PF3D7_0102200 ring-infected erythrocyte surface antigen (RESA)
Disruptability [+]
| Species | Disruptability | Reference | Submitter |
|---|---|---|---|
| P. falciparum 3D7 |
Possible |
15853885 truncation, sensitive to heat shock |
Theo Sanderson, Wellcome Trust Sanger Institute |
| P. falciparum 3D7 |
Possible |
18614010 | Theo Sanderson, Wellcome Trust Sanger Institute |
| P. falciparum 3D7 |
Possible |
USF piggyBac screen (Insert. mut.) | USF PiggyBac Screen |
Mutant phenotypes [+]
None reported yet. Please press the '+' button above to add one.Imaging data (from Malaria Metabolic Pathways)
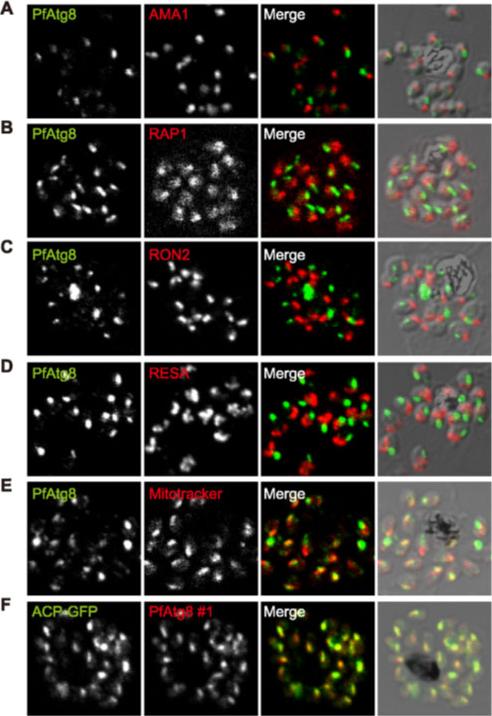
PfAtg8 localizes to the apicoplast. P. falciparum FCR3 (A–E) and P. falciparum 3D7 transfected with ACP-GFP (F–H) were stained with the indicated organelle markers and visualized by confocal microscopy (because ACP-GFP was not uniformly expressed, some merozoites displayed only faint GFP signals). Anti-PfAtg8 antibody #1 was used in (A–F), and anti-PfAtg8 antibody #2 was used in (G). Apical membrane antigen 1 (AMA1) as a microneme marker (A), rhoptry-associated protein 1 (RAP1) as a rhoptry body marker (B), rhoptry neck protein 2 (RON2) as a rhoptry neck marker (C), the ring-infected erythrocyte surface antigen (RESA) as a dense granule marker (D), MitoTrackerRed CMXRos as a mitochondria marker (E), ACPGFP (F–H) and the organellar histone-like protein PfHU (H) as an apicoplast marker were used. Scale bar, 1 mm.Kitamura K, Kishi-Itakura C, Tsuboi T, Sato S, Kita K, Ohta N, Mizushima N. Autophagy-Related Atg8 Localizes to the Apicoplast of the Human Malaria Parasite Plasmodium falciparum. PLoS One. 2012;7(8):e42977.
See original on MMP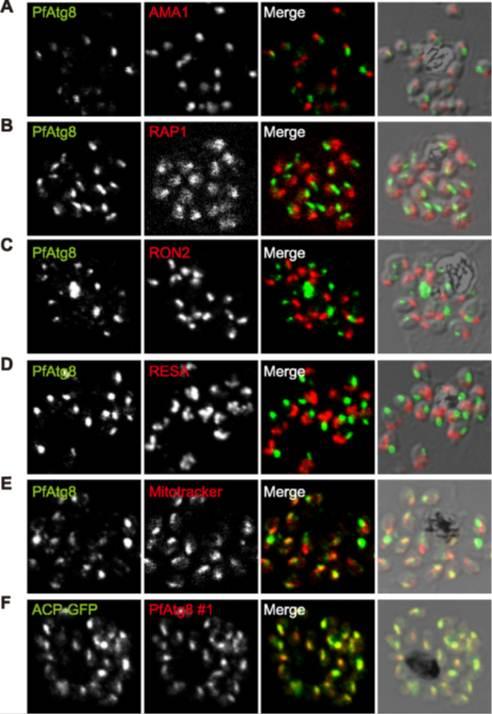
PfAtg8 localizes to the apicoplast. P. falciparum FCR3 (A–E) and P. falciparum 3D7 transfected with ACP-GFP (F–H) were stained with the indicated organelle markers and visualized by confocal microscopy (because ACP-GFP was not uniformly expressed, some merozoites displayed only faint GFP signals). Anti-PfAtg8 antibody #1 was used in (A–F), and anti-PfAtg8 antibody #2 was used in (G). Apical membrane antigen 1 (AMA1) as a microneme marker (A), rhoptry-associated protein 1 (RAP1) as a rhoptry body marker (B), rhoptry neck protein 2 (RON2) as a rhoptry neck marker (C), the ring-infected erythrocyte surface antigen (RESA) as a dense granule marker (D), MitoTrackerRed CMXRos as a mitochondria marker (E), ACPGFP (F–H) and the organellar histone-like protein PfHU (H) as an apicoplast marker were used. Scale bar, 1 mm.Kitamura K, Kishi-Itakura C, Tsuboi T, Sato S, Kita K, Ohta N, Mizushima N. Autophagy-Related Atg8 Localizes to the Apicoplast of the Human Malaria Parasite Plasmodium falciparum. PLoS One. 2012;7(8):e42977
See original on MMP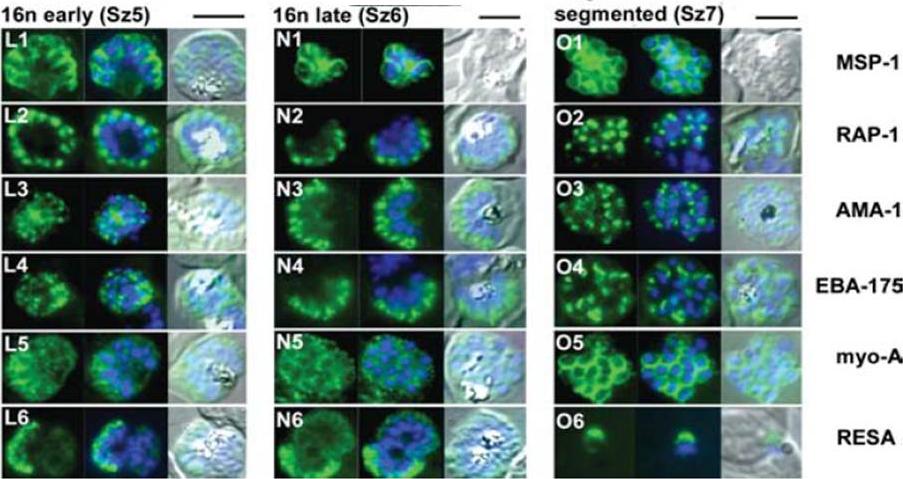
The appearance of Plasmodium falciparum schizonts of the ITO4 line at different times of development. The time of sampling from invasion and the number of nuclei per schizont are indicated. Antibodies to proteins associated with invasion related organelles or the parasite periphery (rhoptries : RAP-1; micronemes: AMA-1, EBA-175; dense granules: RESA; parasite periphery: MSP-1, myo-A) were chosen to follow the development and assembly of merozoites in maturing schizonts.Margos G, Bannister LH, Dluzewski AR, Hopkins J, Williams IT, Mitchell GH. Correlation of structural development and differential expression of invasion-related molecules in schizonts of Plasmodium falciparum. Parasitology. 2004 129:273-87. Copyright Cambridge University Press Journals 2011
See original on MMP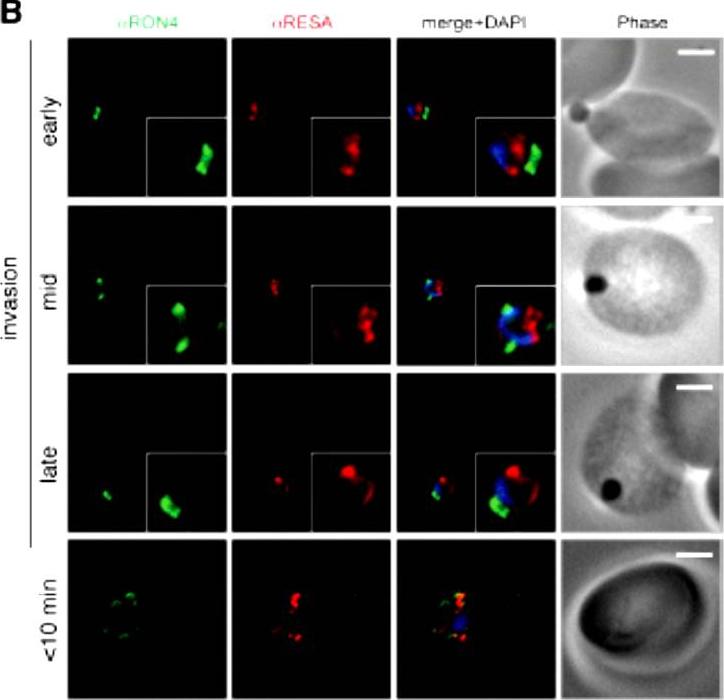
Wide field IFA time course of invasion using anti-RESA/PfRON4. Scale bar = 2.0 μm. Pre-egress shows standard wide-field image. Invasion images show IFA with deconvolution (single slice). RESA is retained within the merozoite body, often at the apical end but frequently basal to rhoptries and micronemes (RESA early). RESA, in contrast, retained its position inside the merozoite, establishing that release of dense granule proteins likely occurs postinvasion (RESA mid). RESA, localization became markedly more peripheral and evenly distributed around the merozoite immediately after invasion (RESA late), supporting the notion that dense granules are released after invasion at the merozoite plasma membrane.Riglar DT, Richard D, Wilson DW, Boyle MJ, Dekiwadia C, Turnbull L, Angrisano F, Marapana DS, Rogers KL, Whitchurch CB, Beeson JG, Cowman AF, Ralph SA, Baum J. Super-resolution dissection of coordinated events during malaria parasite invasion of the human erythrocyte. Cell Host Microbe. 2011 9:9-20.
See original on MMP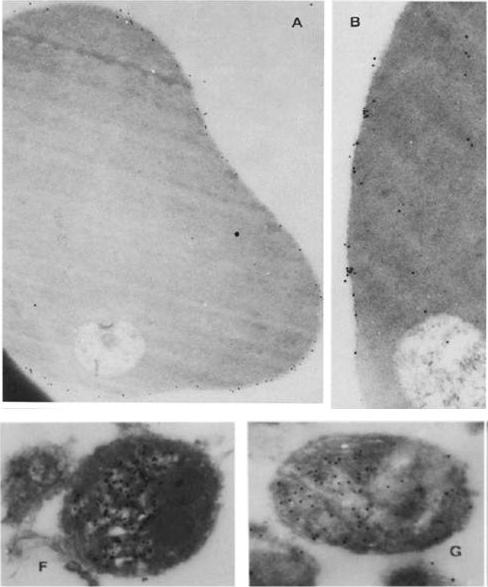
Immunoelectron micrographs showing sections of ring-infected erythrocytes reacted first with rabbit anti-RESA (A) or ~2 mg/ml of affinity-purified human anti-RESA antibodies (B), then with protein A gold; other sections of merozoites were reacted with rabbit anti-RESA (C-F) or 0.5 mg/ml human anti-RESA antibodies (C), and then protein A gold. (A) X 35,600; (B) X 56,500; (F) X 59,200; (G) X 57,500).Brown GV, Culvenor JG, Crewther PE, Bianco AE, Coppel RL, Saint RB, Stahl HD, Kemp DJ, Anders RF. Localization of the ring-infected erythrocyte surface antigen (RESA) of Plasmodium falciparum in merozoites and ring-infected erythrocytes. J Exp Med. 1985 162:774-9.
See original on MMP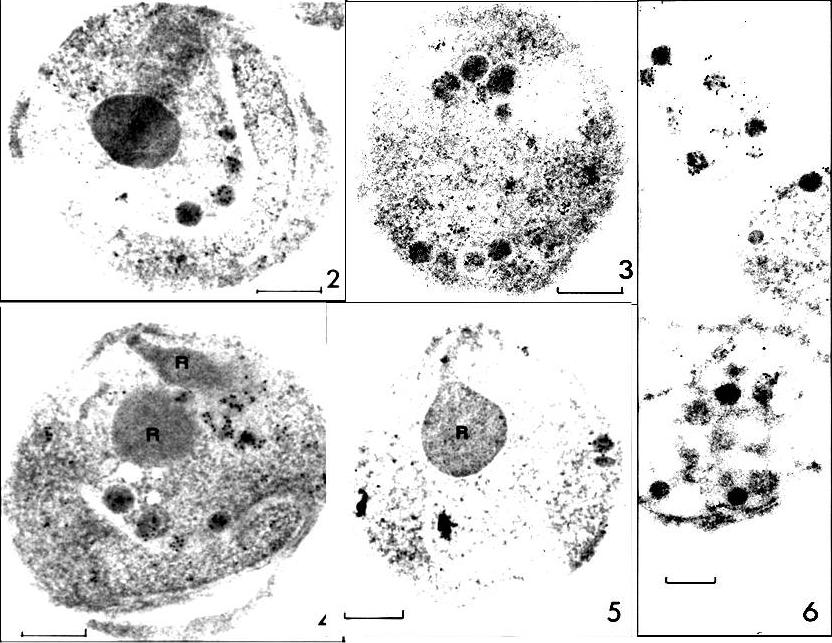
RESA labeling of P. falciparum merozoite dense granules (protein A-5-nm gold; bars, 0.2 ,mm). FIG. 2 and 3. Sections of D10 merozoites with labeled dense granules after incubation with MAb23/9. FIG. 4 and 5. Sections of D10 merozoites with rhoptries (R) in view after incubation with human anti-RESA, preparation H145. FIG. 6. Disrupted D10 merozoites with labeled dense granules (MAb23/9). ports. The studies described here show that P. falciparum RESA is localized to dense round granules (of up to 100 nm in diameter) in merozoites and to similar-sized aggregates of amorphous material in the parasitophorous vacuole of newly invaded erythrocytes.Culvenor JG, Day KP, Anders RF. Plasmodium falciparum ring-infected erythrocyte surface antigen is released from merozoite dense granules after erythrocyte invasion. Infect Immun. 1991 59:1183-1187.
See original on MMP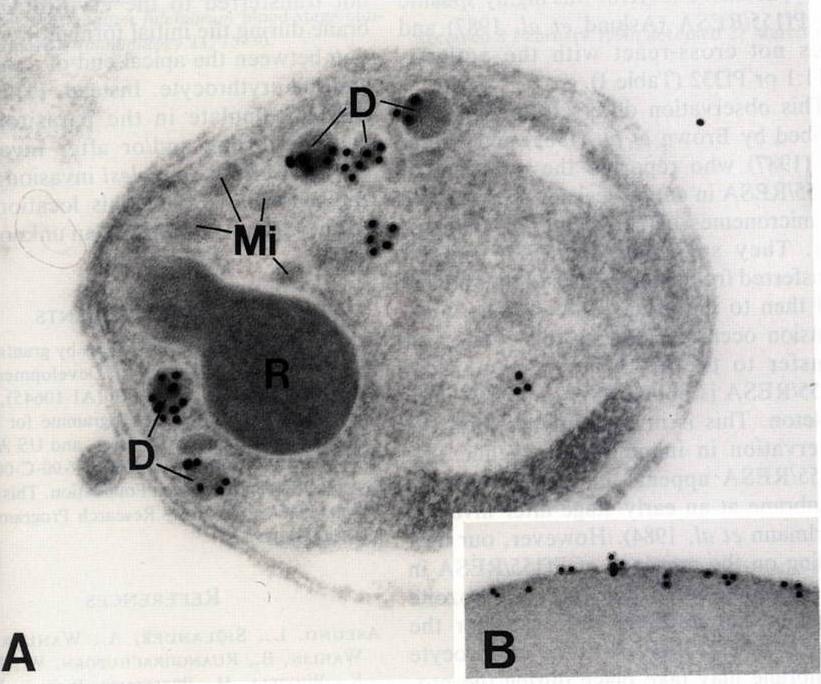
A) Free merozoite. Gold particles which identify the presence of Pf155/RESA are only associated with dense granules (D). Label is not present on rhoptries (R) or micronemes (Mi). X67,000. B) Immunoelectron micrograph showing gold particles on the ring infected erythrocyte. This localization is characteristic of Pf155/RESA> x48,000.Aikawa M, Torii M, Sjölander A, Berzins K, Perlmann P, Miller LH. Pf155/RESA antigen is localized in dense granules of Plasmodium falciparum merozoites. Exp Parasitol. 1990 71:326-9. Copyright Elsevier 2010.
See original on MMP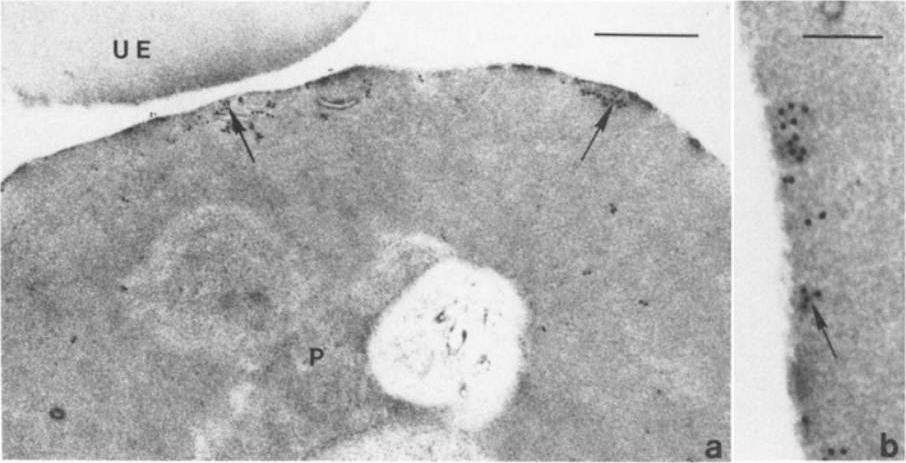
lmmunoelectron micro-graphs showing reactivity of mAb 33G2 (arrow) with P. falciparum (P)-infected erythrocytes. Note the absence of gold particles in an uninfected erythrocyte (UE). Bars: a. 1 mm: b. 0.25 mm. The localization of Ag reacting with mAb 3362 to the membrane of P. falciparum-infected erythrocytes was confirmed. The binding of the mAb was mainly confined to the area just under the erythrocyte membrane. Scattered clusters of labeling were also observed within the intracellular parasite as well as in the erythrocyte cytoplasm. mAb 3362 reacts with RESA and antigens 332 and Pf11.1.Udomsangpetch R, Carlsson J, Wåhlin B, Holmquist G, Ozaki LS, Scherf A, Mattei D, Mercereau-Puijalon O, Uni S, Aikawa M, Berzins K, Perlmann P. Reactivity of the human monoclonal antibody 33G2 with repeated sequences of three distinct Plasmodium falciparum antigens. J Immunol. 1989 142:3620-6. Copyright Journal of Immunology 2009.
See original on MMP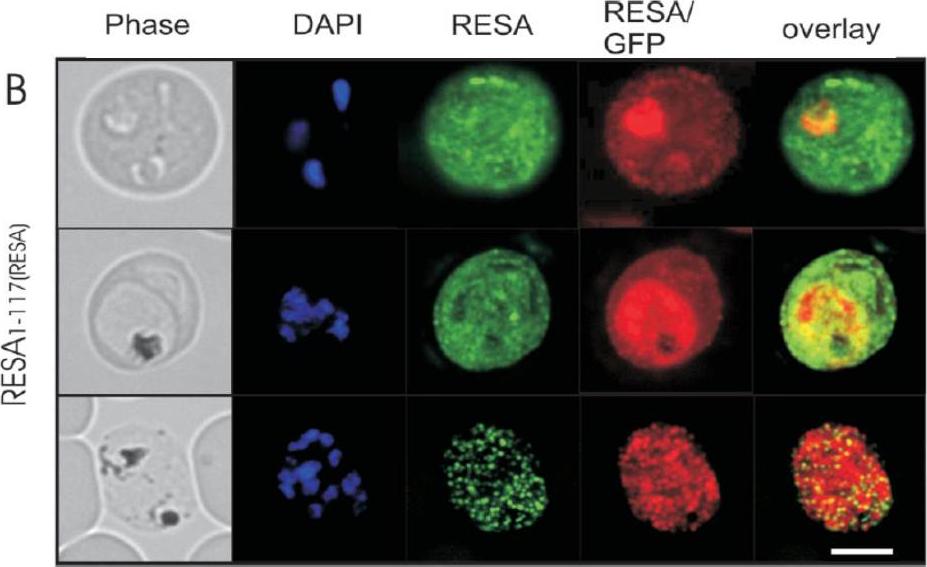
Immunofluorescence microscopy analysis of endogenous RESA and RESA1-117-GFP in transfected parasitized erythrocytes. Erythrocytes infected with RESA1-117-GFPRESA transfectants at the ring stage (top panels), trophozoite stage (middle panels), or schizont stage (bottom panels) were fixed with methanol (which destroys the intrinsic GFP fluorescence) and labeled with rabbit anti-GFP antiserum followed by an Alexa Fluor 568-conjugated anti-rabbit IgG (red fluorescence) and anti-RESA MAb, followed by fluorescein isothiocyanate-conjugated anti-mouse IgG (green fluorescence). Phase-contrast images are shown in the left panels. Overlays of the green and red fluorescence channels are shown in the right panels. Bar, 5 mm. In early-ring stage parasites, the anti-RESA antibody revealed some endogenous RESA associated with the PV which appeared to be relocated to the erythrocyte cytosol as the parasite matured. In schizont stage parasites, the antibody against endogenous RESA revealed a punctate fluorescence pattern that is consistent with labeling of dense granules in individual merozoites. Rug M, Wickham ME, Foley M, Cowman AF, Tilley L. Correct promoter control is needed for trafficking of the ring-infected erythrocyte surface antigen to the host cytosol in transfected malaria parasites. Infect Immun. 2004 72:6095-105.
See original on MMP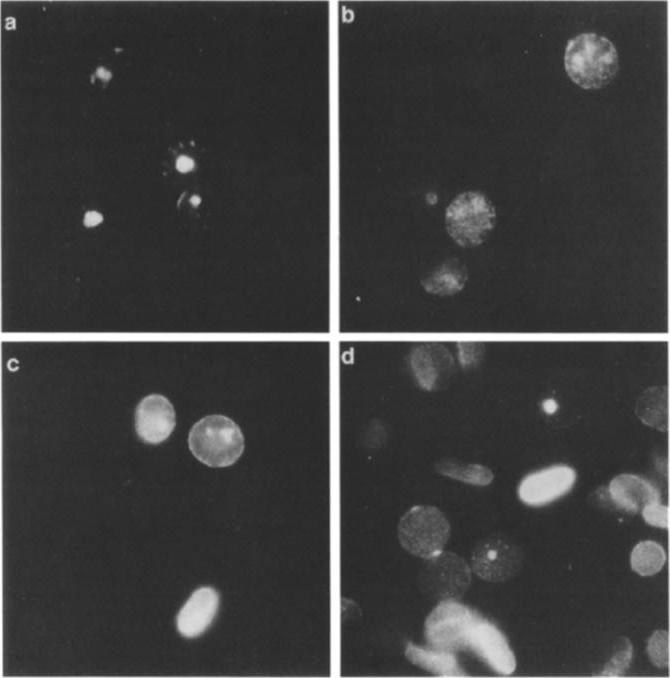
Indirect immunofluorescence using P. falciparum-infected erythrocytes glutaraldehyde fixed in suspension only (a) or subsequently treated with 0.1% saponin (b) or 10% Triton X-100 (c, d). It was previously shown that Pfl55/RESA is not accessible for reaction with antibodies on the surface of intact P. falciparum-infected erythrocytes unless the membrane is modified, e.g., by glutaraldehyde fixation and air dryingProbing was done with the human MAb 3362 (a-c) or the mouse Mab 4C8F6 directed against a cytoplasmic epitope of band 3 (d). The samples were counterstained with ethidium bromide to visualize the intracellular parasites.Ruangjirachuporn W, Udomsangpetch R, Carlsson J, Drenckhahn D, Perlmann P, Berzins K. Plasmodium falciparum: analysis of the interaction of antigen Pf155/RESA with the erythrocyte membrane. Exp Parasitol. 1991 73:62-72. Copyright Elsevier 2010.
See original on MMP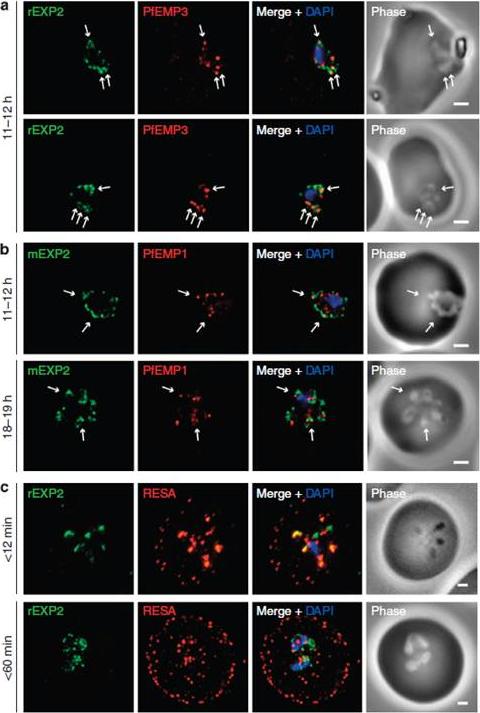
Protein accumulates at sites marked by PTEX during native PEXEL-dependent protein export. Widefield deconvolution microscopy of parasites fixed and labelled by IFA for EXP2 (green), the nucleus (40,6–diamidino-2-phenylindole (DAPI), blue) and (a) PfEMP3 (red), a protein exported by PEXEL-dependent processes, or (b) PfEMP1, a protein likely to undergo PEXEL-independent export, showed some areas of possible association of both exported proteins and EXP2; however, results were inconclusive. Parasite ages are as shown. Scale bars, 1 mm. Parasites were labelled by IFA for EXP2 (green), RESA (red) and the nucleus (DAPI, blue) having been (c) fixed o12 min or 60–90 min after invasion and imaged by widefield deconvolution microscopy (scale bar, 1 mm).Riglar DT, Rogers KL, Hanssen E, Turnbull L, Bullen HE, Charnaud SC, Przyborski J, Gilson PR, Whitchurch CB, Crabb BS, Baum J, Cowman AF. Spatial association with PTEX complexes defines regions for effector export into Plasmodium falciparum-infected erythrocytes. Nat Commun. 2013 Jan 29;4:1415.
See original on MMP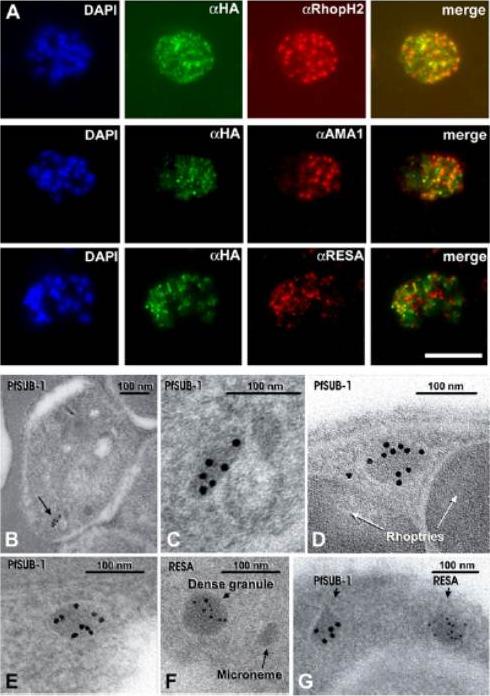
(A) Schizonts of 3D7SUB1HA3 clone C10 dual-labeled with the anti-HA mAb 3F10 (aHA; green) plus mAb 61.3 (aRhopH2), mAb 4G2 (aPfAMA1; micronemes, or mAb 28/2 (aRESA; dense granules. Nuclei were stained with DAPI (blue). Merged images (no DAPI) show that PfSUB1 localizes with none of the other markers. The scale bar represents 5 mm. Identical results were obtained with 3D7SUB1HA3 clone F7 or by using mAbs specific for RAP2 or EBA-175 as markers for rhoptries and micronemes respectively (data not shown). (B–G) Immunoelectron microscopic localization of PfSUB1 in P. falciparum schizonts. (B and C) Labeling of an elongated organelle, arrowed in (B) and at higher magnification in (C), by anti-PfSUB1 antibodies labeled with 10 nm immunogold. (D and E) Two more examples of PfSUB1 positive organelles are shown, also labeled with 10 nm immunogold. These are less elongate than in (B) and (C), reflecting some variation in organelle shape; however, they are typically ellipsoidal, in contrast with the rounded dense granules. (F and G) Staining for RESA with mAb 28/2 and 5 nm immunogold. In (F), an unlabeled microneme is also shown for size comparison. In (G) double staining for PfSUB1 and RESA with different sizes of immunogold shows that the two proteins are in different organelles.Yeoh S, O'Donnell RA, Koussis K, Dluzewski AR, Ansell KH, Osborne SA, Hackett F, Withers-Martinez C, Mitchell GH, Bannister LH, Bryans JS, Kettleborough CA, Blackman MJ. Subcellular discharge of a serine protease mediates release of invasive malaria parasites from host erythrocytes. Cell. 2007 131:1072-83.
See original on MMP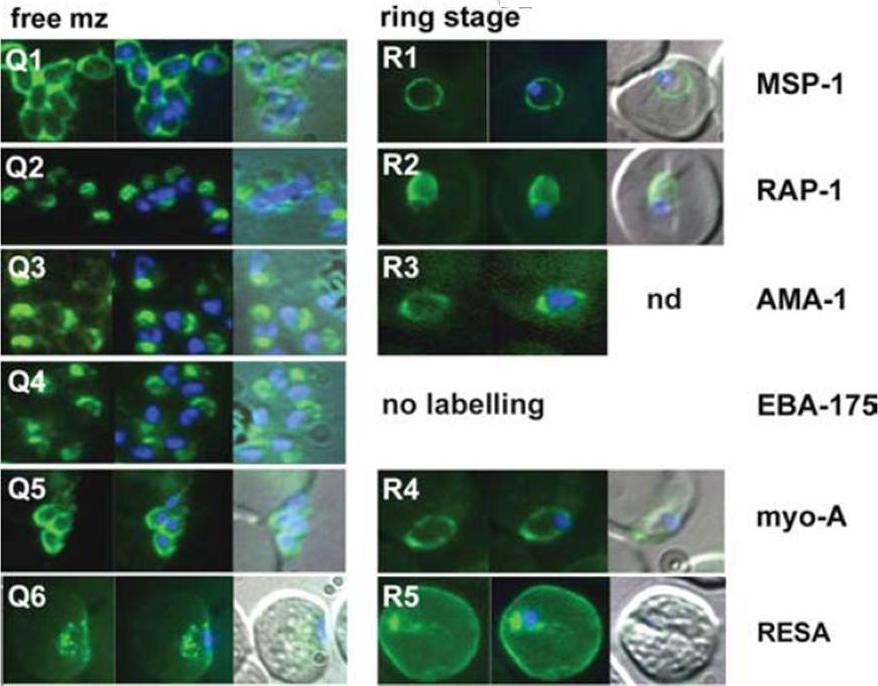
The appearance of Plasmodium falciparum merozoites and rings of the ITO4 line at different times of development. Antibodies to proteins associated with invasion related organelles or the parasite periphery (rhoptries : RAP-1; micronemes: AMA-1, EBA-175; dense granules: RESA; parasite periphery: MSP-1, myo-A) were chosen to follow the development and assembly of merozoites and rings.Margos G, Bannister LH, Dluzewski AR, Hopkins J, Williams IT, Mitchell GH. Correlation of structural development and differential expression of invasion-related molecules in schizonts of Plasmodium falciparum. Parasitology. 2004 129:273-87. Copyright Cambridge University Press Journals 2011
See original on MMP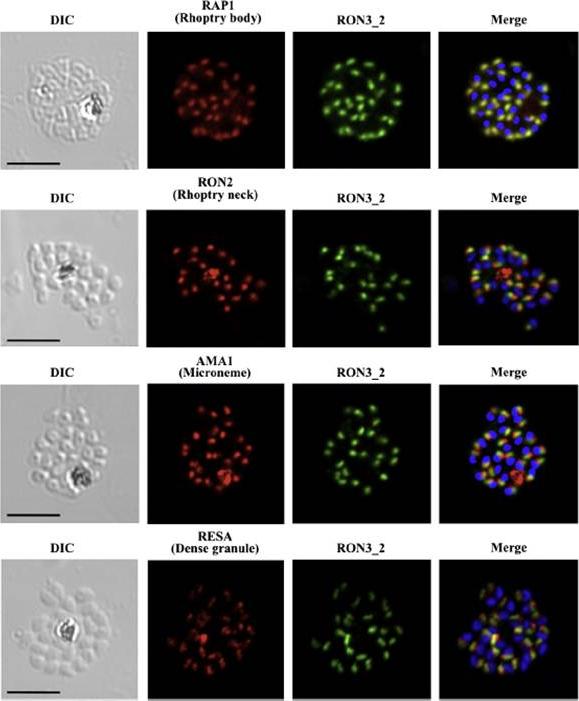
PfRON3 is expressed at the apical end of Plasmodium merozoites. Schizont and merozoite stage parasites were dual-labeled with antisera against PfRON3_2 and either PfRAP1 (rhoptry body marker), or PfRON2 (rhoptry neck marker), or PfAMA1 (microneme marker), or PfRESA (dense granule marker). Nuclei are visualized with DAPI in merged images shown in the right panels. Bars represent 5 μm. PfRON3 is localized to the but in the rhoptry body.Ito D, Han ET, Takeo S, Thongkukiatkul A, Otsuki H, Torii M, Tsuboi T. Plasmodial ortholog of Toxoplasma gondii rhoptry neck protein 3 is localized to the rhoptry body. Parasitol Int. 2011 60(2):132-8.
See original on MMP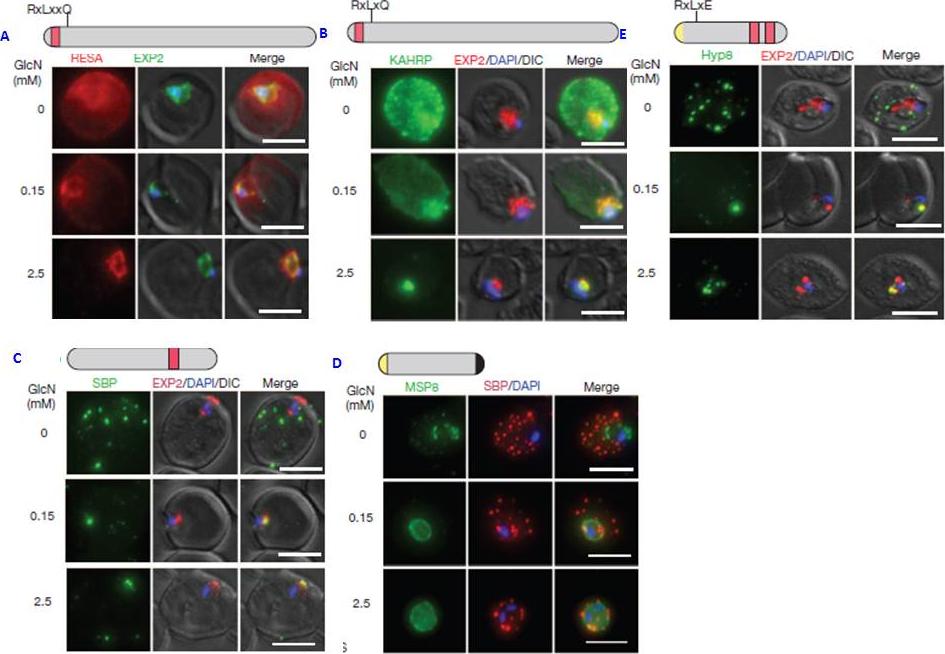
PTEX150 knockdown blocks protein export in P. falciparum. a–e, IFAs (right) and graphs (left) showing a decrease in the export of RESA (a) and KAHRP (b) and of SBP1 (c) and Hyp8 (d) (Maurer’s clefts, MCs) cells for each antibody or GlcN concentration) but similar levels of MSP8 (e) after treatment with GlcN. Scale bars, 5 mm. A strong blockage in export was seen at both 2.5mM and 0.15mM glucosamine in PTEX150-HAglmS parasites (a, b, c and d). No effect on either MSP8 expression or localization to the parasite membrane was observed (e).Elsworth B, Matthews K, Nie CQ, Kalanon M, Charnaud SC, Sanders PR, Chisholm SA, Counihan NA, Shaw PJ, Pino P, Chan JA, Azevedo MF, Rogerson SJ, Beeson JG, Crabb BS, Gilson PR, de Koning-Ward TF. PTEX is an essential nexus for protein export in malaria parasites. Nature. 2014 Jul 16. [Epub ahead of print] PMID: 25043043
See original on MMP
A. Domain architecture of PF3D7 0102200 (RESA), not to scale. PRESAN, domain boundaries defined according to Pfam ID: PF09687; DnaJ domain as defined byPfam ID: PF14308; SBD, spectrin-binding domain. (B–E) Domain architecture and localisation of HT:RESA:GFP reporter constructs. REX31–61, N-terminal sequence of REX3,including its HT motif, which is sufficient to drive protein export. Residue numbers, below each diagram, refer to position within full-length RESA. The left- and right-hand images show GFP localisation and a phase contrast image, respectively. Scale bar, 2 mm. The identity of each parasite line is indicated above the respective images. the extended PRESAN domain of RESA, a known cytoskeleton-associated protein, efficiently targets to the erythrocyte periphery.Tarr SJ, Moon RW, Hardege I, Osborne AR. A conserved domain targets exported PHISTb family proteins to the periphery of Plasmodium infected erythrocytes. Mol Biochem Parasitol. 2014 196(1):29-40
See original on MMP
Time course analysis of PfGEXP5 expression. (b) Representative immunofluorescence images of (top panel) a ring stage-like parasite at 14 h pi positive for PfGEXP5 (green) and negative for Pfg27 (red), and (lower panel) a pigmented double-positive parasite at 38 h pi. (c) Immunofluorescence microscopy of ~18-h gametocyte and asexaul ring-infected RBCs labelled with anti-PfGEXP5 (green) and anti-RESA (red) antibodies. Brightfield images (BF) and nuclear staining are shown at right and left, respectively. Scale bar 5 μm.Tibúrcio M, Dixon MW, Looker O, Younis SY, Tilley L, Alano P. Specific expression and export of the Plasmodium falciparum Gametocyte EXported Protein-5 marks the gametocyte ring stage. Malar J. 2015 14:334. PMID:
See original on MMP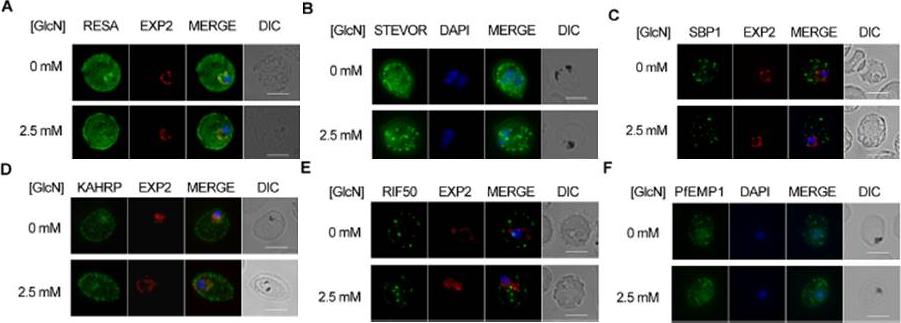
Effect of PTEX88 knockdown on P. falciparum protein export. Immunofluorescence assays showing that exported proteins A: RESA, B: STEVOR, C: SBP1, D: KAHRP, E: RIF50, F: PfEMP1 are still exported after knockdown of PTEX88. RESA (an early expressed PEXEL protein), STEVOR (a PEXEL protein containing transmembrane domains), skeleton binding protein 1 (SBP1; a PNEP), KAHRP (a soluble PEXEL protein), RIF50 (a PEXEL protein containing transmembrane domains) and PfEMP1 (a PNEP containing a transmembrane domain that is exported to the erythrocyte surface.Chisholm SA, McHugh E, Lundie R, Dixon MW, Ghosh S, O'Keefe M, Tilley L, Kalanon M, de Koning-Ward TF. Contrasting Inducible Knockdown of the Auxiliary PTEX Component PTEX88 in P. falciparum and P. berghei Unmasks a Role in Parasite Virulence. PLoS One. 2016 Feb 17;11(2):e0149296.
See original on MMP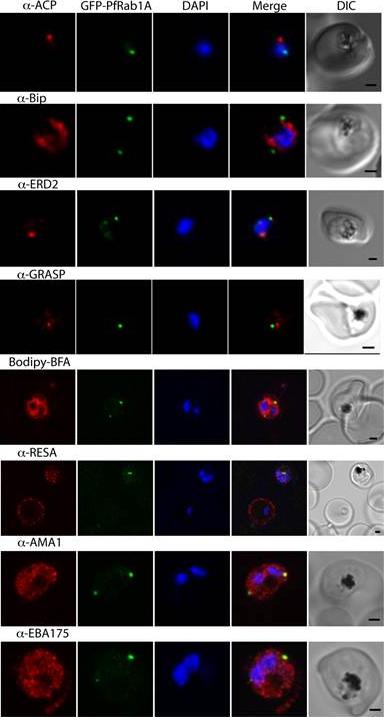
GFP-PfRab1A fluorescence does not co-localize with ER/Golgi, dense granules or microneme markers. GFP-PfRab1A fluorescence is distinct from the localization of markers for the apicoplast (ACP), the ER (Bip), the Golgi (ERD2 and GRASP), as well as from staining of the ER/Golgi with Bodipy BFA. GFP-Rab1A fluorescence is also distinct from the localization of markers for dense granules (RESA) or micronemes (AMA1 and EBA175).Morse D, Webster W, Kalanon M, Langsley G, McFadden GI. Plasmodium falciparum Rab1A Localizes to Rhoptries in Schizonts. PLoS One. 2016 Jun 27;11(6):e0158174
See original on MMP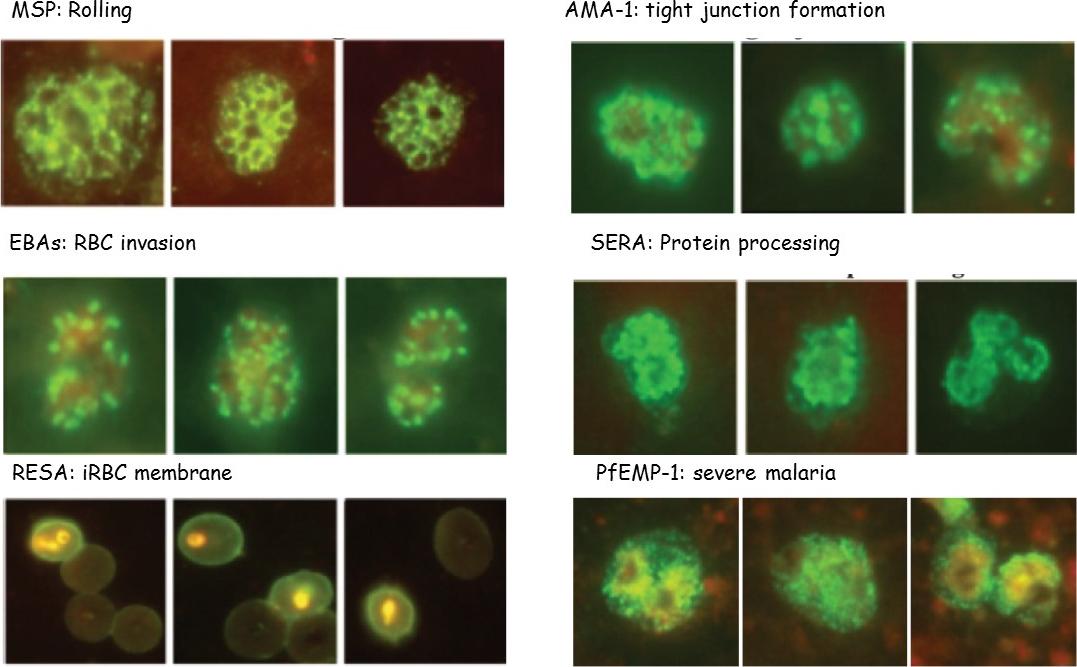
P. falciparum merozoite protein location is shown, as determined by IFA and functions: rolling on the membrane by MSPs, TJ formation mediated by AMA-1-and apical rhoptries proteins, RBC deformation and invasion by microneme EBAs, protein processing by intracytoplasmic SERA-5, iRBC membrane expression by RESA and severe malaria (SM) and echinocytosis by PfEMP-1.Patarroyo ME, Arevalo-Pinzon G, Reyes C, Moreno-Vranich A, Patarroyo MA. Malaria Parasite Survival Depends on Conserved Binding Peptides' Critical Biological Functions. Curr Issues Mol Biol. 2016;18:57-78.
See original on MMP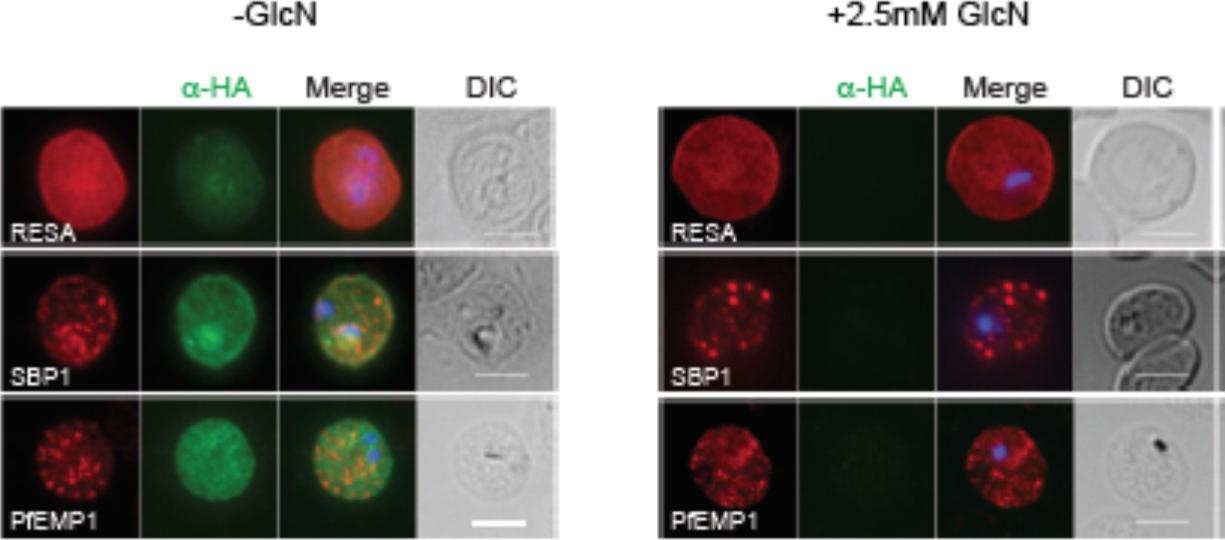
RhopH2 is not involved in the trafficking of exported proteins in the erythrocyte cytoplasm. (a) Representative IFAs of erythrocytes infected with RhopH2-HAglmS parasites grown in 0 mM or 2.5 mM GlcN using the indicated antibodies show trafficking of RESA, SBP1 and PfEMP1 is unaffected upon RhopH2 knockdown. Scale bar = 5 μm. No defect in the export of PfEMP1, or trafficking of either RESA to the erythrocyte membrane or SBP1 to the Maurer’s clefts was evident after knocking down RhopH2 expression with GlcN.Counihan N, Chisholm SA, Bullen HE, Srivastava A, Sanders PR, Jonsdottir TK, Weiss GE, Ghosh S, Crabb BS, Creek DJ, Gilson PR, de Koning-Ward TF. Plasmodium falciparum parasites deploy RhopH2 into the host erythrocyte to obtain nutrients, grow and replicate. Elife. 2017 Mar 2;6. pii: e23217.
See original on MMP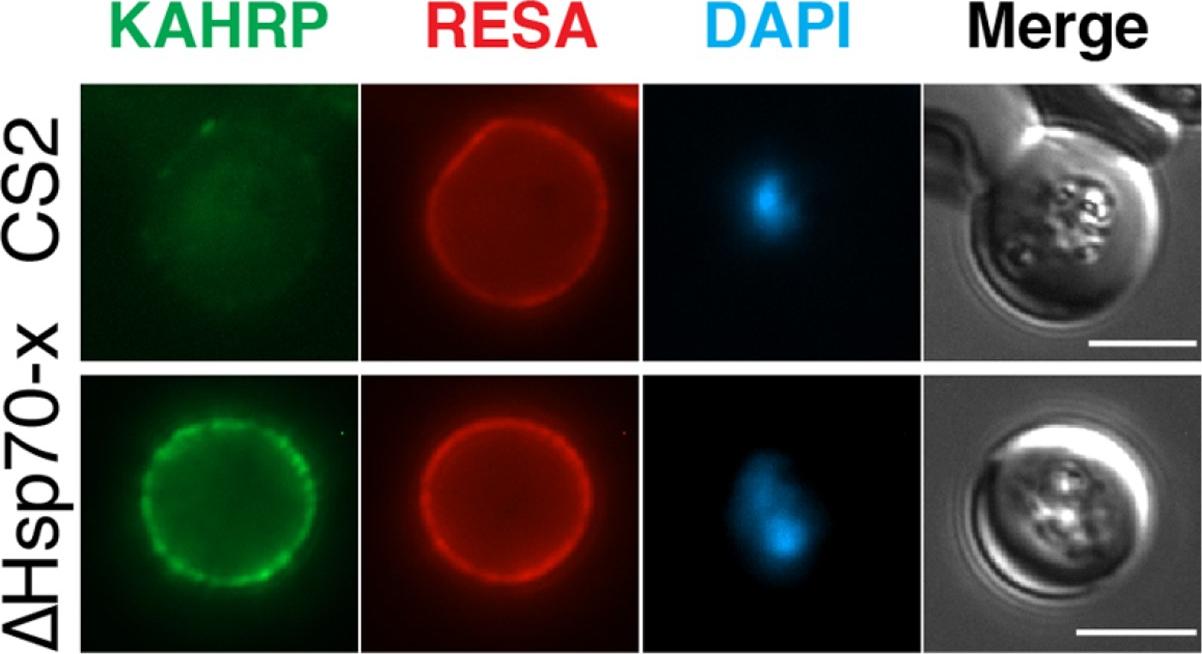
The expression of cytoskeletal binding proteins KAHRP is increased in ΔHsp70-x. (a) Immuno fluorescence microscopy of 16-24 hpi parasites indicates expression of the exported, knob localized KAHRP protein but not RESA appears higher in ΔHsp70-x. Antibodies to the cytoskeletal binding proteins ring exported surface antigen (RESA) and knob associated histidine rich protein (KAHRP) indicated the MFI was significantly higher for KAHRP but not RESA in the mutant. The CS2 parasite line in which the ΔHsp70-x clones were derived from stably expresses the single VAR2CSA PfEMP1 allele, which binds to chondroitin sulfate A (CSA) normally present in the placenta.Charnaud SC, Dixon MWA, Nie CQ, Chappell L, Sanders PR, Nebl T, Hanssen E, Berriman M, Chan JA, Blanch AJ, Beeson JG, Rayner JC, Przyborski JM, Tilley L, Crabb BS, Gilson PR. The exported chaperone Hsp70-x supports virulence functions for Plasmodium falciparum blood stage parasites. PLoS One. 2017 12(7):e0181656.
See original on MMP
PfEH1 and PfEH2 interact with the erythrocyte cytoskeleton. IFA of paraformaldehyde/ glutaraldehyde-fixed, trophozoite/early schizont-stage parasites expressing PfEH1-GFP (A) or PfEH2-GFP (B) under control of the respective endogenous promoter. The GFP-expressing parasites were costained with anti-RESA (red). The nuclei were stained with DAPI (blue). Bars = 5 mm. DIC, differential interference contrast. In asexual parasites, anti-GFP antibody detected PfEH1 and PfEH2 at the periphery of the infected erythrocyte and within the parasite. The fluorescence at the periphery colocalized with ring-exported surface antigen (RESA), an exported protein that interacts with host spectrin at the RBC cytoskeleton.Spillman NJ, Dalmia VK, Goldberg DE. Exported Epoxide Hydrolases Modulate Erythrocyte Vasoactive Lipids during Plasmodium falciparum Infection. MBio. 2016 Oct 18;7(5). pii: e01538-16.
See original on MMP
PfAtg8 localizes to the apicoplast. P. falciparum FCR3 (A–E) and P. falciparum 3D7 transfected with ACP-GFP (F–H) were stained with the indicated organelle markers and visualized by confocal microscopy (because ACP-GFP was not uniformly expressed, some merozoites displayed only faint GFP signals). Anti-PfAtg8 antibody #1 was used in (A–F), and anti-PfAtg8 antibody #2 was used in (G). Apical membrane antigen 1 (AMA1) as a microneme marker (A), rhoptry-associated protein 1 (RAP1) as a rhoptry body marker (B), rhoptry neck protein 2 (RON2) as a rhoptry neck marker (C), the ring-infected erythrocyte surface antigen (RESA) as a dense granule marker (D), MitoTrackerRed CMXRos as a mitochondria marker (E), ACPGFP (F–H) and the organellar histone-like protein PfHU (H) as an apicoplast marker were used. Scale bar, 1 mm.Kitamura K, Kishi-Itakura C, Tsuboi T, Sato S, Kita K, Ohta N, Mizushima N. Autophagy-Related Atg8 Localizes to the Apicoplast of the Human Malaria Parasite Plasmodium falciparum. PLoS One. 2012;7(8):e42977.
See original on MMP
PfAtg8 localizes to the apicoplast. P. falciparum FCR3 (A–E) and P. falciparum 3D7 transfected with ACP-GFP (F–H) were stained with the indicated organelle markers and visualized by confocal microscopy (because ACP-GFP was not uniformly expressed, some merozoites displayed only faint GFP signals). Anti-PfAtg8 antibody #1 was used in (A–F), and anti-PfAtg8 antibody #2 was used in (G). Apical membrane antigen 1 (AMA1) as a microneme marker (A), rhoptry-associated protein 1 (RAP1) as a rhoptry body marker (B), rhoptry neck protein 2 (RON2) as a rhoptry neck marker (C), the ring-infected erythrocyte surface antigen (RESA) as a dense granule marker (D), MitoTrackerRed CMXRos as a mitochondria marker (E), ACPGFP (F–H) and the organellar histone-like protein PfHU (H) as an apicoplast marker were used. Scale bar, 1 mm.Kitamura K, Kishi-Itakura C, Tsuboi T, Sato S, Kita K, Ohta N, Mizushima N. Autophagy-Related Atg8 Localizes to the Apicoplast of the Human Malaria Parasite Plasmodium falciparum. PLoS One. 2012;7(8):e42977
See original on MMP
The appearance of Plasmodium falciparum schizonts of the ITO4 line at different times of development. The time of sampling from invasion and the number of nuclei per schizont are indicated. Antibodies to proteins associated with invasion related organelles or the parasite periphery (rhoptries : RAP-1; micronemes: AMA-1, EBA-175; dense granules: RESA; parasite periphery: MSP-1, myo-A) were chosen to follow the development and assembly of merozoites in maturing schizonts.Margos G, Bannister LH, Dluzewski AR, Hopkins J, Williams IT, Mitchell GH. Correlation of structural development and differential expression of invasion-related molecules in schizonts of Plasmodium falciparum. Parasitology. 2004 129:273-87. Copyright Cambridge University Press Journals 2011
See original on MMP
Wide field IFA time course of invasion using anti-RESA/PfRON4. Scale bar = 2.0 μm. Pre-egress shows standard wide-field image. Invasion images show IFA with deconvolution (single slice). RESA is retained within the merozoite body, often at the apical end but frequently basal to rhoptries and micronemes (RESA early). RESA, in contrast, retained its position inside the merozoite, establishing that release of dense granule proteins likely occurs postinvasion (RESA mid). RESA, localization became markedly more peripheral and evenly distributed around the merozoite immediately after invasion (RESA late), supporting the notion that dense granules are released after invasion at the merozoite plasma membrane.Riglar DT, Richard D, Wilson DW, Boyle MJ, Dekiwadia C, Turnbull L, Angrisano F, Marapana DS, Rogers KL, Whitchurch CB, Beeson JG, Cowman AF, Ralph SA, Baum J. Super-resolution dissection of coordinated events during malaria parasite invasion of the human erythrocyte. Cell Host Microbe. 2011 9:9-20.
See original on MMP
Immunoelectron micrographs showing sections of ring-infected erythrocytes reacted first with rabbit anti-RESA (A) or ~2 mg/ml of affinity-purified human anti-RESA antibodies (B), then with protein A gold; other sections of merozoites were reacted with rabbit anti-RESA (C-F) or 0.5 mg/ml human anti-RESA antibodies (C), and then protein A gold. (A) X 35,600; (B) X 56,500; (F) X 59,200; (G) X 57,500).Brown GV, Culvenor JG, Crewther PE, Bianco AE, Coppel RL, Saint RB, Stahl HD, Kemp DJ, Anders RF. Localization of the ring-infected erythrocyte surface antigen (RESA) of Plasmodium falciparum in merozoites and ring-infected erythrocytes. J Exp Med. 1985 162:774-9.
See original on MMP
RESA labeling of P. falciparum merozoite dense granules (protein A-5-nm gold; bars, 0.2 ,mm). FIG. 2 and 3. Sections of D10 merozoites with labeled dense granules after incubation with MAb23/9. FIG. 4 and 5. Sections of D10 merozoites with rhoptries (R) in view after incubation with human anti-RESA, preparation H145. FIG. 6. Disrupted D10 merozoites with labeled dense granules (MAb23/9). ports. The studies described here show that P. falciparum RESA is localized to dense round granules (of up to 100 nm in diameter) in merozoites and to similar-sized aggregates of amorphous material in the parasitophorous vacuole of newly invaded erythrocytes.Culvenor JG, Day KP, Anders RF. Plasmodium falciparum ring-infected erythrocyte surface antigen is released from merozoite dense granules after erythrocyte invasion. Infect Immun. 1991 59:1183-1187.
See original on MMP
A) Free merozoite. Gold particles which identify the presence of Pf155/RESA are only associated with dense granules (D). Label is not present on rhoptries (R) or micronemes (Mi). X67,000. B) Immunoelectron micrograph showing gold particles on the ring infected erythrocyte. This localization is characteristic of Pf155/RESA> x48,000.Aikawa M, Torii M, Sjölander A, Berzins K, Perlmann P, Miller LH. Pf155/RESA antigen is localized in dense granules of Plasmodium falciparum merozoites. Exp Parasitol. 1990 71:326-9. Copyright Elsevier 2010.
See original on MMP
lmmunoelectron micro-graphs showing reactivity of mAb 33G2 (arrow) with P. falciparum (P)-infected erythrocytes. Note the absence of gold particles in an uninfected erythrocyte (UE). Bars: a. 1 mm: b. 0.25 mm. The localization of Ag reacting with mAb 3362 to the membrane of P. falciparum-infected erythrocytes was confirmed. The binding of the mAb was mainly confined to the area just under the erythrocyte membrane. Scattered clusters of labeling were also observed within the intracellular parasite as well as in the erythrocyte cytoplasm. mAb 3362 reacts with RESA and antigens 332 and Pf11.1.Udomsangpetch R, Carlsson J, Wåhlin B, Holmquist G, Ozaki LS, Scherf A, Mattei D, Mercereau-Puijalon O, Uni S, Aikawa M, Berzins K, Perlmann P. Reactivity of the human monoclonal antibody 33G2 with repeated sequences of three distinct Plasmodium falciparum antigens. J Immunol. 1989 142:3620-6. Copyright Journal of Immunology 2009.
See original on MMP
Immunofluorescence microscopy analysis of endogenous RESA and RESA1-117-GFP in transfected parasitized erythrocytes. Erythrocytes infected with RESA1-117-GFPRESA transfectants at the ring stage (top panels), trophozoite stage (middle panels), or schizont stage (bottom panels) were fixed with methanol (which destroys the intrinsic GFP fluorescence) and labeled with rabbit anti-GFP antiserum followed by an Alexa Fluor 568-conjugated anti-rabbit IgG (red fluorescence) and anti-RESA MAb, followed by fluorescein isothiocyanate-conjugated anti-mouse IgG (green fluorescence). Phase-contrast images are shown in the left panels. Overlays of the green and red fluorescence channels are shown in the right panels. Bar, 5 mm. In early-ring stage parasites, the anti-RESA antibody revealed some endogenous RESA associated with the PV which appeared to be relocated to the erythrocyte cytosol as the parasite matured. In schizont stage parasites, the antibody against endogenous RESA revealed a punctate fluorescence pattern that is consistent with labeling of dense granules in individual merozoites. Rug M, Wickham ME, Foley M, Cowman AF, Tilley L. Correct promoter control is needed for trafficking of the ring-infected erythrocyte surface antigen to the host cytosol in transfected malaria parasites. Infect Immun. 2004 72:6095-105.
See original on MMP
Indirect immunofluorescence using P. falciparum-infected erythrocytes glutaraldehyde fixed in suspension only (a) or subsequently treated with 0.1% saponin (b) or 10% Triton X-100 (c, d). It was previously shown that Pfl55/RESA is not accessible for reaction with antibodies on the surface of intact P. falciparum-infected erythrocytes unless the membrane is modified, e.g., by glutaraldehyde fixation and air dryingProbing was done with the human MAb 3362 (a-c) or the mouse Mab 4C8F6 directed against a cytoplasmic epitope of band 3 (d). The samples were counterstained with ethidium bromide to visualize the intracellular parasites.Ruangjirachuporn W, Udomsangpetch R, Carlsson J, Drenckhahn D, Perlmann P, Berzins K. Plasmodium falciparum: analysis of the interaction of antigen Pf155/RESA with the erythrocyte membrane. Exp Parasitol. 1991 73:62-72. Copyright Elsevier 2010.
See original on MMP
Protein accumulates at sites marked by PTEX during native PEXEL-dependent protein export. Widefield deconvolution microscopy of parasites fixed and labelled by IFA for EXP2 (green), the nucleus (40,6–diamidino-2-phenylindole (DAPI), blue) and (a) PfEMP3 (red), a protein exported by PEXEL-dependent processes, or (b) PfEMP1, a protein likely to undergo PEXEL-independent export, showed some areas of possible association of both exported proteins and EXP2; however, results were inconclusive. Parasite ages are as shown. Scale bars, 1 mm. Parasites were labelled by IFA for EXP2 (green), RESA (red) and the nucleus (DAPI, blue) having been (c) fixed o12 min or 60–90 min after invasion and imaged by widefield deconvolution microscopy (scale bar, 1 mm).Riglar DT, Rogers KL, Hanssen E, Turnbull L, Bullen HE, Charnaud SC, Przyborski J, Gilson PR, Whitchurch CB, Crabb BS, Baum J, Cowman AF. Spatial association with PTEX complexes defines regions for effector export into Plasmodium falciparum-infected erythrocytes. Nat Commun. 2013 Jan 29;4:1415.
See original on MMP
(A) Schizonts of 3D7SUB1HA3 clone C10 dual-labeled with the anti-HA mAb 3F10 (aHA; green) plus mAb 61.3 (aRhopH2), mAb 4G2 (aPfAMA1; micronemes, or mAb 28/2 (aRESA; dense granules. Nuclei were stained with DAPI (blue). Merged images (no DAPI) show that PfSUB1 localizes with none of the other markers. The scale bar represents 5 mm. Identical results were obtained with 3D7SUB1HA3 clone F7 or by using mAbs specific for RAP2 or EBA-175 as markers for rhoptries and micronemes respectively (data not shown). (B–G) Immunoelectron microscopic localization of PfSUB1 in P. falciparum schizonts. (B and C) Labeling of an elongated organelle, arrowed in (B) and at higher magnification in (C), by anti-PfSUB1 antibodies labeled with 10 nm immunogold. (D and E) Two more examples of PfSUB1 positive organelles are shown, also labeled with 10 nm immunogold. These are less elongate than in (B) and (C), reflecting some variation in organelle shape; however, they are typically ellipsoidal, in contrast with the rounded dense granules. (F and G) Staining for RESA with mAb 28/2 and 5 nm immunogold. In (F), an unlabeled microneme is also shown for size comparison. In (G) double staining for PfSUB1 and RESA with different sizes of immunogold shows that the two proteins are in different organelles.Yeoh S, O'Donnell RA, Koussis K, Dluzewski AR, Ansell KH, Osborne SA, Hackett F, Withers-Martinez C, Mitchell GH, Bannister LH, Bryans JS, Kettleborough CA, Blackman MJ. Subcellular discharge of a serine protease mediates release of invasive malaria parasites from host erythrocytes. Cell. 2007 131:1072-83.
See original on MMP
The appearance of Plasmodium falciparum merozoites and rings of the ITO4 line at different times of development. Antibodies to proteins associated with invasion related organelles or the parasite periphery (rhoptries : RAP-1; micronemes: AMA-1, EBA-175; dense granules: RESA; parasite periphery: MSP-1, myo-A) were chosen to follow the development and assembly of merozoites and rings.Margos G, Bannister LH, Dluzewski AR, Hopkins J, Williams IT, Mitchell GH. Correlation of structural development and differential expression of invasion-related molecules in schizonts of Plasmodium falciparum. Parasitology. 2004 129:273-87. Copyright Cambridge University Press Journals 2011
See original on MMP
PfRON3 is expressed at the apical end of Plasmodium merozoites. Schizont and merozoite stage parasites were dual-labeled with antisera against PfRON3_2 and either PfRAP1 (rhoptry body marker), or PfRON2 (rhoptry neck marker), or PfAMA1 (microneme marker), or PfRESA (dense granule marker). Nuclei are visualized with DAPI in merged images shown in the right panels. Bars represent 5 μm. PfRON3 is localized to the but in the rhoptry body.Ito D, Han ET, Takeo S, Thongkukiatkul A, Otsuki H, Torii M, Tsuboi T. Plasmodial ortholog of Toxoplasma gondii rhoptry neck protein 3 is localized to the rhoptry body. Parasitol Int. 2011 60(2):132-8.
See original on MMP
PTEX150 knockdown blocks protein export in P. falciparum. a–e, IFAs (right) and graphs (left) showing a decrease in the export of RESA (a) and KAHRP (b) and of SBP1 (c) and Hyp8 (d) (Maurer’s clefts, MCs) cells for each antibody or GlcN concentration) but similar levels of MSP8 (e) after treatment with GlcN. Scale bars, 5 mm. A strong blockage in export was seen at both 2.5mM and 0.15mM glucosamine in PTEX150-HAglmS parasites (a, b, c and d). No effect on either MSP8 expression or localization to the parasite membrane was observed (e).Elsworth B, Matthews K, Nie CQ, Kalanon M, Charnaud SC, Sanders PR, Chisholm SA, Counihan NA, Shaw PJ, Pino P, Chan JA, Azevedo MF, Rogerson SJ, Beeson JG, Crabb BS, Gilson PR, de Koning-Ward TF. PTEX is an essential nexus for protein export in malaria parasites. Nature. 2014 Jul 16. [Epub ahead of print] PMID: 25043043
See original on MMP
A. Domain architecture of PF3D7 0102200 (RESA), not to scale. PRESAN, domain boundaries defined according to Pfam ID: PF09687; DnaJ domain as defined byPfam ID: PF14308; SBD, spectrin-binding domain. (B–E) Domain architecture and localisation of HT:RESA:GFP reporter constructs. REX31–61, N-terminal sequence of REX3,including its HT motif, which is sufficient to drive protein export. Residue numbers, below each diagram, refer to position within full-length RESA. The left- and right-hand images show GFP localisation and a phase contrast image, respectively. Scale bar, 2 mm. The identity of each parasite line is indicated above the respective images. the extended PRESAN domain of RESA, a known cytoskeleton-associated protein, efficiently targets to the erythrocyte periphery.Tarr SJ, Moon RW, Hardege I, Osborne AR. A conserved domain targets exported PHISTb family proteins to the periphery of Plasmodium infected erythrocytes. Mol Biochem Parasitol. 2014 196(1):29-40
See original on MMP
Time course analysis of PfGEXP5 expression. (b) Representative immunofluorescence images of (top panel) a ring stage-like parasite at 14 h pi positive for PfGEXP5 (green) and negative for Pfg27 (red), and (lower panel) a pigmented double-positive parasite at 38 h pi. (c) Immunofluorescence microscopy of ~18-h gametocyte and asexaul ring-infected RBCs labelled with anti-PfGEXP5 (green) and anti-RESA (red) antibodies. Brightfield images (BF) and nuclear staining are shown at right and left, respectively. Scale bar 5 μm.Tibúrcio M, Dixon MW, Looker O, Younis SY, Tilley L, Alano P. Specific expression and export of the Plasmodium falciparum Gametocyte EXported Protein-5 marks the gametocyte ring stage. Malar J. 2015 14:334. PMID:
See original on MMP
Effect of PTEX88 knockdown on P. falciparum protein export. Immunofluorescence assays showing that exported proteins A: RESA, B: STEVOR, C: SBP1, D: KAHRP, E: RIF50, F: PfEMP1 are still exported after knockdown of PTEX88. RESA (an early expressed PEXEL protein), STEVOR (a PEXEL protein containing transmembrane domains), skeleton binding protein 1 (SBP1; a PNEP), KAHRP (a soluble PEXEL protein), RIF50 (a PEXEL protein containing transmembrane domains) and PfEMP1 (a PNEP containing a transmembrane domain that is exported to the erythrocyte surface.Chisholm SA, McHugh E, Lundie R, Dixon MW, Ghosh S, O'Keefe M, Tilley L, Kalanon M, de Koning-Ward TF. Contrasting Inducible Knockdown of the Auxiliary PTEX Component PTEX88 in P. falciparum and P. berghei Unmasks a Role in Parasite Virulence. PLoS One. 2016 Feb 17;11(2):e0149296.
See original on MMP
GFP-PfRab1A fluorescence does not co-localize with ER/Golgi, dense granules or microneme markers. GFP-PfRab1A fluorescence is distinct from the localization of markers for the apicoplast (ACP), the ER (Bip), the Golgi (ERD2 and GRASP), as well as from staining of the ER/Golgi with Bodipy BFA. GFP-Rab1A fluorescence is also distinct from the localization of markers for dense granules (RESA) or micronemes (AMA1 and EBA175).Morse D, Webster W, Kalanon M, Langsley G, McFadden GI. Plasmodium falciparum Rab1A Localizes to Rhoptries in Schizonts. PLoS One. 2016 Jun 27;11(6):e0158174
See original on MMP
P. falciparum merozoite protein location is shown, as determined by IFA and functions: rolling on the membrane by MSPs, TJ formation mediated by AMA-1-and apical rhoptries proteins, RBC deformation and invasion by microneme EBAs, protein processing by intracytoplasmic SERA-5, iRBC membrane expression by RESA and severe malaria (SM) and echinocytosis by PfEMP-1.Patarroyo ME, Arevalo-Pinzon G, Reyes C, Moreno-Vranich A, Patarroyo MA. Malaria Parasite Survival Depends on Conserved Binding Peptides' Critical Biological Functions. Curr Issues Mol Biol. 2016;18:57-78.
See original on MMP
RhopH2 is not involved in the trafficking of exported proteins in the erythrocyte cytoplasm. (a) Representative IFAs of erythrocytes infected with RhopH2-HAglmS parasites grown in 0 mM or 2.5 mM GlcN using the indicated antibodies show trafficking of RESA, SBP1 and PfEMP1 is unaffected upon RhopH2 knockdown. Scale bar = 5 μm. No defect in the export of PfEMP1, or trafficking of either RESA to the erythrocyte membrane or SBP1 to the Maurer’s clefts was evident after knocking down RhopH2 expression with GlcN.Counihan N, Chisholm SA, Bullen HE, Srivastava A, Sanders PR, Jonsdottir TK, Weiss GE, Ghosh S, Crabb BS, Creek DJ, Gilson PR, de Koning-Ward TF. Plasmodium falciparum parasites deploy RhopH2 into the host erythrocyte to obtain nutrients, grow and replicate. Elife. 2017 Mar 2;6. pii: e23217.
See original on MMP
The expression of cytoskeletal binding proteins KAHRP is increased in ΔHsp70-x. (a) Immuno fluorescence microscopy of 16-24 hpi parasites indicates expression of the exported, knob localized KAHRP protein but not RESA appears higher in ΔHsp70-x. Antibodies to the cytoskeletal binding proteins ring exported surface antigen (RESA) and knob associated histidine rich protein (KAHRP) indicated the MFI was significantly higher for KAHRP but not RESA in the mutant. The CS2 parasite line in which the ΔHsp70-x clones were derived from stably expresses the single VAR2CSA PfEMP1 allele, which binds to chondroitin sulfate A (CSA) normally present in the placenta.Charnaud SC, Dixon MWA, Nie CQ, Chappell L, Sanders PR, Nebl T, Hanssen E, Berriman M, Chan JA, Blanch AJ, Beeson JG, Rayner JC, Przyborski JM, Tilley L, Crabb BS, Gilson PR. The exported chaperone Hsp70-x supports virulence functions for Plasmodium falciparum blood stage parasites. PLoS One. 2017 12(7):e0181656.
See original on MMP
PfEH1 and PfEH2 interact with the erythrocyte cytoskeleton. IFA of paraformaldehyde/ glutaraldehyde-fixed, trophozoite/early schizont-stage parasites expressing PfEH1-GFP (A) or PfEH2-GFP (B) under control of the respective endogenous promoter. The GFP-expressing parasites were costained with anti-RESA (red). The nuclei were stained with DAPI (blue). Bars = 5 mm. DIC, differential interference contrast. In asexual parasites, anti-GFP antibody detected PfEH1 and PfEH2 at the periphery of the infected erythrocyte and within the parasite. The fluorescence at the periphery colocalized with ring-exported surface antigen (RESA), an exported protein that interacts with host spectrin at the RBC cytoskeleton.Spillman NJ, Dalmia VK, Goldberg DE. Exported Epoxide Hydrolases Modulate Erythrocyte Vasoactive Lipids during Plasmodium falciparum Infection. MBio. 2016 Oct 18;7(5). pii: e01538-16.
See original on MMPMore information
| PlasmoDB | PF3D7_0102200 |
| GeneDB | PF3D7_0102200 |
| Malaria Metabolic Pathways | Localisation images Pathways mapped to |
| Previous ID(s) | MAL1P1.13, PFA0110w |
| Orthologs | |
| Google Scholar | Search for all mentions of this gene |