PF3D7_0217500 calcium-dependent protein kinase 1 (CDPK1)
Disruptability [+]
| Species | Disruptability | Reference | Submitter | |
|---|---|---|---|---|
| P. falciparum 3D7 |
Refractory |
18454143 | Theo Sanderson, Wellcome Trust Sanger Institute | |
| P. falciparum 3D7 |
Refractory |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen | |
| P. berghei ANKA |
Possible |
RMgm-1218 | Imported from RMgmDB | |
| P. berghei ANKA |
Possible |
RMgm-966 | Imported from RMgmDB | |
| P. berghei ANKA |
Refractory |
RMgm-518 | Imported from RMgmDB | |
| P. berghei ANKA |
Possible |
PlasmoGEM (Barseq) | PlasmoGEM | |
Mutant phenotypes [+]
| Species | Stage | Phenotype | Reference | Submitter |
|---|---|---|---|---|
| P. falciparum 3D7 | Asexual |
Invasion defect |
28680058 (Knock down)
Reduced attachment and subsequent invasion |
Theo Sanderson, Wellcome Trust Sanger Institute |
| P. falciparum 3D7 | Asexual |
Cell cycle arrest |
23548171 (Knock down) | Theo Sanderson, Francis Crick Institute |
| P. falciparum 3D7 | Gametocyte |
Attenuated |
29311293 (Conditional)
Knocked out on a mutant background after adaptation |
Theo Sanderson, Francis Crick Institute |
| P. berghei ANKA | Asexual |
No difference |
RMgm-1218 | Imported from RMgmDB |
| P. berghei ANKA | Asexual |
No difference |
RMgm-966 | Imported from RMgmDB |
| P. berghei ANKA | Asexual |
No difference |
PlasmoGEM (Barseq) | PlasmoGEM |
| P. berghei ANKA | Oocyst |
Difference from wild-type |
RMgm-966
No oocyst formation |
Imported from RMgmDB |
Imaging data (from Malaria Metabolic Pathways)
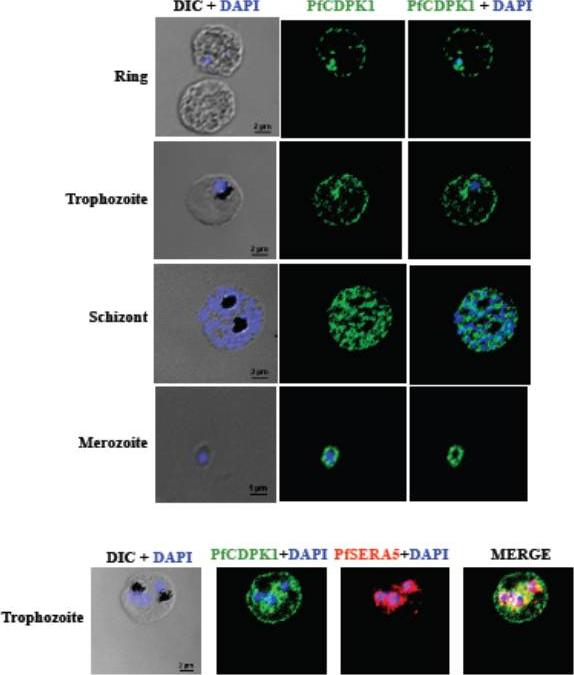
PfCDPK1 is expressed throughout erythrocytic schizogony as detected by by immunofluorescence assay (IFA). PfCDPK1 was detected in blood stages of P. falciparum by IFA using anti-PfCDPK1 mouse sera and Alexa Fluor 488-conjugated anti-mouse IgG goat sera (green). Anti-SERA5 rabbit sera and Alexa Fluor 594-conjugated anti-rabbit IgG goat sera (red) were used to localize the parasitophorous vacuole (PV) protein SERA5. Nuclear DNA was counterstained with DAPI (blue). Bright field (DIC) merged with DAPI (DIC + DAPI), fluorescence images detecting PfCDPK1 (PfCDPK1) and merged fluorescence images detecting PfCDPK1 and DAPI, PfSERA5 and DAPI or PfCDPK1, PfSERA5 and DAPI (Merge) are shown. Bar represents 2 mm (ring, trophozoite, schizont) or 1 μm (merozoite). PfCDPK1 localized to the rim of the infected erythrocyte and appeared to be associated with the erythrocyte membrane in rings and trophozoites (lower panel). In addition, PfCDPK1 was found in the parasitophorous vacuole (PV) and co-localized with SERA5, a known PV protein in trophozoites. Inmerozoites, PfCDPK1 was primarily found at the boundary of the merozoite at the merozoite plasma membraneBansal A, Singh S, More KR, Hans D, Nangalia K, Yogavel M, Sharma A, Chitnis CE. Characterization of Plasmodium falciparum calcium dependent protein kinase 1 (PfCDPK1) and its role in microneme secretion during erythrocyte invasion. J Biol Chem. 2013 288(3):1590-602.
See original on MMP
Expression of functional CDPK1 fusion proteins in 3D7 P. falciparum parasites Immunolocalisation of CDPK1 in late schizont stage parasites showing co-localisation with MSP1-19 and GAP45, proteins which reside on the plasma membrane and inner membrane complex, respectively. CDPK1 was detected with CDPK1-specific mouse monoclonal 3E6 and the other proteins with polyclonal rabbit antibodies .Azevedo MF, Sanders PR, Krejany E, Nie CQ, Fu P, Bach LA, Wunderlich G, Crabb BS, Gilson PR. Inhibition of Plasmodium falciparum CDPK1 by conditional expression of its J-domain demonstrates a key role in schizont development. Biochem J. 2013 452(3):433-41
See original on MMP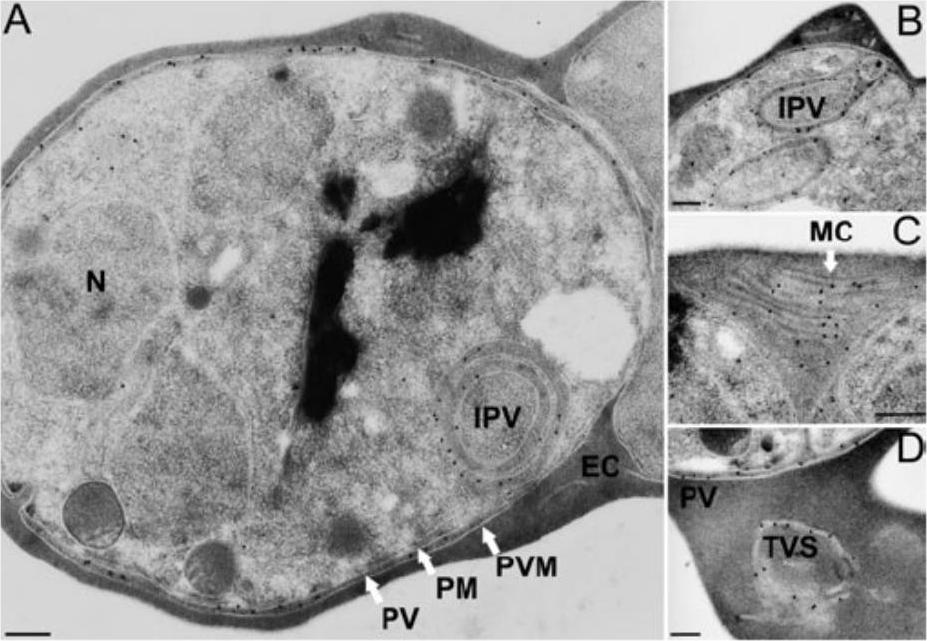
Subcellular localization of PfCDPK1. Sections of P. falciparum (clone K1) erythrocytic stages were incubated with the affinity-purified PfCDPK1-specific antibody 2129 and binding was visualized using gold-conjugated protein A. A and B. Gold particles indicative of antibody binding associated with the parasitophorous vacuole and intracellular vacuoles (intraparasitic vacuole) of schizont stages. Intracellular vacuoles originating from the plasma membrane of the parasite (B). C and D. Sections of erythrocytes infected with two schizonts and one schizont, respectively, showing labelling of the tubovesicular system in the host erythrocyte. The membranous structure visible in the erythrocyte cytosol in (C) is also known as Maurer’s clefts. EC, erythrocyte cytosol; MC, Maurer’s clefts; IPV, intraparasitic vacuole; N, nucleus; PM, parasite plasma membrane; PV, parasitophorous vacuole; PVM, parasitophorous vacuole membrane; TVS, tubovesicular system. Size bars (200 nm) have been inserted in each panel. Möskes C, Burghaus PA, Wernli B, Sauder U, Dürrenberger M, Kappes B. Export of Plasmodium falciparum calcium-dependent protein kinase 1 to the parasitophorous vacuole is dependent on three N-terminal membrane anchor motifs. Mol Microbiol. 2004 54:676-691. Copyright John Wiley & Sons Ltd. 2010.
See original on MMP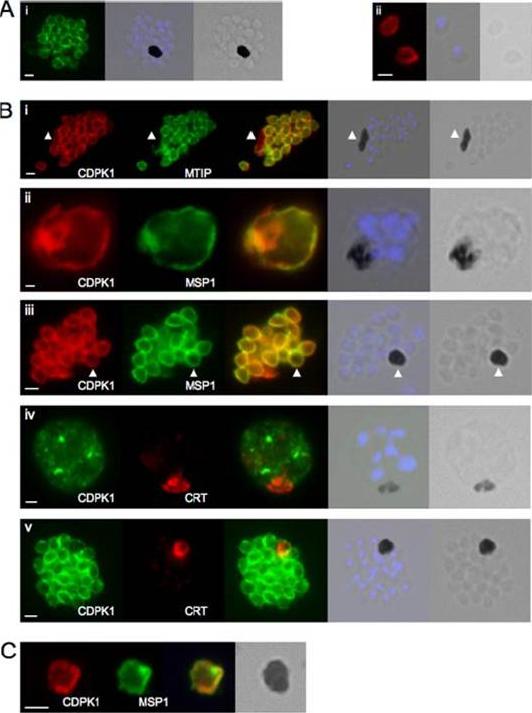
CDPK1 localizes to the plasma membrane of merozoites. A, an anti-CDPK1 antibody detects CDPK1 at the parasite periphery in schizonts (panel i) and free merozoites (panel ii). The second panel shows a merged image of nuclear staining with 4’,6’-diamino-2-phenylindole (blue) and the bright field image, and the third panel shows the bright field image. B, panel i, CDPK1 colocalizes with MTIP around the periphery of merozoites but is also found around the residual body, marked with an arrowhead, from which MTIP is absent. In young (panel ii) and segmented (panel iii) schizonts, CDPK1 colocalizes with the plasma membrane marker MSP1. Both proteins are also detected around the residual body, marked with an arrowhead. CDPK1 does not colocalize with the food vacuole marker PfCRT in either immature (panel iv) or mature (panel v) schizonts. The third panels in all of the images show the merged images of the antibody staining, beside which is a merged image of the bright field image and the nuclei of the parasites stained with 4’,6’-diamino-2-phenylindole (blue), followed by the bright field image alone. C, both CDPK1 (red) and MSP1 (green) can be detected on residual bodies that are released upon schizont rupture. The merged images of the antibody staining are shown in the third panels, followed by the bright field image. In all cases the white scale bar in the first panel of each set of images represents 1 mm.Green JL, Rees-Channer RR, Howell SA, Martin SR, Knuepfer E, Taylor HM, Grainger M, Holder AA. The motor complex of Plasmodium falciparum: phosphorylation by a calcium-dependent protein kinase. J Biol Chem. 2008 7;283(45):30980-9. PMID:
See original on MMP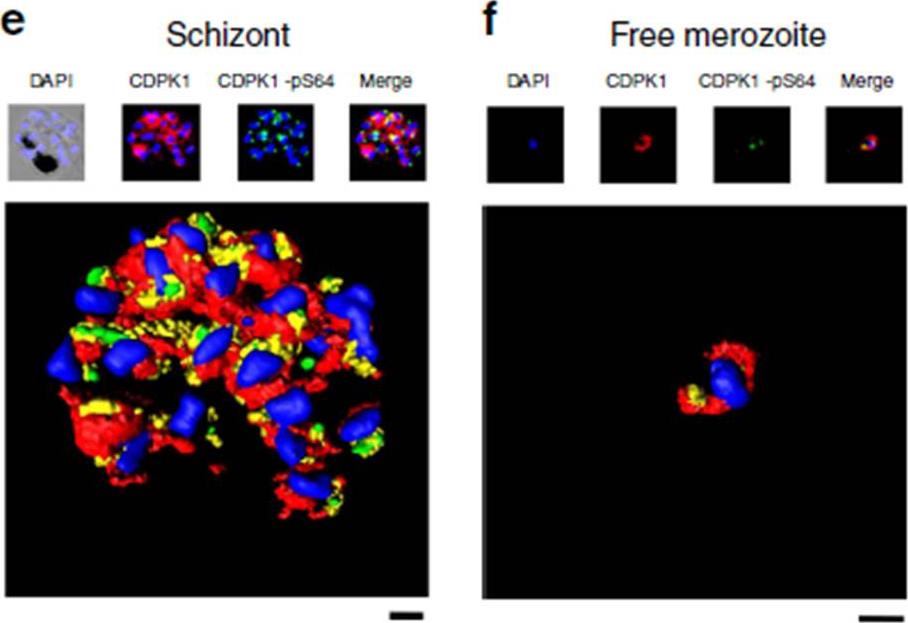
Phosphorylation of PfCDPK1 at S64 (phosphorylation at Serine 64) occurs during the schizont (Schiz) stage of parasite development. (e) A rendered image where deconvoluted z stacks were reconstructed in 3D, with interpolation, of wild-type parasites stained with DAPI (blue), the structural CDPK1 antibody to reveal the total pool of PfCDPK1 (red) and the CDPK1-pS64 antibody (green). (f) Same as e but in this case the images for z stacks are of a free merozoite. These results are representative of at least three experiments. Scale bars, e-1 mm, f-0.5 mm. Immunofluorescence using an anti-HA antibody that detected PfCDPK1-HA in transgenic parasites revealed the peripheral (membrane) localization of PfCDPK1 in the nascent merozoites contained within the late-stage schizonts or free merozoites. In contrast, S64-phosphorylated PfCDPK1 appeared as discrete ‘punctate spots’ at the periphery of merozoites within late Schizonts .Alam MM, Solyakov L, Bottrill AR, Flueck C, Siddiqui FA, Singh S, Mistry S, Viskaduraki M, Lee K, Hopp CS, Chitnis CE, Doerig C, Moon RW, Green JL, Holder AA, Baker DA, Tobin AB. Phosphoproteomics reveals malaria parasite Protein Kinase G as a signalling hub regulating egress and invasion. Nat Commun. 2015 6:7285
See original on MMP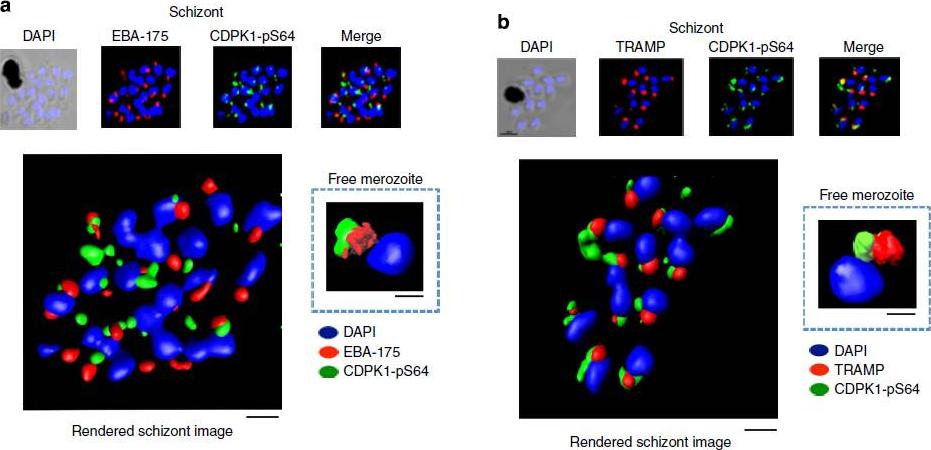
PfCDPK1 phosphorylated at serine-64 is associated with apical parasite structures. (a) A schizont stage parasite stained with DAPI to reveal the nuclei (blue), an antibody to PfEBA-175 to reveal the micronemes (red) and the phospho-specific CDPK1-pS64 antibody (green). A merge of images from all three stains is shown on the far right; this is a representative image from at least three experiments. Also shown is a rendered image where deconvoluted z stacks were reconstructed in 3D, with interpolation. The inset shows a rendered image of a free merozoite from the same preparation . (b) The same as a, but instead of probing with an anti-EBA-175 antibody the preparation was probed with an antibody to the rhoptrymarker PfTRAMP . Scale bars, a-1 mm (insert-0.5 mm), b-1 mm (insert-0.5 mm).Alam MM, Solyakov L, Bottrill AR, Flueck C, Siddiqui FA, Singh S, Mistry S, Viskaduraki M, Lee K, Hopp CS, Chitnis CE, Doerig C, Moon RW, Green JL, Holder AA, Baker DA, Tobin AB. Phosphoproteomics reveals malaria parasite Protein Kinase G as a signalling hub regulating egress and invasion. Nat Commun. 2015 6:7285
See original on MMP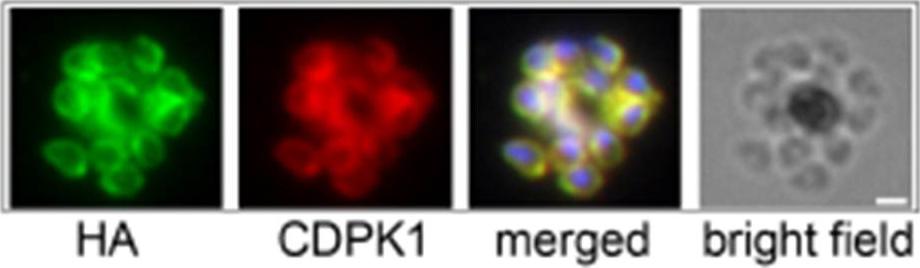
Inhibition of PfCDPK1 has no effect on asexual parasite growth. Immunofluorescence using anti-HA and anti-CDPK1 antibodies confirms that CDPK1 T145G-HA (a gatekeeper mutant) localizes correctly to the plasma membrane of parasites. Scale bar is 1 μm.Ansell KH, Jones HM, Whalley D, Hearn A, Taylor DL, Patin EC, Chapman TM, Osborne SA, Wallace C, Birchall K, Large J, Bouloc N, Smiljanic-Hurley E, Clough B, Moon RW, Green JL, Holder AA. Biochemical and antiparasitic properties of inhibitors of the Plasmodium falciparum calcium-dependent protein kinase PfCDPK1. Antimicrob Agents Chemother. 2014 58(10):6032-43.
See original on MMP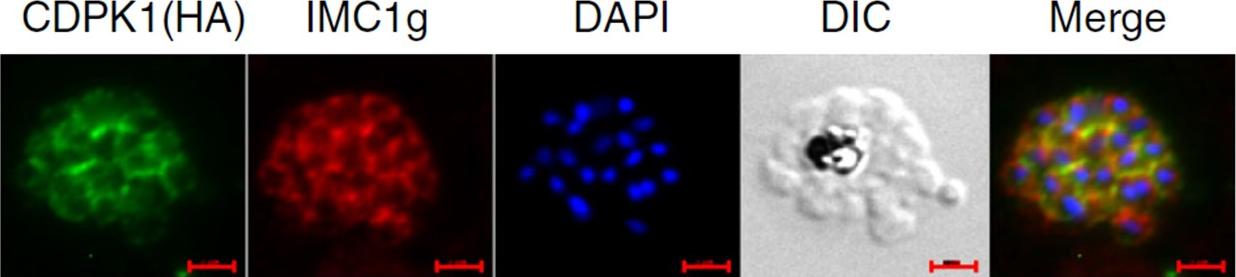
IFA was performed on PfCDPK1-3HA-DD parasites using anti-HA antibody to detect CDPK1 and anti-IMC1g antisera. Scale bar is 2 μm. Data strongly suggest that PfCDPK1 is a major kinase that phosphorylates IMC1g in the parasite. Moreover, PfCDPK1 exhibited localization in close proximity with IMC1g in the parasite, which was reflected by overlapping fluorescence in IFA.Kumar S, Kumar M, Ekka R, Dvorin JD, Paul AS, Madugundu AK, Gilberger T, Gowda H, Duraisingh MT, Keshava Pr asad TS, Sharma P. PfCDPK1 mediated signaling in erythrocytic stages of Plasmodium falciparum. Nat Commun. 2017;8(1):63.
See original on MMPMore information
| PlasmoDB | PF3D7_0217500 |
| GeneDB | PF3D7_0217500 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | PF02_0166, PFB0815w |
| Orthologs | PBANKA_0314200 , PCHAS_0316300 , PKNH_0403400 , PVP01_0407500 , PVX_002665 , PY17X_0314700 |
| Google Scholar | Search for all mentions of this gene |