PF3D7_0423500 glideosome associated protein with multiple membrane spans 2 (GAPM2)
Disruptability [+]
| Species | Disruptability | Reference | Submitter |
|---|---|---|---|
| P. falciparum 3D7 |
Possible |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen |
Mutant phenotypes [+]
| Species | Stage | Phenotype | Reference | Submitter |
|---|---|---|---|---|
| P. falciparum 3D7 | Asexual |
Invasion defect |
https://www.researchsquare.com/article/rs-87875/v1
(Knock down)
\'fPhIP and PfGAPM2 depleted parasites revealed abrogation of reorientation/gliding, although initial attachment with human RBCs was not affected in these knock-down parasites.\' |
Theo Sanderson, Francis Crick Institute |
Imaging data (from Malaria Metabolic Pathways)
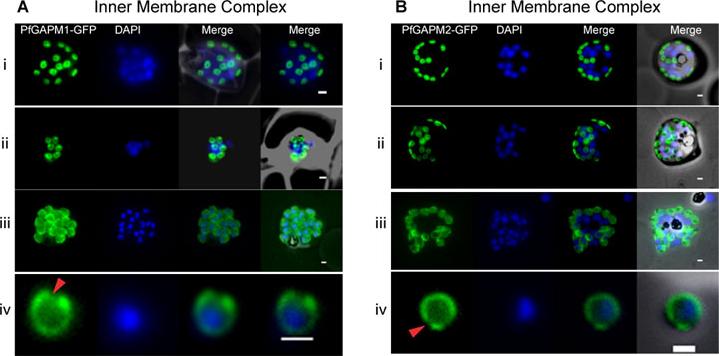
Live cell imaging of GFP tagged GAPMs (Glideosome Associated Protein with Multiple membrane spans) in Plasmodium falciparum indicate that they localize to the inner membrane complex. Developmental series of fluorescent images of live P. falciparum schizonts expressing PfGAPM1-GFP (A) or PfGAPM2-GFP (B) fusion proteins showing typical inner membrane complex morphology. During early schizogony, PfGAPM proteins are apparent as one or two small discs associated with each nucleus (i). Each disc is perforated by one or two small holes that may correspond to the future apical tip of the merozoite (ii). As the parasites mature the discs grow, ultimately encapsulating the mature merozoite except at the apical tip (red arrowhead) (iii,iv). Bullen HE, Tonkin CJ, O'Donnell RA, Tham WH, Papenfuss AT, Gould S, Cowman AF, Crabb BS, Gilson PR. A novel family of apicomplexan glideosome associated proteins with an inner-membrane anchoring role. J Biol Chem. 2009 284(37):25353-63.
See original on MMP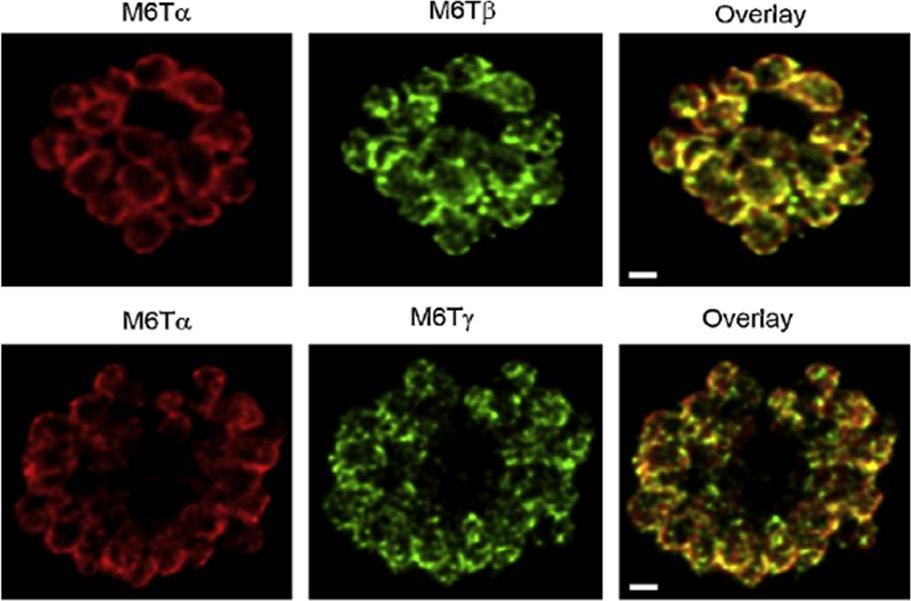
PfM6T proteins localize to the merozoite IMC. Confocal immunofluorescence images demonstrating location of each paralog, as indicated above each image. Images show a separate schizont-infected erythrocyte containing many merozoites; overlay of the red and green channels (rightmost column) shows colocalization of PfM6Ta with each other paralog. Red and green images reflect detection using specific antibodies raised in rabbits and mice, respectively. White scale bars represent 1mm.Rayavara K, Rajapandi T, Wollenberg K, Kabat J, Fischer ER, Desai SA. A complex of three related membrane proteins is conserved on malarial merozoites. Mol Biochem Parasitol. 2009 167:135-43. Copyright Elsevier 2010
See original on MMP
Upper panel: Co-localization of the group A protein PFD1110w (anti-PFD1110w, red) with the group B alveolin PFE1285w (PFE1285w-GFP, green) emphasizes the differential nature of the two structures in the nascent IMC and their spatial relation during the ongoing schizogony (T1-3). Nuclei stained with DAPI. Scale bar 2 μm.Lower panel: A double transgenic parasite line was generated expressing a different combination of proteins, MAL13P1.130-GFP (group A) and PF10_0039-mCherry. The dynamic distribution of these two members confirmed the spatial distinction but intimate proximity of the group A and group B defined IMC sub-compartments during the early phase of IMC biogenesis.Kono M, Herrmann S, Loughran NB, Cabrera A, Engelberg K, Lehmann C, Sinha D, Prinz B, Ruch U, Heussler V, Spielman T, Parkinson J, Gilberger TW. Evolution and Architecture of the Inner Membrane Complex in Asexual and Sexual Stages of the Malaria Parasite. Mol Biol Evol. 2012 29(9):2113-32
See original on MMP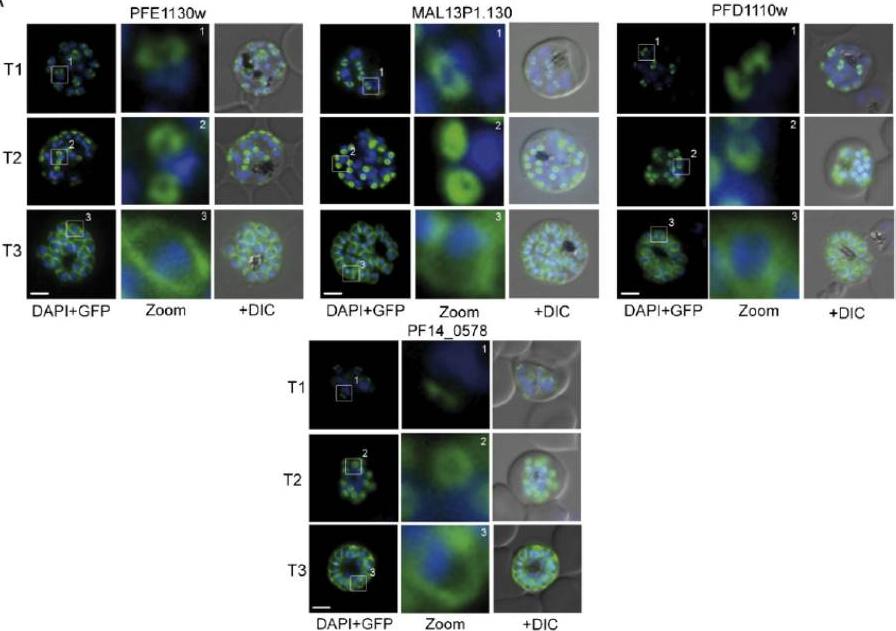
Several IMC proteins co-localize with GAP45 during schizogony. (A) Using GFP as a fluorescence tag, the TMD proteins PFE1130w, MAl13P1.130, PFD1110w, as well as PF14_0578 reveal three distinct structures during ongoing schizogony. Commencing as cramplike structures (T1), they transform to small ring-shaped formations (T2) that toward the end of schizogony expand and are then equally distributed underneath the plasma membrane (T3). Enlargement of these distinct structures are marked (white square) and shown in second row (Zoom) for each cell line.Kono M, Herrmann S, Loughran NB, Cabrera A, Engelberg K, Lehmann C, Sinha D, Prinz B, Ruch U, Heussler V, Spielmann T, Parkinson J, Gilberger TW. Evolution and architecture of the inner membrane complex in asexual and sexual stages of the malaria parasite. Mol Biol Evol. 2012 Sep;29(9):2113-32. PMID:
See original on MMP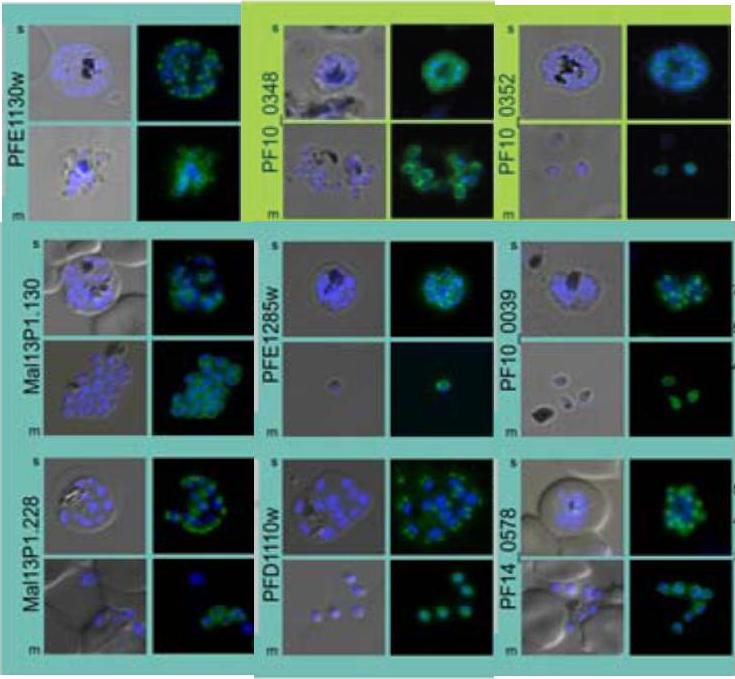
Subcellular distribution of proteins predicted to be involved in invasion. All proteins were localized in schizonts (s) and free merozoites (m) using GFP-fusion proteins and grouped according to their predominant GFP localization: surface (green), inner membrane complex (turquoise),Hu G, Cabrera A, Kono M, Mok S, Chaal BK, Haase S, Engelberg K, Cheemadan S, Spielmann T, Preiser PR, Gilberger TW, Bozdech Z. Transcriptional profiling of growth perturbations of the human malaria parasite Plasmodium falciparum. Nat Biotechnol. 2010 28(1):91-8.
See original on MMP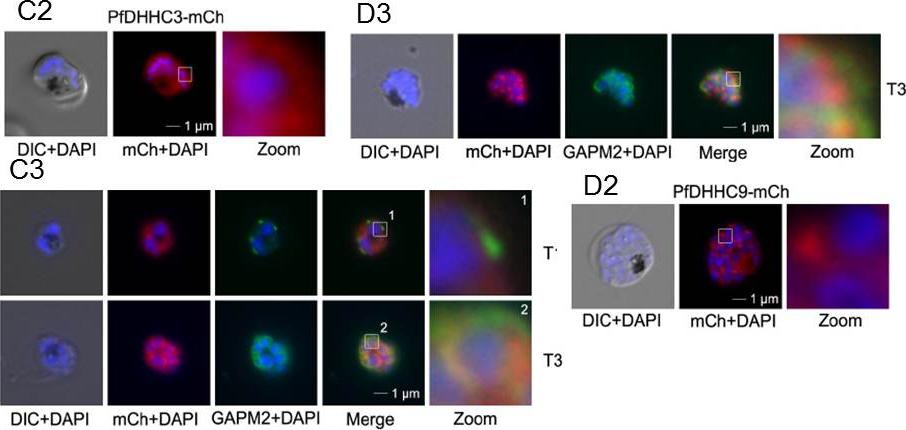
Over-expression and localization of PfDHHC3 and PFDHHC9 (PF11_0167) in late stage parasites. C. Expression of PfDHHC3-mCherry. C. Expression of PfDHHC3-mCherry. C2. Microscopic analysis located this GFP-fusion protein in the periphery of the nascent merozoites. C3. Co-localization with the IMC marker GAPM2 (anti-GAPM2, green) in fixed cells shows differential localization of PfDHHC2-mCherry with the IMC localization in early stages (T1) and colocalization in nascent merozoites (T3) consistent with plasma membrane association. Scale bar, 1 μm. D.Expression of PfDHHC9-mCherry. D2. Microscopy localized this fusion protein mainly in apical structures in the parasite. D3. Co-localization with the IMC marker GAPM2 (anti-GAPM2, green) in fixed cells shows differential localization of PfDHHC9 with the IMC (T3). Scale bar, 1 μm.Wetzel J, Herrmann S, Swapna LS, Prusty D, Peter AT, Kono M, Saini S, Nellimarla S, Wong TW, Wilcke L, Ramsay O, Cabrera A, Biller L, Heincke D, Mossman K, Spielmann T, Ungermann C, Parkinson J, Gilberger TW. The role of palmitoylation for protein recruitment to the inner membrane complex of the malaria parasite. J Biol Chem. 2014 Nov 25.
See original on MMP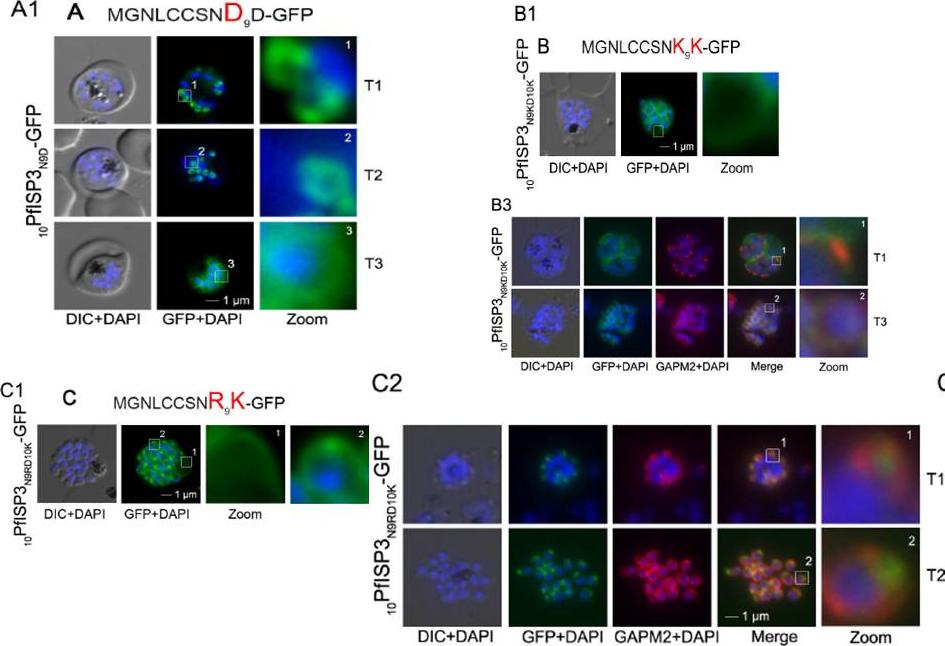
Re-direction of the IMC 10PfISP3-GFP fusion protein to the rhoptry membrane. A. Substitution of N9 with D (10PfISP3N9D-GFP, A) does not interfere with correct IMC recruitment of the GFP-fusion protein (A1). Nuclei stained with DAPI (blue). B-C. The substitution of N9 and D10 with lysine and its expression in the parasite (10PfISP3N9KD10K-GFP) leads to a peripheral localization (B1). The zoom highlights the additional association of this mutant protein with the food vacuole membrane. B2. Western blot analysis using anti-GFP antibodies. B3 Co-localization with the IMC marker GAPM2. Nascent IMC (anti-GAPM2, red) is clearly distinguishable from the peripheral associated 11PfISP1D11K-GFP in early stages (T1) and congruent in late stages (T3). C. In contrast, the substitution of N9 with arginine (10PfISP3N9RD10K-GFP) re-directs the fusion protein to cell periphery (zoom 1) and the apical pole (zoom 2). C2. Co-localization with the rhoptry marker RALP (anti-RALP, red) in fixed cells confirmed PfDHHC1-GFP IMC localization shown here in an early stage of TMC biogenesis (T1). Wetzel J, Herrmann S, Swapna LS, Prusty D, Peter AT, Kono M, Saini S, Nellimarla S, Wong TW, Wilcke L, Ramsay O, Cabrera A, Biller L, Heincke D, Mossman K, Spielmann T, Ungermann C, Parkinson J, Gilberger TW. The role of palmitoylation for protein recruitment to the inner membrane complex of the malaria parasite. J Biol Chem. 2014 Nov 25. PMID:
See original on MMP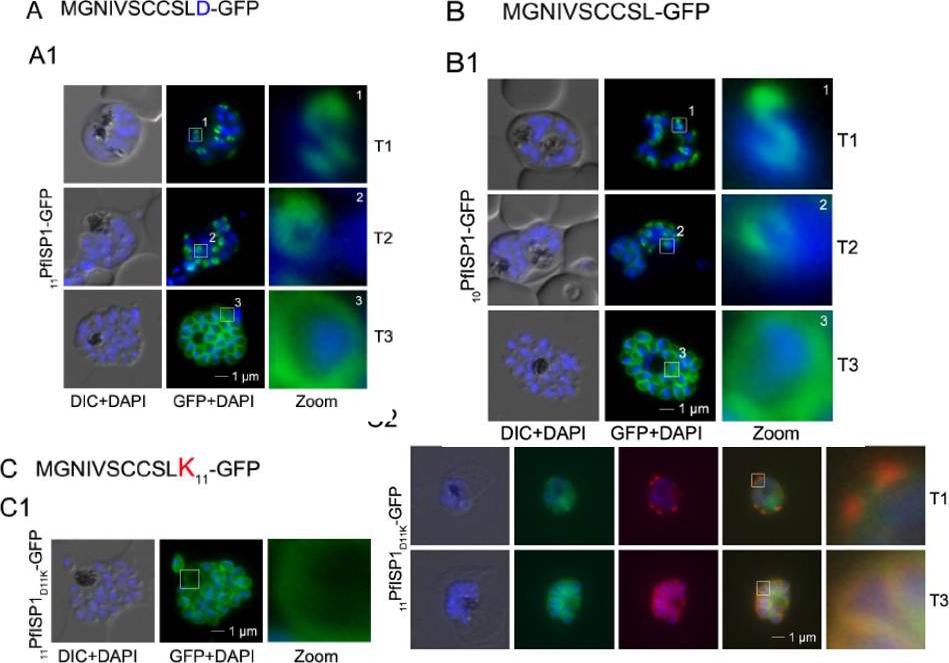
Mutational analysis of the minimal sequence requirements for IMC association of PfISP1. AB. Expression of the first 11 (11PfSP1-GFP, A) or 10 (10PfISP1-GFP, B) amino acids are sufficient to direct GFP to the IMC (A1-B1). C. Substitutionof D11 with lysine (11PfISP1D11K-GFP, C) abolishes IMC targeting (C1) and leads to peripheral localization. The zoom highlights its additional association with the food vacuole membrane consistent with plasma membrane association. C2. Co-localization with the IMC marker GAPM2. Nascent IMC (anti-GAPM2, red) is clearly distinguishable from the peripheral associated 11PfISP1D11K-GFP in early stages (T1) and congruent in late stages (T3). Wetzel J, Herrmann S, Swapna LS, Prusty D, Peter AT, Kono M, Saini S, Nellimarla S, Wong TW, Wilcke L, Ramsay O, Cabrera A, Biller L, Heincke D, Mossman K, Spielmann T, Ungermann C, Parkinson J, Gilberger TW. The role of palmitoylation for protein recruitment to the inner membrane complex of the malaria parasite. J Biol Chem. 2014 Nov 25.
See original on MMP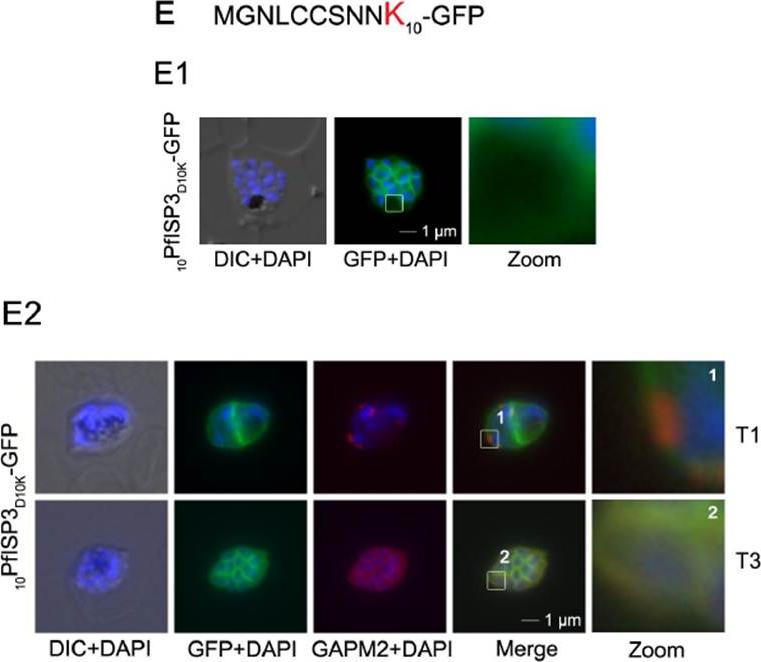
Mutational analysis of the minimal sequence requirements for IMC association of PfISP3. E. Substitution of D10 with a positively charged lysine re-directs the GFP-fusion protein to the periphery of the nascent merozoites. The zoom highlights the additional association with the food vacuole membrane characteristic for plasma membrane proteins (E1). Co-localization with the IMC marker GAPM2. Nascent IMC (anti-GAPM2, red) is clearly distinguishable from the peripheral associated 10PfISP3D10K-GFP in early stages (T1) and congruent in late stages (T3). Wetzel J, Herrmann S, Swapna LS, Prusty D, Peter AT, Kono M, Saini S, Nellimarla S, Wong TW, Wilcke L, Ramsay O, Cabrera A, Biller L, Heincke D, Mossman K, Spielmann T, Ungermann C, Parkinson J, Gilberger TW. The role of palmitoylation for protein recruitment to the inner membrane complex of the malaria parasite. J Biol Chem. 2014 Nov 25. PMID:
See original on MMP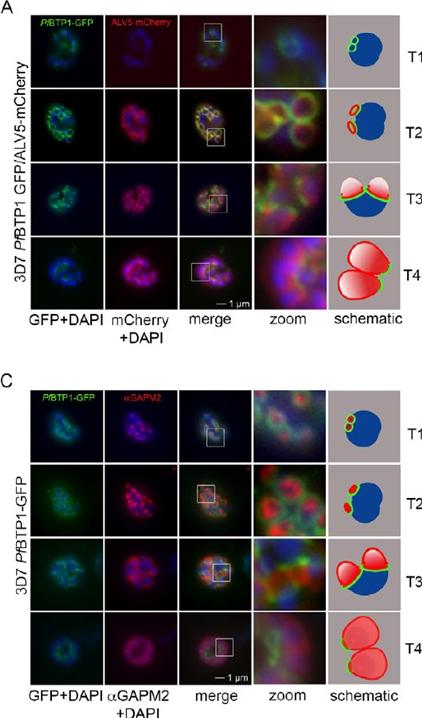
Colocalization of BTP1 with ALV5 and GAPM2 during inner membrane complex (IMC) biogenesis. (A) Colocalization of BTP1–GFP with ALV5–mCherry (red) in unfixed parasites. The initial ring structure marked by BTP1–GFP precedes the appearance of Alveolin 5 (ALV5) (T1). The emerging Alveolin-5-defined ring is associated but distinct from that marked by BTP1 (T2). During IMC expansion, BTP1 remains rim associated (T3) and, at the end of daughter cell formation, is concentrated at the basal end with ALV5 equally distributed within the IMC (T4). (C) Colocalization of BTP1–GFP with GAPM2 (red) in fixed parasites. Both proteins appeared at about the same time, marking the onset of IMC biogenesis (T1). GAPM2 stains small ring–shaped formations that are encircled by BTP1–GFP (T2). With elongation and propagation of the IMC from the apical towards the basal end of the forming daughter cell, GAPM2 is equally distributed within the forming IMC, but BTP1 is only present at the rim of the nascent IMC (T3) and, at the end of schizogony, is concentrated at the basal pole of the daughter cell (T4). Nuclei were stained with DAPI (blue). Enlargement of selected areas are marked with a white square and referred to as ‘zoom’ (a fourfold magnification). The bottom row shows a schematic representation of the spatio-temporal distribution of BTP1 and GAPM2. Kono M, Heincke D, Wilcke L, Wong TW, Bruns C, Herrmann S, Spielmann T, Gilberger TW. Pellicle formation in the malaria parasite. J Cell Sci. 2016 129(4):673-80.
See original on MMPMore information
| PlasmoDB | PF3D7_0423500 |
| GeneDB | PF3D7_0423500 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | MAL4P1.217, PFD1110w |
| Orthologs | PBANKA_0523900 , PCHAS_0524100 , PKNH_0515400 , PVP01_0532000 , PVX_090215 , PY17X_0525300 |
| Google Scholar | Search for all mentions of this gene |