both cases we could observe a mixed fluorescent phenotype, with a small bright “dot” within the parasite, and additionally a circular rim of fluorescencesurrounding the parasite (A, upper and lower panels). Analysisusing antisera against the apicoplast marker ACP verified the dotted fluorescence as the apicoplast. The fluorescence surrounding the parasite probably represents the lumen of the parasitophorous vacuole, the default target of secretory proteins lacking any further targeting signals.Meyer C, Barniol L, Hiss JA, Przyborski JM. The N-terminal extension of the P. falciparum GBP130 signal peptide is irrelevant for signal sequence function. Int J Med Microbiol. 2017 S1438-4221(17)30195-9.
See original on MMP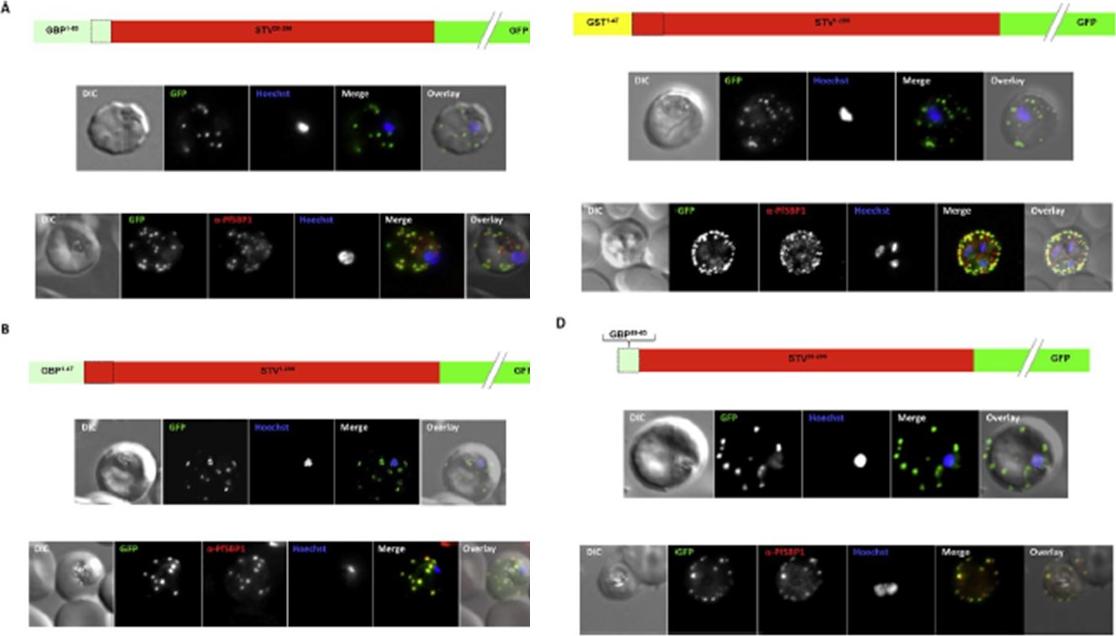
Effect of signal peptide type on export of a membrane protein. A schematic of eachconstruct is shown above the respective images. Upper panels show live cells, lowerpanels fixed cells. DIC, differential interference contrast; GFP, green fluorescent protein; Hoechst, nuclear staining. In merge and overlay: green, GFP; blue, Hoechst; red, anti-SBP1. Having excluded an important role for N-terminal extensions in the export of soluble proteins, we wished to study whether the C- domain from GBP130 is alone capable of driving entry of a membrane protein to the secretory pathway. For this reason we expressed a construct composed of the C- domain of GBP130 (aa48-65) fused to STEVOR lacking the endogenous signal peptide. Once again we observed a high level of export to the Maurer’s clefts, and co-localisation with PfSBP1Meyer C, Barniol L, Hiss JA, Przyborski JM. The N-terminal extension of the P. falciparum GBP130 signal peptide is irrelevant for signal sequence function. Int J Med Microbiol. 2017 S1438-4221(17)30195-9.
See original on MMPMore information
| PlasmoDB | PF3D7_1016300 |
| GeneDB | PF3D7_1016300 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | PF10_0159 |
| Orthologs | |
| Google Scholar | Search for all mentions of this gene |