PF3D7_0302200 cytoadherence linked asexual protein 3.2 (CLAG3.2)
Disruptability [+]
| Species | Disruptability | Reference | Submitter |
|---|---|---|---|
| P. falciparum 3D7 |
Possible |
21320181 | Theo Sanderson, Wellcome Trust Sanger Institute |
| P. falciparum 3D7 |
Possible |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen |
Mutant phenotypes [+]
None reported yet. Please press the '+' button above to add one.Imaging data (from Malaria Metabolic Pathways)
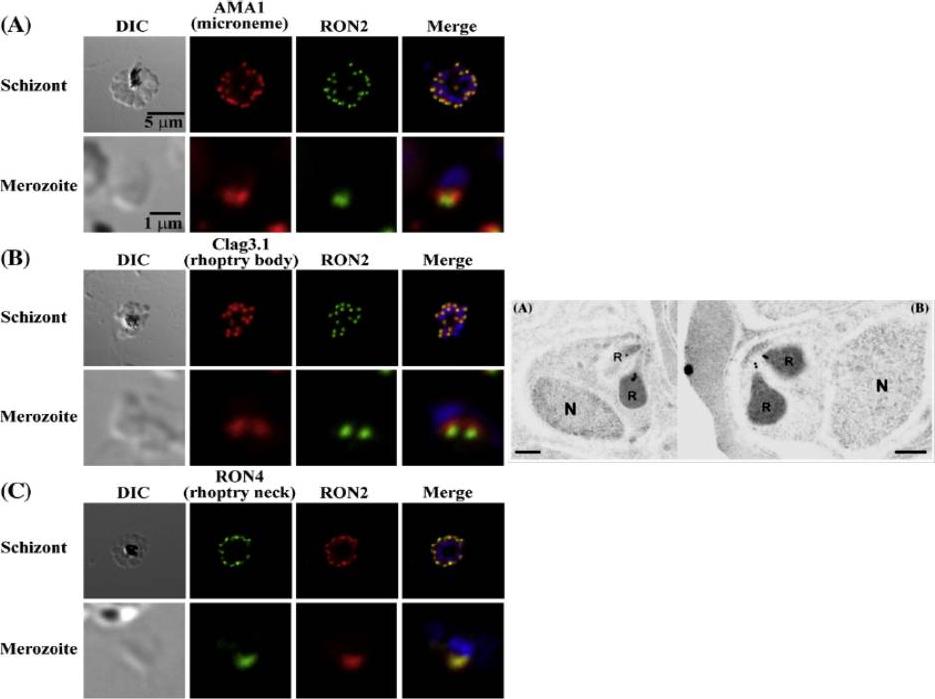
Left: PfRON2 is expressed at the apical end of Plasmodium merozoites. Schizont infected erythrocytes and merozoites were dual-labeled with antisera against PfRON2 and PfAMA1 PF11_0344 (A), PfClag3.1 PFC0110w (B), or PfRON4 PF11_0168 (C). Merged images are shown in the right panels. All segmented schizonts and merozoites are positive for PfRON2. Nuclei are counterstained with DAPI. Colocalization of PfRON2 with PfRON4 (rhoptry neck marker) was observed but neither colocalized with PfClag3.1 (rhoptry body marker) nor PfAMA1 (microneme marker). Right: Longitudinally sectioned merozoites in schizont-infected erythrocytes were labeled with anti-PfRON2 serum followed by secondary Ab conjugated with gold particles. Gold particles were restricted to the narrow neck portion of the rhoptries (R). Two different images are shown (A and B). N indicates nucleus. Bars=200 nm. Cao J, Kaneko O, Thongkukiatkul A, Tachibana M, Otsuki H, Gao Q, Tsuboi T, Torii M. Rhoptry neck protein RON2 forms a complex with microneme protein AMA1 in Plasmodium falciparum merozoites. Parasitol Int. 2009 58:29-35.
See original on MMP
Ppolyclonal antibodies to a carboxy-terminal recombinant fragment conserved between the two clag3 products were prepared. Confocal microscopy with this antibody confirmed reports localizing these proteins to the host cytosol and possibly the erythrocyte membrane as well as within rhoptries of invasive merozoites.Nguitragool W, Bokhari AA, Pillai AD, Rayavara K, Sharma P, Turpin B, Aravind L, Desai SA. Malaria parasite clag3 genes determine channel-mediated nutrient uptake by infected red blood cells. Cell. 2011 145:665-77.
See original on MMP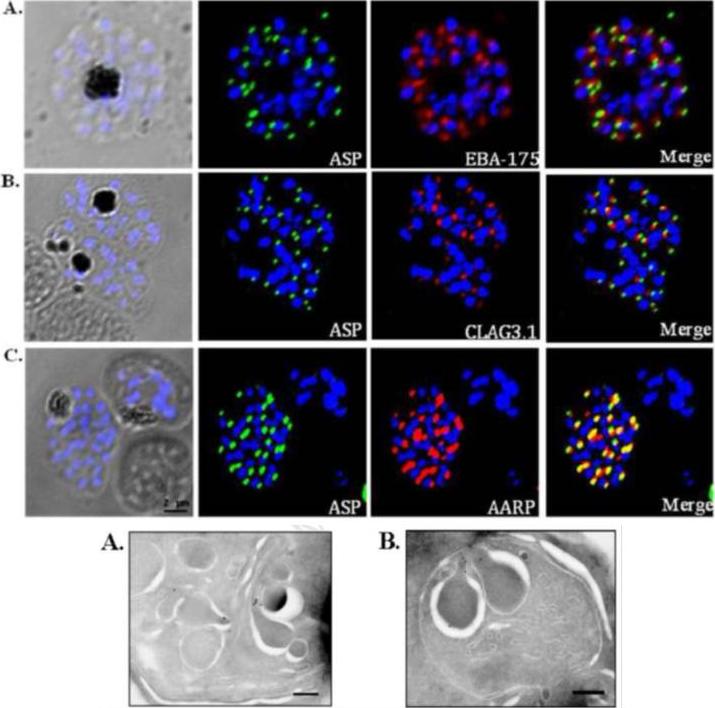
Co-localization of PfASP in late stage P. falciparum schizonts with microneme protein, EBA-175 (A); rhoptry bulb protein, Clag3.1 (B); and rhoptry neck protein, PfAARP (C). P. falciparum schizonts were stained with anti-PfASP-RIV mouse sera (green) and either anti-EBA-175 (red), anti-CLAG3.1 (red) or anti PfAARP (red) rabbit sera. Parasite nuclei were stained with DAPI (blue). PfASP did not co-localize with microneme protein EBA-175 or rhoptry bulb protein CLAG3.1 but co-localized with rhoptry neck protein PfAARP in segmented P. falciparum merozoites within late-stage schizonts. Bottom: Ultra thin sections of late-stage P. falciparum schizonts containing segmented merozoites were labelled with anti-PfASP-RIV mouse sera and gold-conjugated anti-mouse IgG. Labelling was observed in the neck of rhoptries in segmented merozoites of late-stage P. falciparum schizonts. Scale bar = 250 nm.Srivastava A, Singh S, Dhawan S, Alam MM, Mohmmed A, Chitnis CE. Localization of apical Sushi Protein in Plasmodium falciparum merozoites. Mol Biochem Parasitol. 2010 74(1):66-9 Copyright Elsevier2011
See original on MMP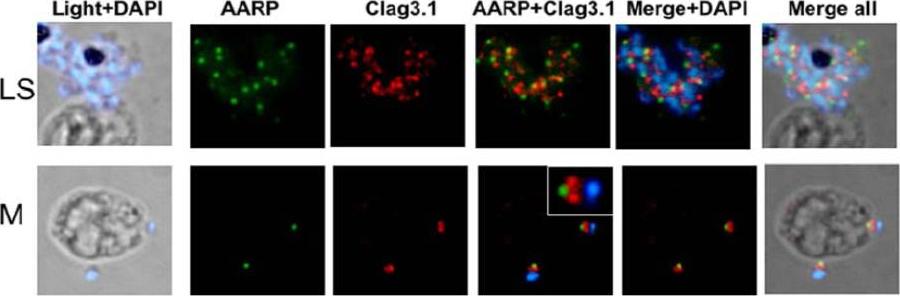
Spatial localization of PfAARP by co-immunostaining studies with rhoptry resident protein Clag3.1. P. falciparum parasites were coimmuno-stained with anti- PfAARP (green) and anti-Clag3.1 (red) antibodies. The parasite nuclei were stained with DAPI (blue). Clag3.1 staining was present in the rhoptry bulb in the free and invading merozoites (lower panel). PfAARP was localized just above these two rhoptry bulbs towards the apex of the merozoites. Enlarged image of merozoite invading in the host erythrocyte is shown in the insetWickramarachchi T, Devi YS, Mohmmed A, Chauhan VS. Identification and characterization of a novel Plasmodium falciparum merozoite apical protein involved in erythrocyte binding and invasion. PLoS ONE. 2008 Mar 5;3(3):e1732.
See original on MMP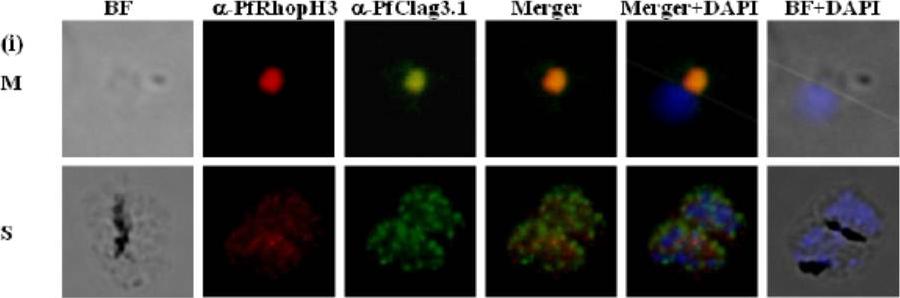
Immunofluorescence assay to show localization of PfRhopH3-C in rhoptries. (i) Co-localization of PfRhopH3 and PfClag3.1 using their respective antibodies at schizont (S) and free merozoites (M) stages. Staining with preimmune sera and anti-GST antibodies respectively were used as controls. The parasite nuclei were stained with DAPI (blue). PfRhopH3 and PfClag3.1 were localized at the apical tip.Ranjan R, Chugh M, Kumar S, Singh S, Kanodia S, Hossain MJ, Korde R, Grover A, Dhawan S, Chauhan VS, Reddy VS, Mohmmed A, Malhotra P. Proteome analysis Rrveals a large merozoite surface protein-1 associated complex on the Plasmodium falciparum merozoite surface. J Proteome Res. 2010 10(2):680-91 Copyrihgt American Chemical Society 2011.
See original on MMP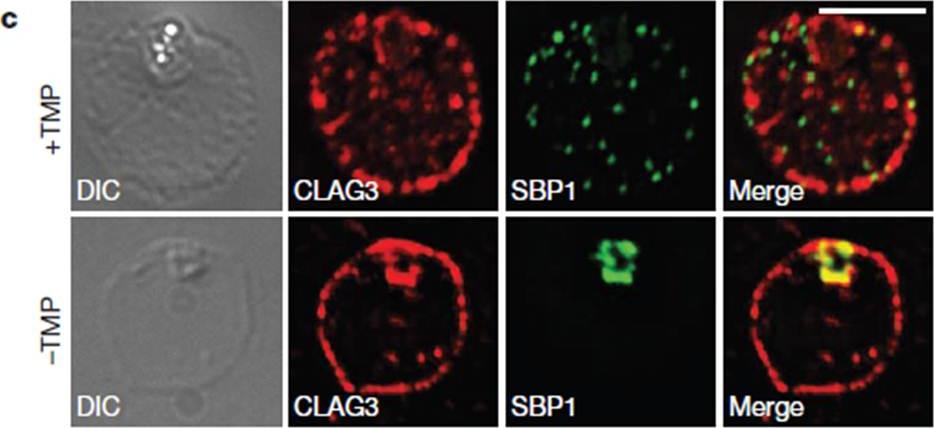
HSP101 is required for activation of PSAC but not trafficking of CLAG3 to the RBC periphery. CLAG3 proteins localize to the infected RBCperiphery and are the only parasite proteins known to be involved in PSAC activation IFA of ring-stage 13F10 parasites showing CLAG3 localization to the RBC membrane with or without TMP (DDD-stabilizing small molecule trimethoprim). TMP was removed in the late schizont stage and parasites were allowed to develop 18 h before fixation with 90% acetone/10% methanol. Scale bar, 5 mm. Beck JR, Muralidharan V, Oksman A, Goldberg DE. PTEX component HSP101 mediates export of diverse malaria effectors into host erythrocytes. Nature. 2014 Jul 16. [Epub ahead of print]
See original on MMP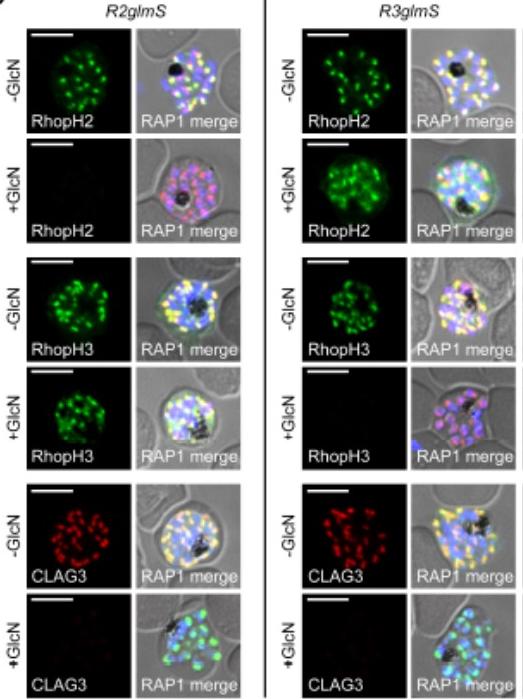
IFA of indicated proteins in R2glmS (RopH2 knockdown) and R3glmS (RopH3 knockdown) at schizont stage; colocalization with RAP1 and apical puncta in daughter merozoites indicates rhoptry trafficking. Scale bars, 5 μm. GlcN treatment of either R2glmS or R3glmS abolished not only the targeted gene product but also CLAG3, a third better-characterized member of the RhopH complex. Indirect immunofluorescence microscopy confirmed this surprising finding and provided additional insights. While RhopH2 was still detected in the R3glmS knockdown, it did not fully colocalize with RAP1 but instead had a more diffuse distribution; this suggests RhopH2 may be retained in the parasite endoplasmic reticulum if it fails to interact with RhopH3. In contrast, RhopH3 appeared to traffic normally inGlcN-treated R2glmS parasites, as indicated by apical colocalization with RAP1 inmerozoites. Thus, CLAG3, RhopH2 and RhopH3 assemble into a complex either during or immediately after translation.Ito D, Schureck MA, Desai SA. An essential dual-function complex mediates erythrocyte invasion and channel-mediated nutrient uptake in malaria parasites. Elife. 2017 Feb 21;6.
See original on MMP
RhopH proteins are deposited in the PV and exported via PTEX. IFA of 13F10 showing export of RhopH proteins after growth with or without TMP for 48 h. Colocalization with exported HRP2 or SBP1, as indicated. Scale bars, 5 μm. Export of RhopH2 and RhopH3 were both fully blocked in trophozoite-stage parasites grown for 48 h without TMP. Colocalization with HRP2, a parasite protein carrying the canonical PEXEL export signal showed both proteins trapped in the vacuole upon PTEX knockdown. CLAG3 export requires PTEX activity as TMP removal prevented its movement out of the PV (bottom row); colocalization with SBP1, another PTEX substrate.Ito D, Schureck MA, Desai SA. An essential dual-function complex mediates erythrocyte invasion and channel-mediated nutrient uptake in malaria parasites. Elife. 2017 Feb 21;6.
See original on MMP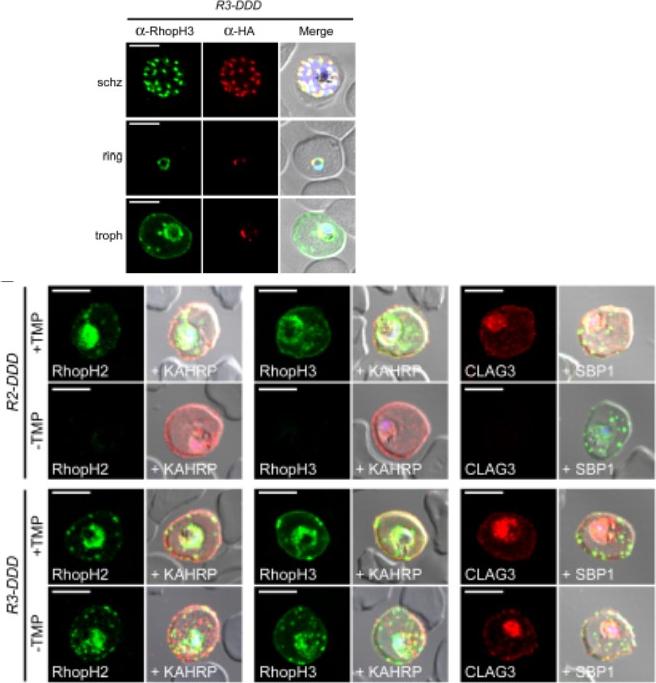
(D) IFA of R3-DDD (conditional RopH3 mutant) probed with indicated antibodies at each parasite stage. Note that both antibodies detect protein in schizont rhoptries, but that only the anti-RhopH antibody detects export to the host membrane in trophozoites. Scale bars, 5 μm. (E) Trophozoite-stage IFAs of indicated parasites cultivated with and without TMP for 48 h, showing that knockdown abolishes each member of the complex in R2-DDD (conditional RopH2 mutant) but not R3-DDD. Co-localization is shown with exported parasite proteins KAHRP or SBP1 (red or green in merge panels, respectively). Scale bars, 5 μm. The RhopH complex appears to be secreted into the PV (upper panel), suggesting that its member proteins must cross the bounding PVM to enter host cytosol and eventually reach the erythrocyte surface. While TMP removal rendered all three RhopH proteins undetectable in the R2-DDD parasite, these proteins and their transfer to trophozoites were preserved inR3-DDD.Ito D, Schureck MA, Desai SA. An essential dual-function complex mediates erythrocyte invasion and channel-mediated nutrient uptake in malaria parasites. Elife. 2017 Feb 21;6. PMID: 28221136.
See original on MMP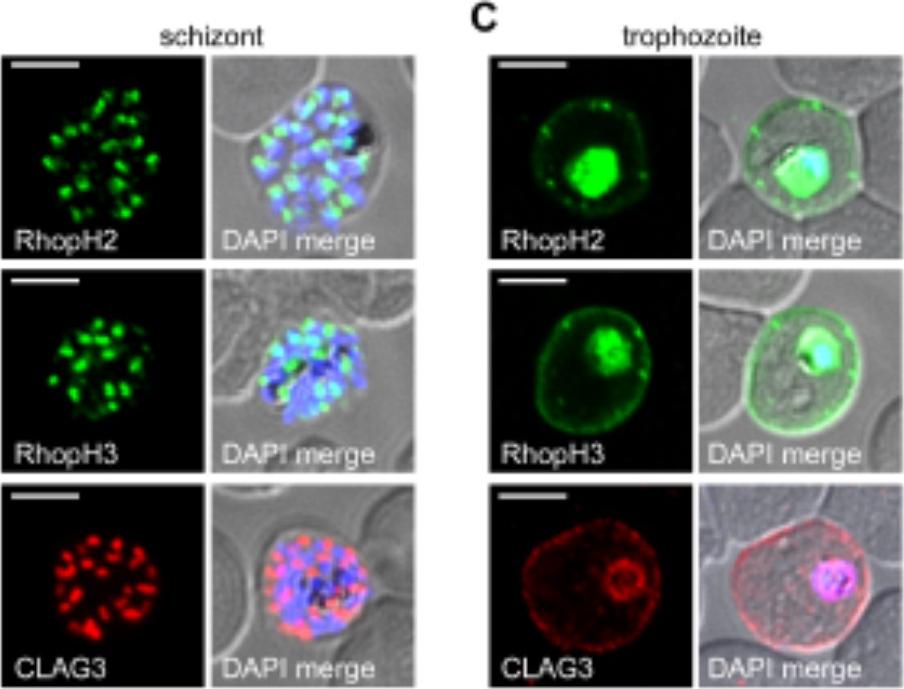
RhopH2 and RhopH3 are membrane-associated proteins. IFA of indicated proteins in R2glmS (RopH2 knockdown) and R3glmS (RopH3 knockdown) at the trophozoite stage. IFA of mature schizont-infected cells with indicated antibodies. Punctate labeling indicates that these proteins localize to rhoptry organelles in daughter merozoites. Scale bar, Scale bars, 5 μm. IFA of trophozoite-infected cells, showing that each protein localizes to the host membrane and to the vacuole surrounding the intracellular parasite. Scale bar, 5 μmImmunofluorescence microscopy confirmed packaging into rhoptries within schizont-infected cells (B). Imaging of maturing trophozoite-stage parasites also showed that these proteins traffic to the host membrane (C). Because each gene is transcribed and translated only within schizonts, the protein found in these maturing trophozoites reflects transfer from the merozoite during invasion.Ito D, Schureck MA, Desai SA. An essential dual-function complex mediates erythrocyte invasion and channel-mediated nutrient uptake in malaria parasites. Elife. 2017 Feb 21;6.
See original on MMPMore information
| PlasmoDB | PF3D7_0302200 |
| GeneDB | PF3D7_0302200 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | MAL3P1.2, PFC0110w |
| Orthologs | |
| Google Scholar | Search for all mentions of this gene |