PF3D7_0302500 cytoadherence linked asexual protein 3.1 (CLAG3.1)
Disruptability [+]
| Species | Disruptability | Reference | Submitter |
|---|---|---|---|
| P. falciparum 3D7 |
Possible |
21320181 (Knock down) | Theo Sanderson, Wellcome Trust Sanger Institute |
| P. falciparum 3D7 |
Possible |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen |
Mutant phenotypes [+]
| Species | Stage | Phenotype | Reference | Submitter |
|---|---|---|---|---|
| P. falciparum 3D7 | Asexual |
Possible |
32069335 (Knock down)
Few differences noted, even on a background lacking CLAG3.2 |
Theo Sanderson, Francis Crick Institute |
Imaging data (from Malaria Metabolic Pathways)
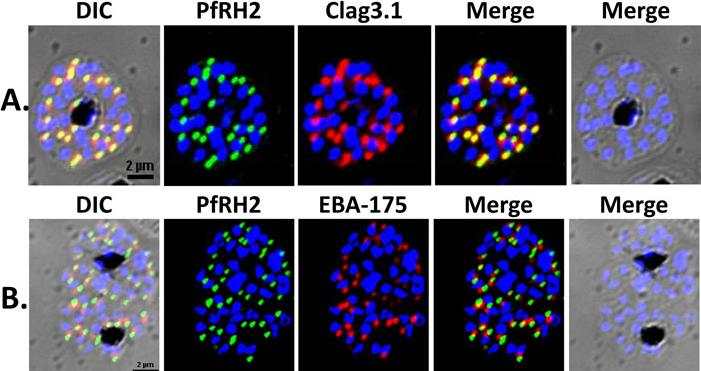
Localization of PfRH2a/b by immunofluorescence confocal microscopy. (A) 3D7 schizonts were dual labeled with anti-rPfRH240 mice sera and anti-clag3.1 rabbit sera. Mature schizonts immunolabeled with anti-rPfRH240 were stained with Alexa 488 linked anti-mouse IgG secondary antibody (green). Schizonts labeled with anti-clag3.1 rabbit sera were stained with Alexa 594 linked anti-rabbit IgG secondary antibody (red). (B) 3D7 mature schizonts were dual labeled with anti-rPfRH240 mouse sera and anti-EBA175 rabbit sera. Schizonts labeled with anti-EBA-175 antibodies were stained with Alexa 594 linked anti-rabbit IgG secondary antibody (red). PfRH2a/b co-localizes with the known rhoptry bulb marker protein, clag3.1 demonstrating that it is localized in the rhoptries, and not with the microneme marker protein, EBA-175.Sahar T, Reddy KS, Bharadwaj M, Pandey AK, Singh S, Chitnis CE, Gaur D. Plasmodium falciparum reticulocyte binding-like homologue protein 2 (PfRH2) is a key adhesive molecule involved in erythrocyte invasion. PLoS One. 2011 6(2):e17102.
See original on MMP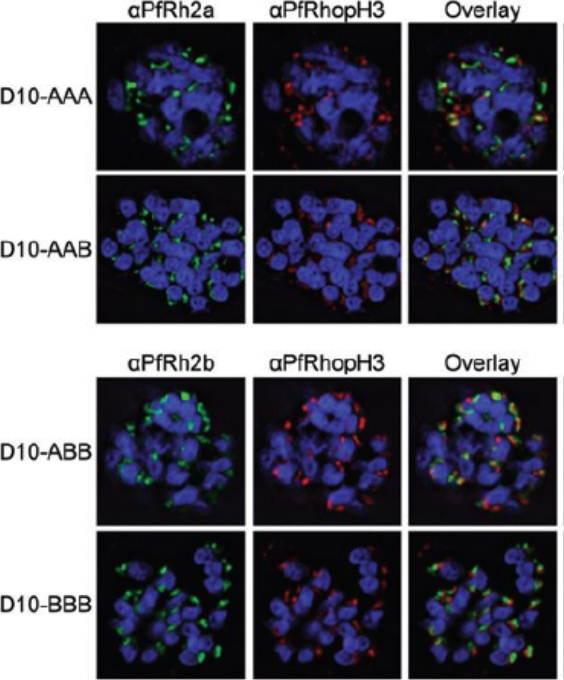
Chimeric protein localization and processing. D10-AAA, D10-AAB, D10-ABB and D10-BBB schizonts were methanol-fixed and stained with rabbit anti-PfRh2a or rabbit anti-PfRh2b, mouse anti-PfRhopH3 and DAPI. Images were obtained using deconvolution microscopy. The chimeric proteins from both D10-AAA and D10-AAB localized to the apical organelles (Fig. 3A, left panel) and in close proximity to PfRhopH3 (middle panel). Fluorescence overlays showed some direct colocalization between the PfRh2a/b proteins and PfRhopH3 (right panel). PfRh2a and PfRh2b are known to localize to the rhoptry neck whereas PfRhopH3 localizes to the rhoptry bulb.Dvorin JD, Bei AK, Coleman BI, Duraisingh MT. Functional diversification between two related Plasmodium falciparum merozoite invasion ligands is determined by changes in the cytoplasmic domain. Mol Microbiol. 2010 75:990-1006.
See original on MMP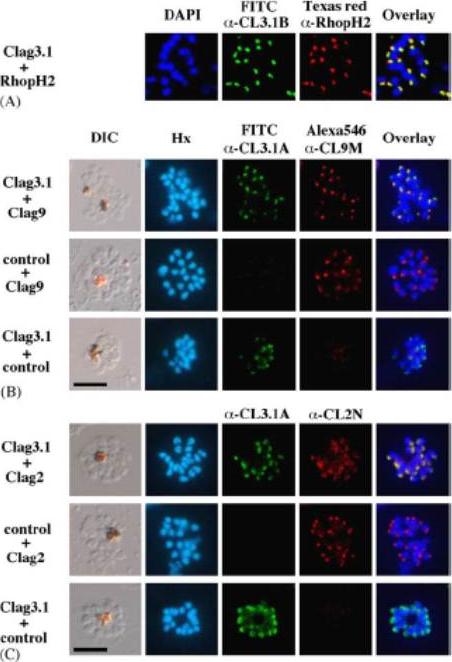
Apical expression of RhopH1/Clag members -2, -3.1, and -9 in P. falciparum segmented schizonts. Schizont-infected erythrocytes were duallabeled with a-CL3.1B and a-RhopH2 mAb 61.3 in the 3D7 parasite line (Panel A); a-CL3.1A and a-CL9M (Panel B) and a-CL3.1A and a-CL2N (Clag2) (Panel C) in Dd2 parasite line. Overlaid images are shown in the right-hand panels. All segmented schizont-stage parasites are positive for the antisera against Clag2, -3.1, and -9. Nuclei are counterstained with either DAPI or Hoechst-33342 (Hx). Scale bar represents 5 mm. Overlaid images of the double-staining for anti-Clag3.1 and anti-PfRhopH2 or anti-Clag3.1 and anti-Clag9 sera reacting with segmented schizonts showed indistinguishable patterns of localization for these proteins. Clag3.1 and Clag2 are localized within the rhoptries of segmented schizonts. Kaneko O, Yim Lim BY, Iriko H, Ling IT, Otsuki H, Grainger M, Tsuboi T, Adams JH, Mattei D, Holder AA, Torii M. Apical expression of three RhopH1/Clag proteins as components of the Plasmodium falciparum RhopH complex. Mol Biochem Parasitol. 2005 143(1):20-8.
See original on MMP
Expression of rhoptry protein CLAG3.1 on surface of P. falciparum merozoites isolated in intracellular buffer (5 mM NaCl, 140 mM KCl, 1 mM EGTA), detected by IFA using anti-CLAG3.1 rabbit sera followed by FITC-conjugated (green) anti-rabbit IgG goat sera. Nuclear DNA was counterstained with DAPI (blue). Bright field, bright field merged with DAPI staining and merged fluorescence images (DAPI and FITC) are shown. Black bar indicates 2 mm.Singh S, Alam MM, Pal-Bhowmick I, Brzostowski JA, Chitnis CE. Distinct external signals trigger sequential release of apical organelles during erythrocyte invasion by malaria parasites. PLoS Pathog. 2010 Feb 5;6(2):e1000746.
See original on MMP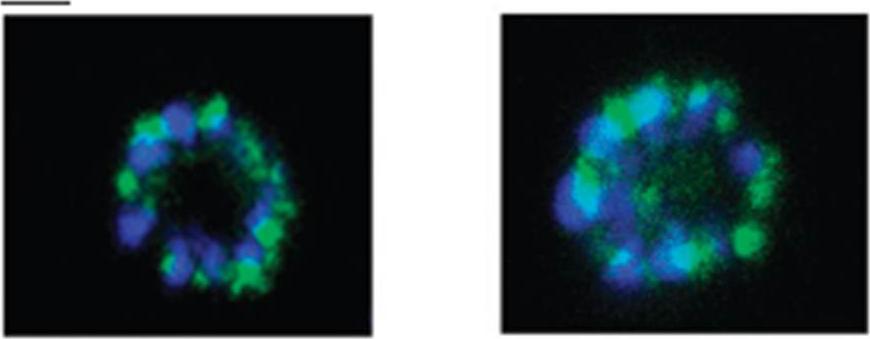
The anti-phospho-CLAG 3.1 antibody was used to determine the localization of phospho-CLAG 3.1 by immunocytochemistry of paraformaldehyde-fixed schizont stage parasites. Shown are two examples of schizont (segmenter) parasites stained with the phospho-CLAG 3.1 antibody. The samples were also stained with 4,6-diamidino-2-phenylindole (blue) to label the nuclei. The scale bar indicates 2 μM. The experiments shown are representative of at least three independent experiments. This antibody identified a punctuate localization of phospho-CLAG 3.1 consistent with rhoptry localization within merozoites of a mature schizont.Solyakov L, Halbert J, Alam MM, Semblat JP, Dorin-Semblat D, Reininger L, Bottrill AR, Mistry S, Abdi A, Fennell C, Holland Z, Demarta C, Bouza Y, Sicard A, Nivez MP, Eschenlauer S, Lama T, Thomas DC, Sharma P, Agarwal S, Kern S, Pradel G, Graciotti M, Tobin AB, Doerig C. Global kinomic and phospho-proteomic analyses of the human malaria parasite Plasmodium falciparum. Nat Commun. 2011 2:565.
See original on MMP
Ppolyclonal antibodies to a carboxy-terminal recombinant fragment conserved between the two clag3 products were prepared. Confocal microscopy with this antibody confirmed reports localizing these proteins to the host cytosol and possibly the erythrocyte membrane as well as within rhoptries of invasive merozoites.Nguitragool W, Bokhari AA, Pillai AD, Rayavara K, Sharma P, Turpin B, Aravind L, Desai SA. Malaria parasite clag3 genes determine channel-mediated nutrient uptake by infected red blood cells. Cell. 2011 145:665-77.
See original on MMP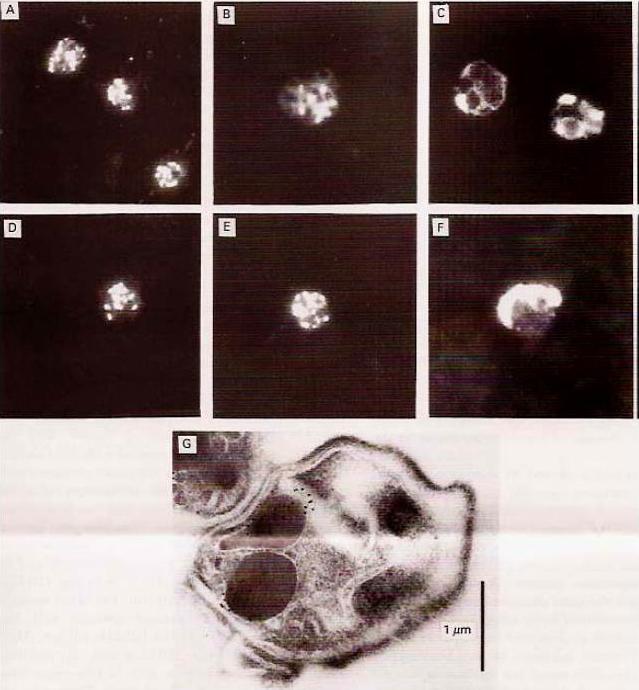
Immunofluorescence and immunoelectron microscopy on late asexual blood stages. Reaction of mAb 84 RhopH3 with (A) air-dried or (B) aceton fixed FCCE1-Niger Plasmodium flaciparum parasites or (C) air-dried P. chabaudi parasites. (D) reaction of mAb 47 with FCCE1-Niger P. falciparum parasites: reaction with mAb 109 with air-dried FCCE1-Niger P. falciparum parasites or (F) P. vivax parasites. (G) Immuno-electron microscopy using mAb 91 RhopH3. RhopH3 localized to the rhoptries.Doury JC, Bonnefoy S, Roger N, Dubremetz JF, Mercereau-Puijalon O. Analysis of the high molecular weight rhoptry complex of Plasmodium falciparum using monoclonal antibodies. Parasitology. 1994 108:269-80. Cambridge University Press © 2009.
See original on MMP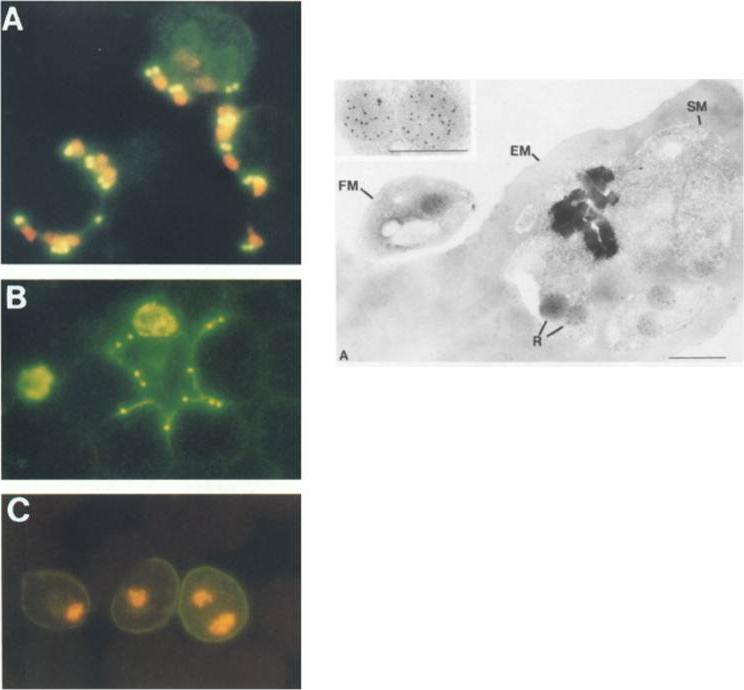
Left row: mAb 1B9 Immunofluorescence of merozoite invasion. Thin smears were incubated with mAb IB9, FITC-goat anti-mouse IgG and counterstained with ethidium bromide. (A and C) to identify parasite nucleus. (A) Merozoites attached to erythrocytes. Erythrocyte at upper right has been invaded by two merozoites. (B) Merozoites in the process of invading; rhoptry protein is discharged around erythrocyte surface. (C) Erythrocytes invaded by merozoites. Rhoptry protein is localized in infected erythrocyte membrane.Right picture: Immunoelectron microscopic localization of the rhoptry protein using mAb 1B9 and protein A-gold. (A) post-embedded immunolabeling of mature schizont-infected erythrocyte showing localization of 5-nm gold particles over rhoptries. EM, Erythrocyte membrane; SM, schizont membrane; FM, free merozoite; R, rhoptries. (Inset) High magnification of paired rhoptry organelles showing localizaltion of rhoptry antigen.Sam-Yellowe TY, Shio H, Perkins ME. Secretion of Plasmodium falciparum rhoptry protein into the plasma membrane of host erythrocytes. J Cell Biol. 1988 1061507-13.
See original on MMP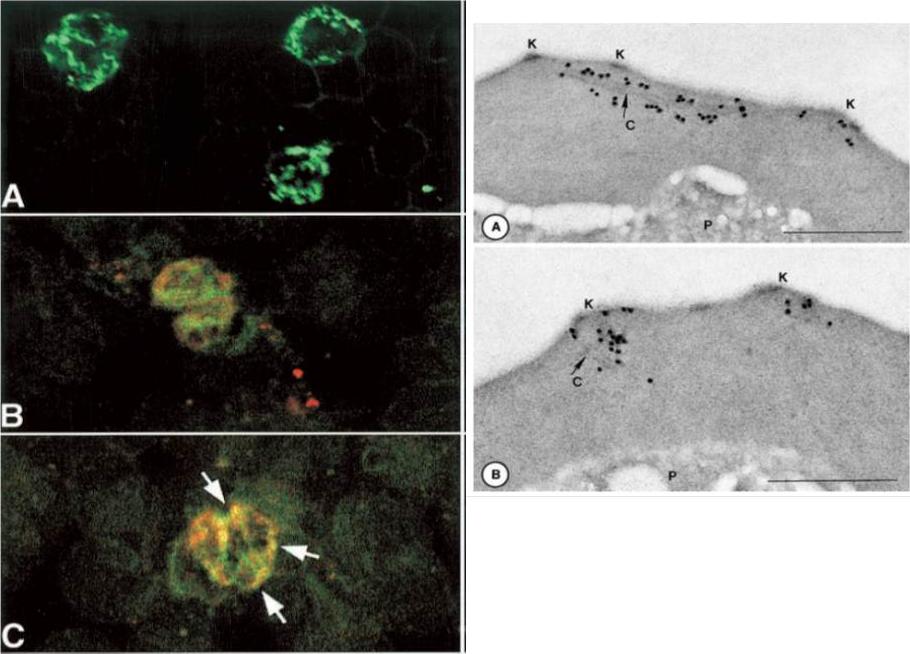
Right panel:Immunodetection of P. falciparum proteins by immuno-electron microscopy, using mAB SP1AG5. Stage-specific-infected erythrocytes were processed as described. Schizont-infected erythrocytes were reactive with antibody as shown. Gold particles (15 nm) in panel label longitudinal clefts (Maurer’s clefts, C); and panel B. They label knobs (K):Stage-specific IFA using mAbs. A. mAb with fluoresceinLeft panelisothiocyanate-conjugated staining. B. mAb co-localize with Bodipy ceramide in trophozoites and schizonts (C). This suggests that Rhop3 localize in lipid-rich areas of the parasite, putatively within the intra-host cell cellular network.Sam-Yellowe TY, Fujioka H, Aikawa M, Hall T, Drazba JA. A Plasmodium falciparum protein located in Maurer's clefts underneath knobs and protein localization in association with Rhop-3 and SERA in the intracellular network of infected erythrocytes. Parasitol Res. 2001 87:173-85. With kind permission of Springer Science+Business Media.
See original on MMP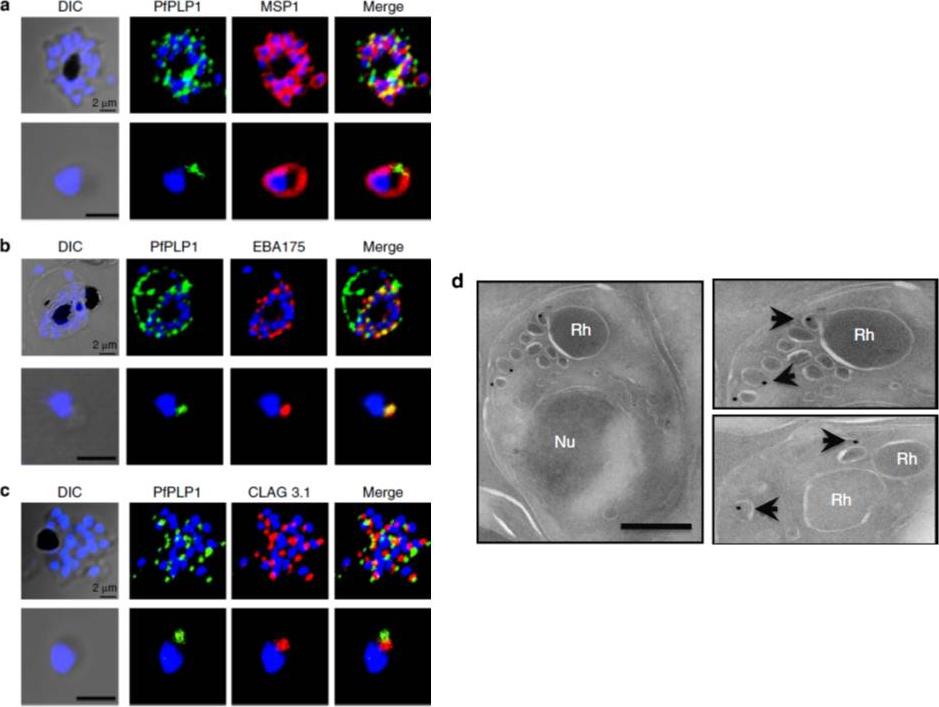
Subcellular localization of PfPLP1 in schizonts and merozoites of P. falciparum. (a) Co-immunostaining of PfPLP1 (green) with merozoite surface protein MSP1 (red). (b) Co-immunostaining of PfPLP1 (green) with micronemal protein EBA-175 (red). PfPLP1 colocalizes with EBA-175 (colocalization coefficient being more than 0.75). (c) Co-immunostaining of PfPLP1 (green) with rhoptry protein CLAG3.1 (red). PfPLP1 is not localized in rhoptry. Nuclei were counterstained with DAPI and Scale bar, 2 mm. (d) Immunoelectron microscopy of PfPLP1. Ultrathin sections of merozoites and late-stage schizonts were labelled with anti-PfPLP1 mouse sera followed by gold-labelled (15 nm) anti-mouse IgG secondary antibodies. Gold particles were detected in elongated, membrane-enclosed micronemes towards the apical end in schizonts and merozoites. Scale bar, 500 nm. PfPLP1 is localized to the micronemes of merozoites.Garg S, Agarwal S, Kumar S, Shams Yazdani S, Chitnis CE, Singh S. Calcium-dependent permeabilization of erythrocytes by a perforin-like protein during egress of malaria parasites. Nat Commun. 2013 Apr 16;4:1736.
See original on MMP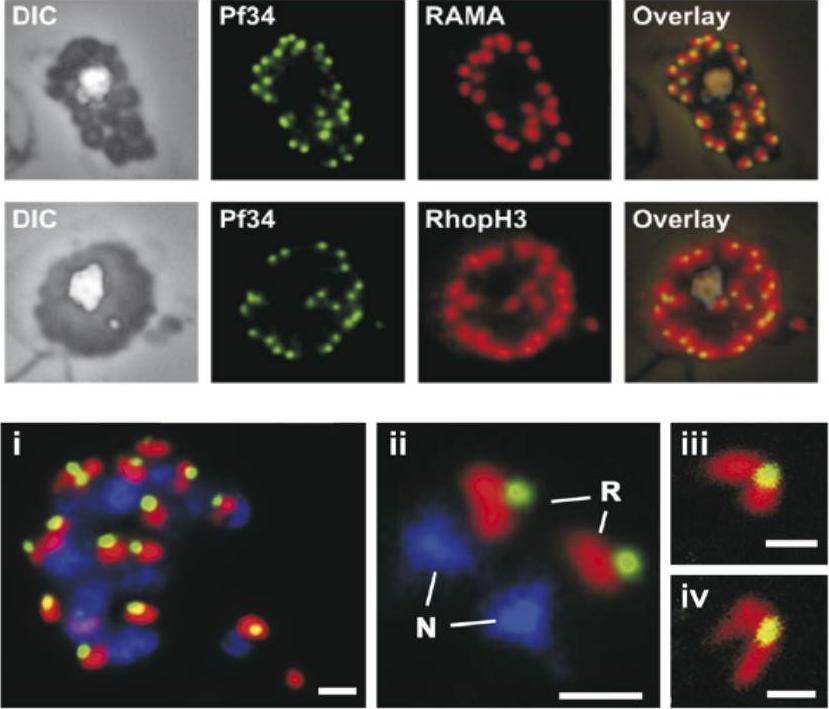
Upper 2 rows: Localization of Pf34 to the rhoptries was demonstrated in late stage parasites by immunostaining infected blood smears with anti-Pf34 and co-localising with either anti-RAMA or anti-RhopH3 (both rhoptry markers) or anti-AMA-1 (microneme marker) antibodies.Lower row: A mature schizont (i) and two extracellular merozoites (ii) showing the close apposition of Pf34-positive (green) and RAMA-positive (red) structures in the apex of the merozoite. Panels (iii) and (iv) show the apex of merozoites and the two rhoptries connected to the Pf34-positive structure at the anterior when visualised by confocal microscopy. N, nucleus; R, rhoptry. Bars represent 1 mm in (i) and (ii) and 500 nm in (iii) and (iv).Proellocks NI, Kovacevic S, Ferguson DJ, Kats LM, Morahan BJ, Black CG, Waller KL, Coppel RL. Plasmodium falciparum Pf34, a novel GPI-anchored rhoptry protein found in detergent-resistant microdomains. Int J Parasitol. 2007 37:1233-41. Copyright Elsevier
See original on MMP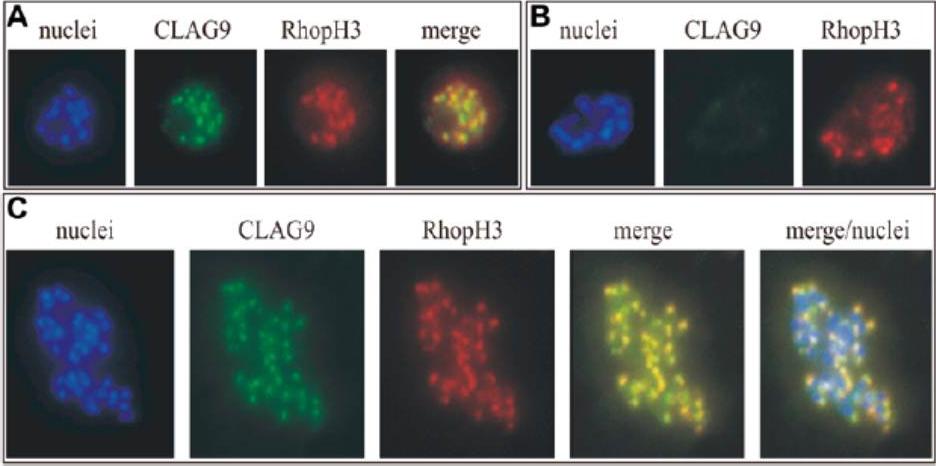
Immunofluorescence co-localization assay of Plasmodium falciparum clones 3D7 and the isogenic CLAG 9 mutant clone 11E. Antibody raised in mice to CLAG 9 peptide C were used at 1/1,000 dilution in conjunction with antibodies raised in rabbits to the rhoptry protein, RhopH3, at a dilution of 1/600. This was followed by anti-mouse antibodies conjugated to cy2 and anti-rabbit antibodies conjugated to Texas red at a dilution of 1/150. The parasite nuclei were visualized with Hoechst dye (DAPI). The slides were examined with a microscope with filters appropriate for the three fluorescent dyes,and the images merged. A Specific green staining of a schizont of clone 3D7 with the CLAG 9 antiserum, and the specific red staining of the schizont with RhopH3 antiserum. The merging of these two giving a yellow staining indicates that CLAG 9 colocalizes with RhopH3. B Rupturing schizonts of 3D7 showing multiple nuclei of individual merozoites and the co-localization of CLAG 9 and RhopH3 to the rhoptries. C CLAG 9 isogenic mutant clone 11E showing that there was no staining of the rhoptries in this parasite with the CLAG 9 antisera, although there still was specific staining with the RhopH3 antisera.Gardiner DL, Spielmann T, Dixon MW, Hawthorne PL, Ortega MR, Anderson KL, Skinner-Adams TS, Kemp DJ, Trenholme KR. CLAG 9 is located in the rhoptries of Plasmodium falciparum. Parasitol Res. 2004 93:64-7. Copyright Springer 2010
See original on MMP
PfTRAMP localizes to the rhoptries of P. falciparum schizonts and merozoites.(A) Co-localization of PfTRAMP with MSP1, EBA175 and Rh2b in schizonts and free merozoites by IFA. PfTRAMP (green) co-localizes with rhoptry marker PfRh2b (red) but does not co-localize with EBA175 (red) or merozoite surface marker MSP1 (red) in schizonts and merozoites. (B)Co-localization of PfTRAMP-GFP in transgenic P. falciparum 3D7 with MSP1, AMA1 and Clag3.1. PfTRAMP-GFP (green) co-localizes with rhoptry marker Clag 3.1 (red) but not with microneme markerPfAMA1 (red) or merozoite suface marker PfMSP1 (red) in schizonts and merozoites. Nuclei were stained with DNA intercalating dye DAPI.Siddiqui FA, Dhawan S, Singh S, Singh B, Gupta P, Pandey A, Mohmmed A, Gaur D, Chitnis CE. A Thrombospondin Structural Repeat Containing Rhoptry Protein from Plasmodium falciparum Mediates Erythrocyte Invasion. Cell Microbiol. 2013 15(8):1341-56
See original on MMP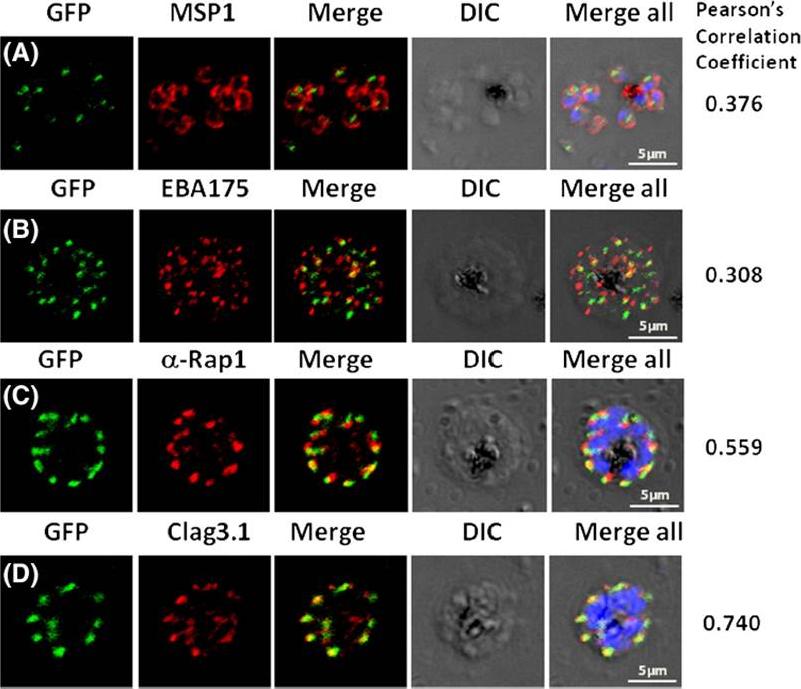
Pfμ1 co-localizeswith rhoptrymarker proteins in schizont stage parasites. Transgenic parasites expressing Pfμ1–GFPwere immunostained with antibodies specific to the Merozoite surface localized MSP1 (A), Microneme localized EBA175 (B), and Rhoptry localized RAP1 (C) and Clag3.1 (D). The parasite nuclei were stained with DAPI and slides were visualized by confocal microscopy. Representative images are shown for each antibody, together with DIC images; scale bars denote 5 μM. To quantify co-localisation, Pearson correlation coefficients of the individual stains were calculated and are shown in the right panel of each image. IFA with antibodies to rhoptry (RAP1 and Clag3.1), microneme (EBA175), and surface markers (MSP1). IFAwith anti-MSP1 antibody showed no overlap in staining betweenMSP1 and Pfμ1 (A). Similar results were seen with antibodies to EBA175 (B). Importantly, anti-RAP1 and anti-Clag3.1 showed co-localization with the Pfμ1–GFP chimeric protein (C and D), suggesting a potential role for Pfμ1 in rhoptry trafficking. Co-localization between Pfμ1 and RAP1 was first observed ~24 h post invasion in budding vesicles near the Golgi. As nuclear division commenced (32 h), Golgi multiplication occurred as well, and this resulted in apical distribution of Pfμ1 along with RAP1 in the rhoptries. Kaderi Kibria KM, Rawat K, Klinger CM, Datta G, Panchal M, Singh S, Iyer GR, Kaur I, Sharma V, Dacks JB, Mohmmed A, Malhotra P. A role for adaptor protein complex 1 in protein targeting to rhoptry organelles in Plasmodium falciparum. Biochim Biophys Acta. 2015 1853(3):699-710.
See original on MMP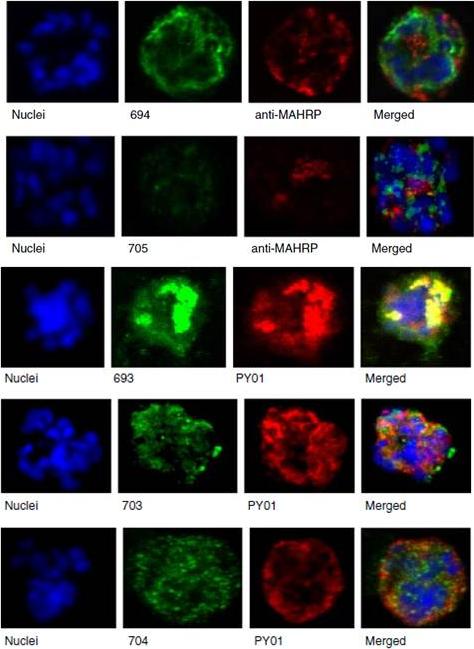
Upper panel: Mouse antisera against MAHRP1 was incubated with antisera against PfMC-2TM peptides and antisera. Lower panel: Immunofluorescence assay of mouse antisera against Rhop-3 (PY01) was incubated with rabbit antibodies against PfMC-2TM on smears of mature schizonts;Tsarukyanova I, Drazba JA, Fujioka H, Yadav SP, Sam-Yellowe TY. Proteins of the Plasmodium falciparum two transmembrane Maurer's cleft protein family, PfMC-2TM, and the 130 kDa Maurer's cleft protein define different domains of the infected erythrocyte intramembranous network. Parasitol Res. 2009 104(4):875-91.
See original on MMP
Upper panel: Mouse antisera against MAHRP1 was incubated with antisera against PfMC-2TM peptides and antisera. Lower panel: Immunofluorescence assay of mouse antisera against Rhop-3 (PY01) was incubated with rabbit antibodies against PfMC-2TM on smears of mature schizonts;Tsarukyanova I, Drazba JA, Fujioka H, Yadav SP, Sam-Yellowe TY. Proteins of the Plasmodium falciparum two transmembrane Maurer's cleft protein family, PfMC-2TM, and the 130 kDa Maurer's cleft protein define different domains of the infected erythrocyte intramembranous network. Parasitol Res. 2009 104(4):875-91.
See original on MMP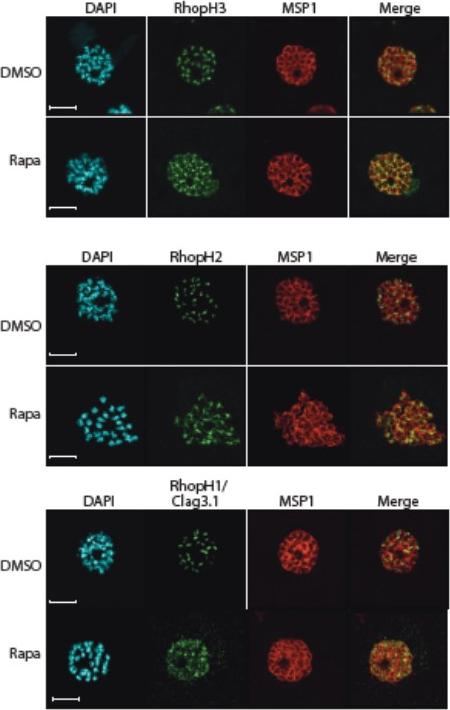
Truncation of RhopH3 leads to mistrafficking of components of the RhopH complex. IFA of mature schizonts of control (DMSO) and rapamycin-treated rhopH3-loxP parasites, probed with MSP1-specific antibodies (either mAb 89.1 or rabbit polyclonal anti-MSP1 antibodies; red) and antibodies to the three indicated RhopH components (green). Mis-localisation of the RhopH proteins was observed in all cases, and in the case of RhopH3 the protein often appeared to reside external to the plasma membrane of intracellular merozoites. Parasite nuclei were visualized by staining with DAPI. Note that, for clarity, the merge panels do not include the DAPI signal. Scale bar, 5 μm.Sherling ES, Knuepfer E, Brzostowski JA, Miller LH, Blackman MJ, van Ooij C. The Plasmodium falciparum rhoptry protein RhopH3 plays essential roles in host cell invasion and nutrient uptake. Elife. 2017 6. pii: e23239.
See original on MMP
Truncation of RhopH3 leads to mistrafficking of components of the RhopH complex and loss of complex formation. A) IFA showing colocalization of RhopH3, RhopH2 and RhopH1/Clag3.1 with the rhoptry marker RAP2 in schizonts of control (DMSO) rhopH3-loxP parasites but loss of colocalization following rapamycin (Rapa) treatment. Parasite nuclei were visualized by staining with 4,6-diamidino-2-phenylindole (DAPI). Scale bar, 5 μm. Immunofluorescence analysis (IFA) showed that, as expected, RhopH3 colocalized with the rhoptry marker RAP2 in mature schizonts of control rhopH3-loxP parasites. However, in rapamycin-treated (RhopH3Δ4-219 6) parasites, this colocalization was lost, although RAP2 was still detected in a punctate, apically-disposed pattern typical of rhoptries B) Colocalization of the members of the RhopH complex. RhopH3, RhopH2 and Rhop 1/Clag3.1 colocalize in rhopH3-loxP parasites treated with DMSO, but this colocalization is lost in parasites treated with rapamycin. Scale bar, 2 μm. Neither RhopH2 nor RhopH1/Clag3.1 colocalized with 233 RhopH3Δ4-6 in the mutant parasites. The RhopH2 and RhopH1/Clag3.1 signals were also distinct in the mutant parasites, although in this case some limited colocalization of these proteins was apparent (bottom images).Sherling ES, Knuepfer E, Brzostowski JA, Miller LH, Blackman MJ, van Ooij C. The Plasmodium falciparum rhoptry protein RhopH3 plays essential roles in host cell invasion and nutrient uptake. Elife. 2017 6. pii: e23239.
See original on MMP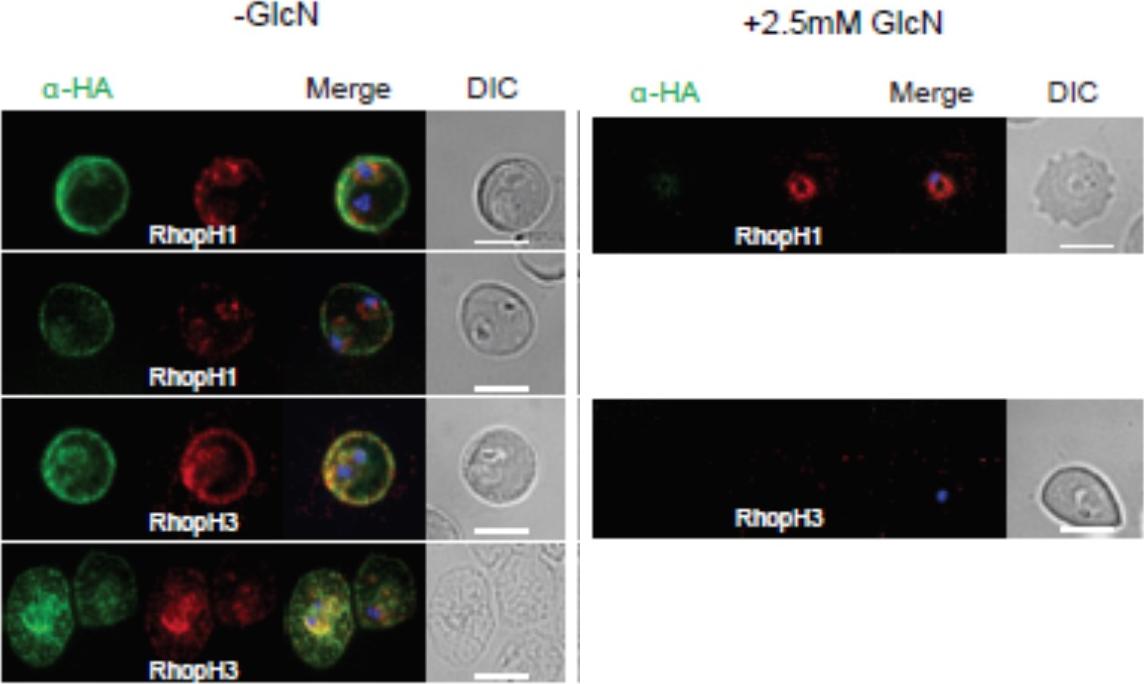
Localization of RhopH1/clag3 and RhopH3 in infected erythrocytes when RhopH2 expression is knocked down. Representative immunofluorescence analysis of erythrocytes infected with RhopH2-HAglmS parasites grown in 0 mM or 2.5 mM GlcN. Cells fixed with acetone/methanol and labelled with anti-HA antibody to detect RhopH2 IFAs and other antibodies, as indicated. the localization of RhopH3 and to a lesser extent RhopH1/clag3 was affected when RhopH2 expression was knocked down.Counihan N, Chisholm SA, Bullen HE, Srivastava A, Sanders PR, Jonsdottir TK, Weiss GE, Ghosh S, Crabb BS, Creek DJ, Gilson PR, de Koning-Ward TF. Plasmodium falciparum parasites deploy RhopH2 into the host erythrocyte to obtain nutrients, grow and replicate. Elife. 2017 Mar 2;6. pii: e23217
See original on MMP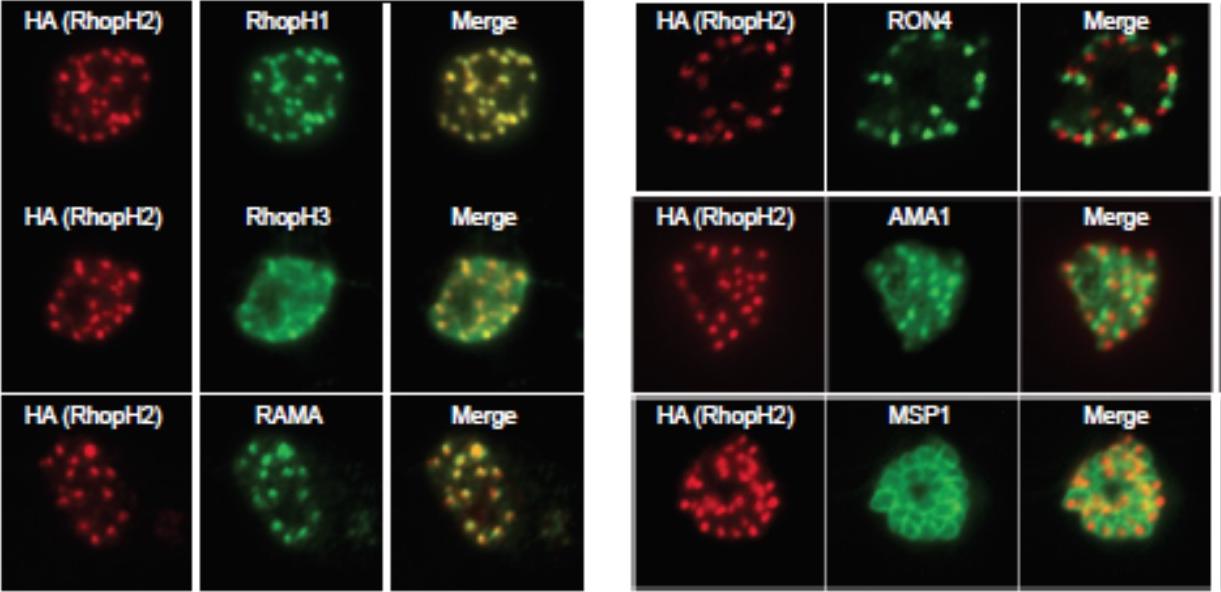
Generation of transgenic parasites in which RhopH2 is epitope-tagged. Immunofluorescence analysis (IFA) on schizonts fixed with acetone/methanol and labelled with anti-HA antibody to detect RhopH2 and other antibodies, as indicated. Immunofluorescence analysis (IFA) confirmed RhopH2-HA localized to the rhoptry and co-localized with other rhoptry bulb proteins, RhopH1, RhopH3 and RAMA but not with the rhoptry neck protein, RON4, the micronemal marker, AMA-1 or the plasma membrane protein MSP1.Counihan N, Chisholm SA, Bullen HE, Srivastava A, Sanders PR, Jonsdottir TK, Weiss GE, Ghosh S, Crabb BS, Creek DJ, Gilson PR, de Koning-Ward TF. Plasmodium falciparum parasites deploy RhopH2 into the host erythrocyte to obtain nutrients, grow and replicate. Elife. 2017 Mar 2;6. pii: e23217
See original on MMPMore information
| PlasmoDB | PF3D7_0302500 |
| GeneDB | PF3D7_0302500 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | MAL3P1.5, PFC0120w |
| Orthologs | |
| Google Scholar | Search for all mentions of this gene |