PF3D7_0532400 lysine-rich membrane-associated PHISTb protein (LyMP)
Disruptability [+]
| Species | Disruptability | Reference | Submitter |
|---|---|---|---|
| P. falciparum 3D7 |
Possible |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen |
Mutant phenotypes [+]
| Species | Stage | Phenotype | Reference | Submitter |
|---|---|---|---|---|
| P. falciparum 3D7 | Asexual |
Possible |
26916885 (Conditional)
strongly reduced adhesion to CD36 |
Jan D. Warncke, Swiss Tropical and Public Health Institute |
Imaging data (from Malaria Metabolic Pathways)

Localization of LyMP in iRBCs. A) Localization of LyMP in trophozoites, with either anti-LyMP (green) or anti-HA (red) showing that LyMP is exported into the RBC cytosol. 4’,6’-Diamidino-2-phenylidole (DAPI) is a nuclear stain (blue). B) Colocalization of LyMP (green) with a variety of iRBC markers (red) that are indicated in the panel. DAPI is a nuclear stain (blue). C) High-resolution structured illumination fluorescence microscopy (OMX) with anti-LyMP (green) and either anti-KAHRP, anti-MESA, or anti-spectrin (red) colocalizations. Corresponding overlay images are shown for all. D) Immunoelectron micrograph of gold-labeled Ha-tagged parasites. Red asterisks indicate knobs and the arrow indicates gold labeling of LyMP at the RBC membrane skeleton. P, parasite; MC, Maurer’s clefts. LyMP showed a pattern of distribution similar to that for MESA, KAHRP. LyMP did not consistently colocalize with MCs or to other punctate structures previously identified in the iRBC cytosol such as those seen for FIKK4.2 (R45) or for J-dots (KAHsp40).Proellocks NI, Herrmann S, Buckingham DW, Hanssen E, Hodges EK, Elsworth B, Morahan BJ, Coppel RL, Cooke BM. A lysine-rich membrane-associated PHISTb protein involved in alteration of the cytoadhesive properties of Plasmodium falciparum-infected red blood cells. FASEB J. 2014 Apr 4. [Epub ahead of print]
See original on MMP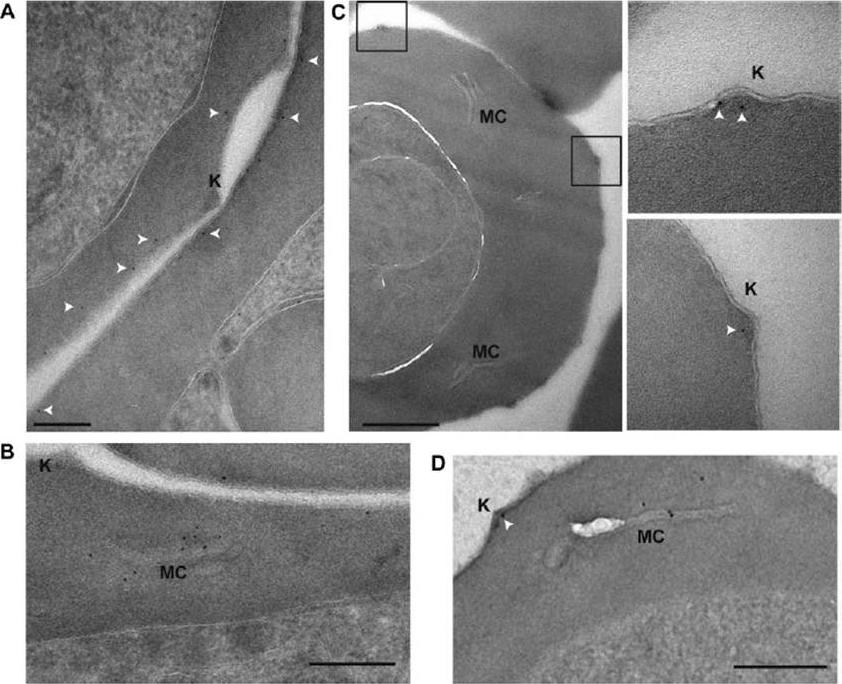
PFE1605w localizes to knobs. Shown here are postembedding immunoelectron microscopy images of iRBCs expressing PFI1780w-GFP (A, B), PFE1605w-HA (C), and PFE1605w-GFP (D). Insets: enlarged views of boxed sections. White arrows, 5 nM gold. Knobs (K) and Maurer’s clefts (MC) are labeled. Scale bars = 200 nm (A); 500 nm (C); 250 nm (B, D). Gold labeling of PFI1780w-GFP was found in proximity to the iRBC membrane but was clearly absent from knobs (A) and frequently at Maurer’s clefts (B). In contrast, PFE1605w-3xHA (C) and PFE1605w-GFP were clearly localized in knobs, and in trophozoite stage parasites, PFE1605w-GFP was also frequently found in Maurer’s clefts (D), suggesting that it localizes underneath the iRBC membrane, whereas PFE1605w localizesto knobs, but both proteins seem to be transiently transported through the Maurer’s clefts.Oberli A, Slater LM, Cutts E, Brand F, Mundwiler-Pachlatko E, Rusch S, Masik MF, Erat MC, Beck HP, Vakonakis I. A Plasmodium falciparum PHIST protein binds the virulence factor PfEMP1 and comigrates to knobs on the host cell surface. FASEB J. 2014 [Epub ahead of print] PMID:
See original on MMP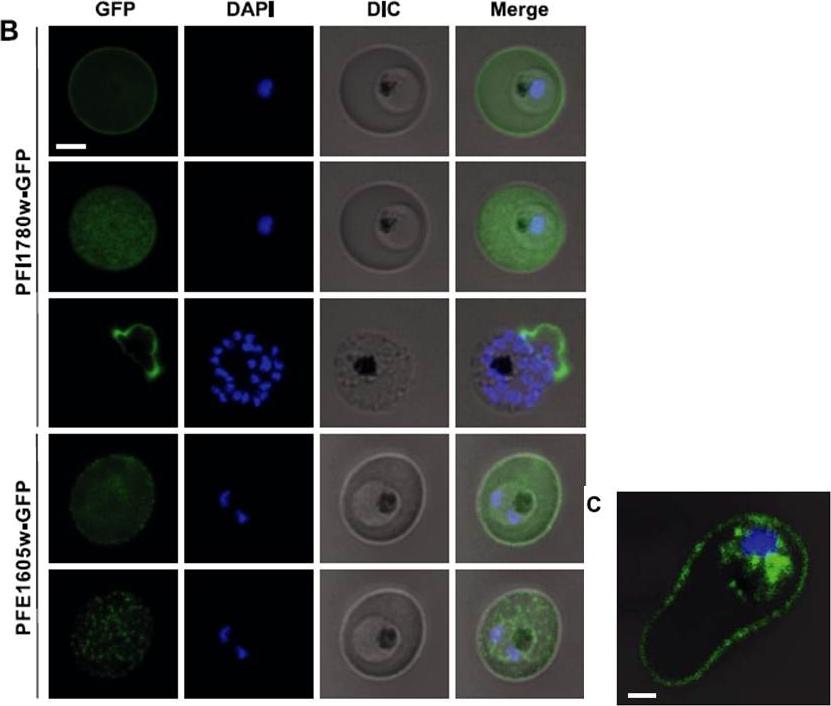
Live cell imaging of 3D7 parasites expressing PFI1780w-GFP and PFE1605w-GFP. For panels 2 and 5, the focal plane of the GFP signal was set on the surface of the iRBC, and the DAPI/differential interference contrast (DIC) microscopy signal was kept on the previous focal plane. Nuclei were stained with DAPI. Scale bar=2 mm. Panel 3 shows a rare schizont with already ruptured erythrocyte membrane. C) Confocal immunofluorescence analysis of 3D7 parasite expressing PFI1780w-GFP. Merge image of GFP/DAPI/DIC channels representing stack 32 of 69 in total. Scale bar =1mm. PFI1780w-GFP was exported to the iRBC cytosol but full-lengthPFI1780w-GFP additionally revealed fluorescence at the periphery of iRBCs (Fig. 2B; top panel), suggesting a localization close or adjacent to the erythrocyte membrane (panel 2). Immunofluorescent 3-dimensional reconstructions of fixed iRBCs with PFI1780w-GFPexpressing parasites showed focal fluorescence in parasite cytosol and uniform luorescence around the biconcave rim of the iRBC (C).Oberli A, Slater LM, Cutts E, Brand F, Mundwiler-Pachlatko E, Rusch S, Masik MF, Erat MC, Beck HP, Vakonakis I. A Plasmodium falciparum PHIST protein binds the virulence factor PfEMP1 and comigrates to knobs on the host cell surface.FASEB J. 2014 [Epub ahead of print]
See original on MMP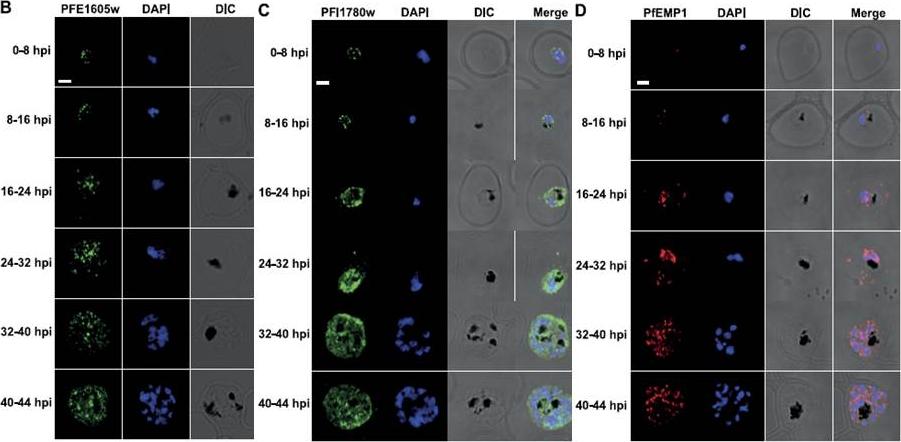
PFE1605w is cotranslocated with PfEMP1. Confocal immunofluorescence analysis of tightly synchronized 3D7 parasites. Samples were collected at 8-h intervals and stained with antibodies recognizing PFE1605w (B), PFI1780w (C), or PfEMP1 (D) and costained with DAPI. Scale bars = 2 mm. DIC, differential interference contrast microscopy. PfEMP1 is known to display a necklace of beads pattern at the parasite surface at ~8–11 hpi which was confirmed (D) and was similar to the pattern observed for PFE1605w (B). Both proteins transiently associated with Maurer’s clefts at ~16–24 hpi (D) before being transferred to the iRBC membrane, suggesting cotransport of PFE1605w with PfEMP1. In contrast to PFE1605w, PFI1780w was found within the parasite cytosol until ~24–32 hpi with limited focal fluorescence in the iRBC cytosol (C). In schizonts, PFI1780w was exported to the iRBC surface as shown in live cell imaging (B) with no distinct intermediate locations.Oberli A, Slater LM, Cutts E, Brand F, Mundwiler-Pachlatko E, Rusch S, Masik MF, Erat MC, Beck HP, Vakonakis I. A Plasmodium falciparum PHIST protein binds the virulence factor PfEMP1 and comigrates to knobs on the host cell surface. FASEB J. 2014 [Epub ahead of print]
See original on MMP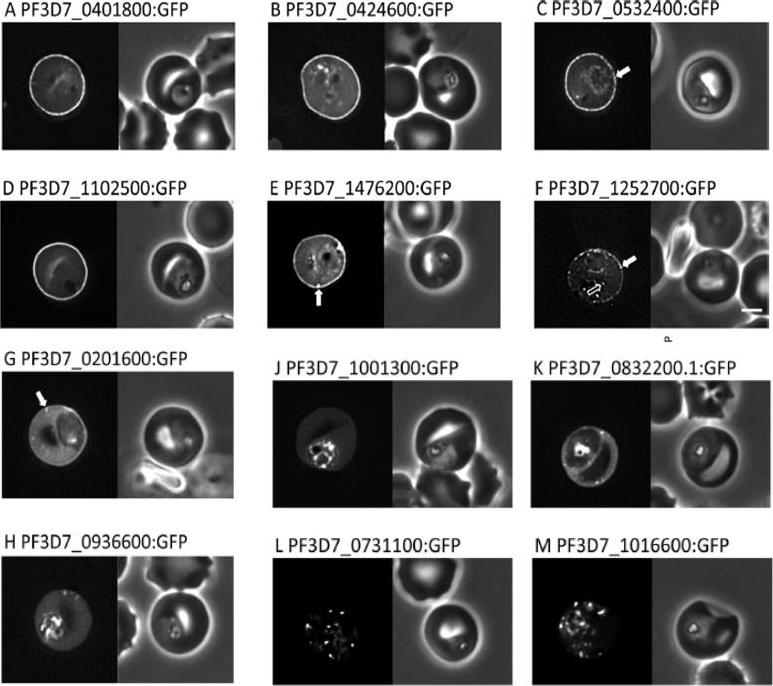
Localisation of PHIST:GFP proteins. The left- and right-hand images show GFP localisation and a phase contrast image, respectively. White filled arrows: peripheral GFP puncta; White unfilled arrow: GFP puncta in host erythocyte cytosol. Scale bar, 2 mm. GFP-tagged PF3D7_0401800, PF3D7_0424600, PF3D7_0532400, PF3D7_1102500 and PF3D7_1476200 were all exported to the host cell and displayed a striking localisation at the edge of the host erythrocyte (1A–E), PF3D7 1252700 was clearly peripheral in the host erythrocyte (F). PF3D7_0201600 (G) shows weak accumulation at the erythrocyte periphery, but the majority of the protein was localised in the RBC cytosol. PF3D7_0936600 was localised in the erythrocyte cytosol, not at the host cell periphery (H).Tarr SJ, Moon RW, Hardege I, Osborne AR. A conserved domain targets exported PHISTb family proteins to the periphery of Plasmodium infected erythrocytes. Mol Biochem Parasitol. 2014 196(1):29-40 PMID: 25106850
See original on MMP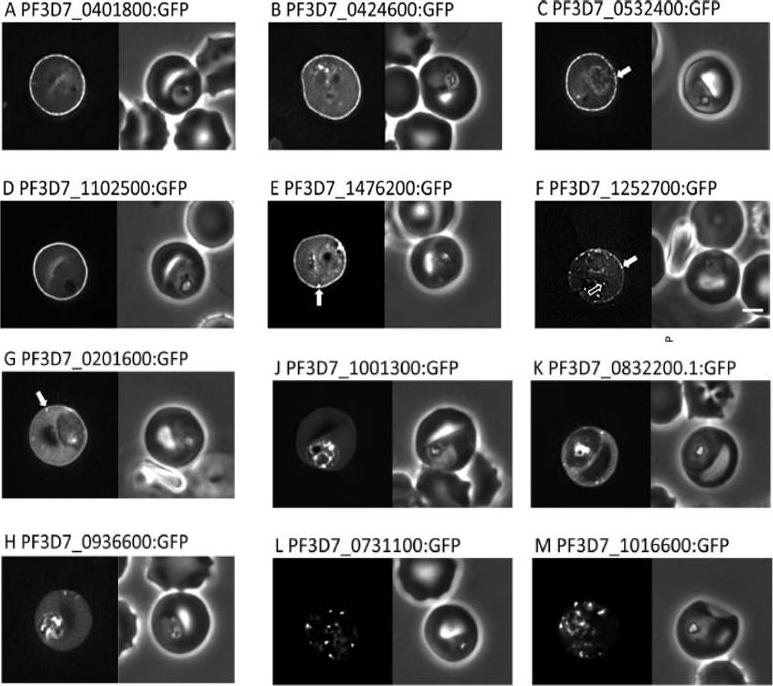
Localisation of PHIST:GFP proteins. The left- and right-hand images show GFP localisation and a phase contrast image, respectively. White filled arrows: peripheral GFP puncta; White unfilled arrow: GFP puncta in host erythocyte cytosol. Scale bar, 2 mm. GFP-tagged PF3D7_0401800, PF3D7_0424600, PF3D7_0532400, PF3D7_1102500 and PF3D7_1476200 were all exported to the host cell and displayed a striking localisation at the edge of the host erythrocyte (1A–E), PF3D7 1252700 was clearly peripheral in the host erythrocyte (F). PF3D7_0201600 (G) shows weak accumulation at the erythrocyte periphery, but the majority of the protein was localised in the RBC cytosol. PF3D7_0936600 was localised in the erythrocyte cytosol, not at the host cell periphery (H).Tarr SJ, Moon RW, Hardege I, Osborne AR. A conserved domain targets exported PHISTb family proteins to the periphery of Plasmodium infected erythrocytes. Mol Biochem Parasitol. 2014 196(1):29-40
See original on MMP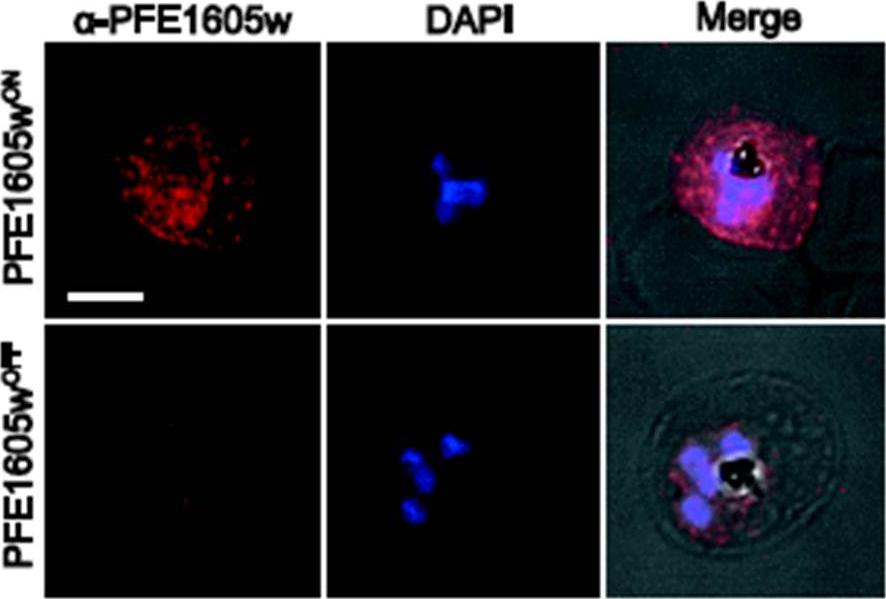
Conditional depletion of PFE1605w (A) Confocal immunofluorescence and Western blot. analysis of synchronized 3D7 parasites expressing endogenous PFE1605w as a C-terminally tagged DD fusion protein grown for 96 hours in presence (PFE1605wON) or absence (PFE1605wOFF) of 625 nM Shield-1. Nuclei were stained with DAPI. Scale bar = 3 μm. Immunofluorescence assays showed the expected localisation of tagged PFE1605w at Maurer’s clefts and at the iRBC membrane. In parasites grown for 96 hours without Shield-1(PFE1605wOFF), PFE1605w levels were highly reduced and residual protein was only visible within the parasite. Parasites grown for 96 hours under the presence of Shield-1 (PFE1605wON) displayed normal levels and distribution of PFE1605w.Oberli A, Zurbrügg L, Rusch S, Brand F, Butler ME, Day JL, Cutts EE, Lavstsen T, Vakonakis I, Beck HP. Plasmodium falciparum PHIST Proteins Contribute to Cytoadherence and Anchor PfEMP1 to the Host Cell Cytoskeleton. Cell Microbiol. 2016 Feb 24. [Epub ahead of print]
See original on MMP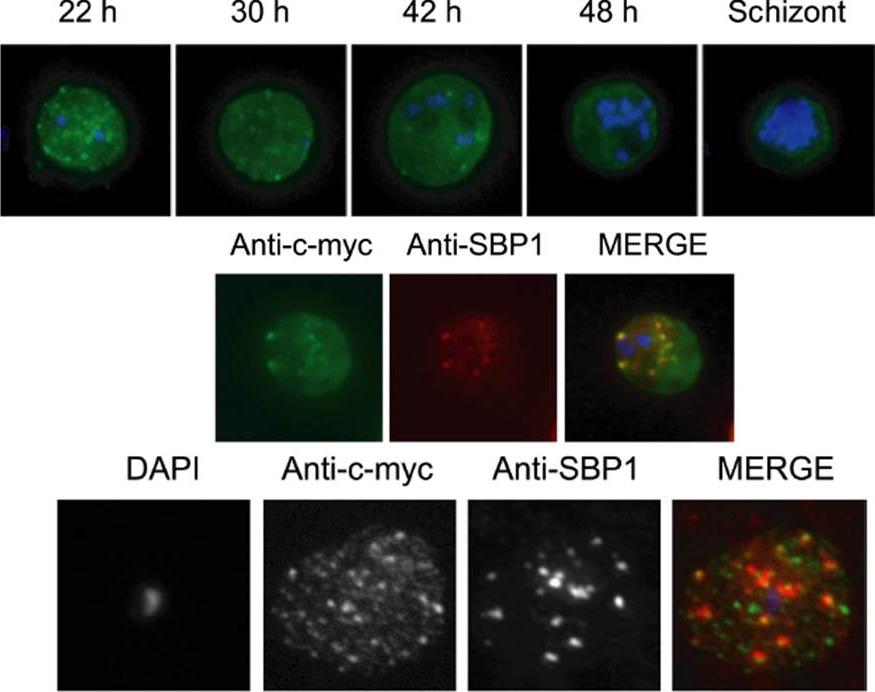
Immunolocalization of c-myc epitope-tagged PFE1605w PHIST protein within infected erythrocytes. A) Synchronized parasites were harvested at different time points after Percoll-sorbitol purification, air-dried and fixed with ice-cold methanol. Epitope-tagged proteins were stained with anti-c-myc monoclonal antibodies that were conjugated with FITC. Nuclei were stained with DAPI (blue). The abbreviations used to describe parasite stages are as follows: 22 h, late ring stage; 30 h, early-trophozoite stage; 42 h, mid-trophozoite stage; 48 h, late trophozoite stage; and schizont stage. B) Co-localization was assayed using rabbit polyclonal SBP1 followed by goat anti-rabbit IgG conjugated with Alexa 594 (red). C) The pattern of expression of c-myc epitope tagged PFE1605w PHIST protein is dependent on the fixation protocol. When infected erythrocytes are fixed in solution with 1% paraformaldehyde, 0.075% glutaraldehyde, then PHIST protein shows a broader punctate expression than the Maurer’s cleft marker, SPB1. Moreira CK, Naissant B, Coppi A, Bennett BL, Aime E, Franke-Fayard B, Janse CJ, Coppens I, Sinnis P, Templeton TJ. The Plasmodium PHIST and RESA-Like Protein Families of Human and Rodent Malaria Parasites. PLoS One. 2016 Mar 29;11(3):e0152510.
See original on MMP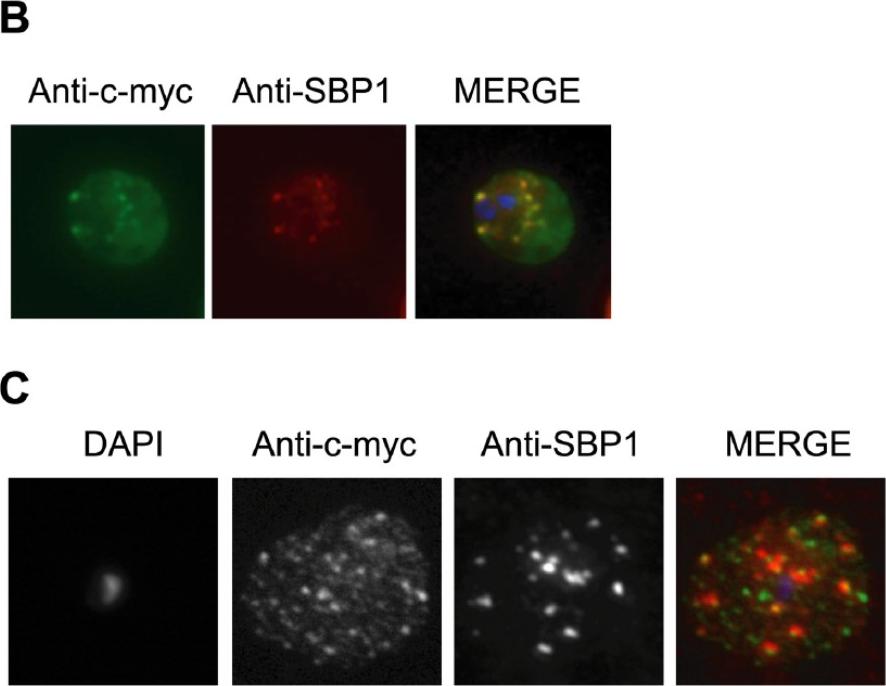
Immunolocalization of c-myc epitope-tagged PFE1605w PHIST protein within infected erythrocytes. B) Co-localization was assayed using rabbit polyclonal SBP1 followed by goat anti-rabbit IgG conjugated with Alexa 594 (red). C) The pattern of expression of c-myc epitope tagged PFE1605w PHIST protein is dependent on the fixation protocol. When infected erythrocytes are fixed in solution with 1% paraformaldehyde, 0.075% glutaraldehyde, then PHIST protein shows a broader punctate expression than the Maurer’s cleft marker, SBP1. At the late ring stage, PHIST proteins were observed exported to the erythrocyte cytoplasm, at least in partial co-localization with the marker of Maurer’s clefts, SBP1 (B). PFE1600w was abundantly observed in the ER of late stage parasites (data not shown); and it is possible that this localization represents aberrant trafficking due to over-expression of the transcript and thereby protein, perhaps due to the use of a heterologous promoter. The localization to the erythrocyte cytoplasm validates trafficking that is predicted by the presence of a PEXEL/HT motif.Moreira CK, Naissant B, Coppi A, Bennett BL, Aime E, Franke-Fayard B, Janse CJ, Coppens I, Sinnis P, Templeton TJ. The Plasmodium PHIST and RESA-Like Protein Families of Human and Rodent Malaria Parasites. PLoS One. 2016 11(3):e0152510
See original on MMPMore information
| PlasmoDB | PF3D7_0532400 |
| GeneDB | PF3D7_0532400 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | MAL5P1.315, PFE1605w |
| Orthologs | |
| Google Scholar | Search for all mentions of this gene |